Abstract
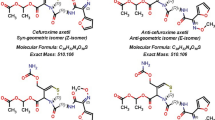
N-methyl-pyrrolidine (NMP) a potential impurity in the cephalosporin antibiotic cefepime is analysed using a rapid thermal desorption – ion mobility spectrometry (TD-IMS) method. The thermal desorption approach is shown to be capable of rapidly extracting NMP from the cefepime at 80 °C without causing thermal degradation of the cefepime. The ion mobility method has an analysis time of 1 min and demonstrates good linearity over a range from 0.3–3.0 μg ml−1 of NMP, with limits of detection and quantification of 0.056 and 0.1875 μg ml−1 respectively. The developed method was applied to the analysis of a cefepime sample and determined that NMP was present in a cefepime sample at a level of 0.0376 % with a percentage relative standard deviation (n = 6) of 3.2 %. This was compared with a LC-UV method which was in close agreement measuring NMP at 0.0384 % in the cefepime sample with a percentage RSD (n = 6) of 5.7 %. These results show that the TD-IMS method gives comparable data to the established LC methods and demonstrates the potential of TD-IMS for rapid measurement of volatile compounds in pharmaceutical matrices.






Similar content being viewed by others
References
Singh S, Handa T, Narayanam M, Sahu A, Junwal M, Shah RP (2012) A critical review on the use of modern sophisticated hyphenated tools in the characterization of impurities and degradation products. J Pharm Biomed Anal 69:148–173
Barbhaiya RH, Forgue ST, Gleason CR, Knupp CA, Pittman KA, Weidler DJ, Movahhed H, Tenney J, Martin RR (1992) Pharmacokinetics of cefepime after single and multiple intravenous administrations in healthy subjects. Antimicrob. Agents Chemother 36:552–557
Van der Auwera P, Santella PJ (1993) Pharmacokinetics of cefepime: a review. Antimicrob. Agents Chemother 32:103–115
Sprauten PF, Beringer PM, Louie SG, Synold TW, Gill MA (2003) Stability and antibacterial activity of cefepime during continuous infusion. Antimicrob. Agents Chemother 47:1991–1994
Yayan J, Ghebremedhin B, Rasche K (2016) Cefepime shows good efficacy and no antibiotic resistance in pneumonia caused by Serratia marcescens and Proteus mirabilis - an observational study. BMC Pharmacol Toxicol 17. doi:10.1186/s40360-016-0056-y
Legout L, Senneville E, Stern R, Yazdanpanah Y, Savage C, Roussel-Delvalez M, Rosele B, Migaud H, Mouton Y (2006) Treatment of bone and joint infections caused by gram-negative bacilli with a cefepime-fluoroquinolone combination. Clin Microbiol Infect 12:1030–1033
Han SB, Hae BY, Lee JW, Lee DG, Chung NG, Jeong DC, Cho B, Kang JH, Kim HK (2013) Clinical characteristics and antimicrobial susceptibilities of viridans streptococcal bacteremia during febrile neutropenia in patients with hematologic malignancies: a comparison between adults and children. BMC Infect Dis 13. doi:10.1186/1471-2334-13-273
Kumar VJ, Gupta PB, Kumar KSRP, Rao KVVP, Rao KR, Prasanna SJ, Sharma HK, Mukkanti K (2010) Identification and characterization ofnew degradation products of cefepime dihydrochloride monohydrate drug substance during stress stability studies. Anal Sci 26:1081–1086
Shahid SK (2010) Cefepime: a review of its use in the treatment of serious bacterial infections. Clin Med Rev Therapeutics 2:1–10
Viane E, Chanteux H, Servais H, Mingeout-Leclercq MP, Tulkens PM (2002) Comparative stability studies of Antipseudomonal β-lactams for potential administration through portable elastomeric pumps (home therapy for cystic fibrosis patients) and motor-operated syringes (intensive care units) Antimicrob. Agents Chemother 46:2327–2332
Fubara JO, Notari RE (1998) Influence of pH, temperature and buffers on cefepime degradation kinetics and stability predictions in aqueous solutions. J Pharm Sci 87:1572–1576
United States Pharmacopoeia, USP monograph: Cefepime for injection, USP 29 NF24, 410
United States Pharmacopoeia, USP monograph: Cefepime hydrochloride, USP 29 NF24, 409
Calahorra B, Campanero MA, Sadaba B, Azanza JR (1999) Rapid high-performance liquid chromatographic determination of cefepime in human plasma. Biomed Chromatogr 13:272–275
Page N, Stevenson R, Powell M (2014) Analysis of N-methylpyrrolidine in cefepime hydrochloride by ion chromatography using suppressed conductivity detection with solid-phase extraction pre-treatment. Anal Methods 6:1248–1253
Prasanna SJ, Sharma HK, Mukkanti K, Kumar VJ, Raja G, Sivakumaran M (2010) Validation of capillary electrophoresis method for determination of N-methylpyrrolidine in cefepime for injection. J Chromatogr Sci 48:830–834
Fardjzadeh MA, Goushjuii L, Bashour Y (2010) A simple and rapid dispersive liquid-liquid microextraction method followed by GC-FID for determination of N-methylpyrrolidine in cefepime. J Sep Sci 33:3767–3773
Smith RW, Cox LB, Yudin A, Reynolds JC, Powell M, Creaser CS (2015) Rapid determination of N-methylpyrrolidine in cefepime by combining direct infusion electrospray ionisation-time-of-flight mass spectrometry with field asymmetric waveform ion mobility spectrometry. Anal Methods 7:34–39
Borsdorf H, Eiceman GA (2006) Ion mobility spectrometry: principles and applications. Appl Spectrosc Rev 41:323–375
Armenta S, Alcala M, Blanco M, Gonzalez JM (2013) Ion mobility spectrometry for the simultaneous determination of diacetyl midecamycin and detergents in cleaning validation. J Pharm Biomed Anal 83:265–272
Strege MA (2009) Total residue analysis of swabs by ion mobility spectrometry. Anal Chem 81:4576–4580
Verkouteren J, Staymates JL (2011) Reliability of ion mobility spectrometry for qualitative analysis of complex, multicomponent illicit drug samples. Foren Sci Int 206:190–196
O’Donnell RM, Sun X, Harrington PB (2008) Pharmaceutical applications of ion mobility spectrometry. Trends Anal Chem 27:44–53
Harris GA, Graf S, Knochenmuss R, Fernandez FM (2012) Coupling laser ablation/desorption electrospray ionization to atmospheric pressure drift tube ion mobility spectrometry for the screening of antimalarial drug quality. Analyst 137:3039–3044
Roscioli KM, Tufariello JA, Zhang X, Li SX, Goetz GH, Cheng G, Siems WF, Hill HH (2014) Desorption electrospray ionization (DESI) with atmospheric pressure ion mobility spectrometry for drug detection. Analyst 139:1740–1750
Dunn JD, Gryniewicz-Ruzicka CM, Mans DJ, Mecker-Pogue LC, Kauffman JF, Westenberger BJ, Buhse LF (2012) Qualitative screening for adulterants in weight-loss supplements by ion mobility spectrometry. J Pharm Biomed Anal 71:18–26
Eckers C, Laures AMF, Giles K, Major H, Pringle S (2007) Evaluating the utility of ion mobility separation in combination with high-pressure liquid chromatography/mass spectrometry to facilitate detection of trace impurities in formulated drug products. Rapid Commun Mass Spectrom 21:1255–1263
Howdle MD, Eckers C, Laures AMF, Creaser CS (2010) The effect of drift gas on the separation of active pharmaceutical ingredients and impurities by ion mobility–mass spectrometry. Int J Mass Spectrom 298:72–77
Howdle MD, Eckers C, Laures AMF, Creaser CS (2009) The use of shift reagents in ion mobility-mass spectrometry: studies on the complexation of an active pharmaceutical ingredient with polyethylene glycol excipients. J Am Soc Mass Spectrom 20:1–9
Zamora D, Alcala M, Blanco M (2011) Determination of trace impurities in cosmetic intermediates by ion mobility spectrometry. Anal Chim Acta 708:69–74
Puton J, Nousiainen M, Sillanpaa M (2008) Ion mobility sopectrometers with doped gases. Talanta 76:978–987
Wang S, Dapeng Z, Zhang J, Li H, Liu H (2006) Rapid identification of Z/E cefepime dihydrochloride by high performance liquid chromatography / tandem time of flight mass spectrometry. Chin J Anal Chem 34(9):1278–1282
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 22 kb)
Rights and permissions
About this article
Cite this article
Reynolds, J.C., Giddings, L., Usen, I.C. et al. Rapid analysis of N-methylpyrrolidine in cefepime with thermal desorption ion mobility spectrometry. Int. J. Ion Mobil. Spec. 19, 209–217 (2016). https://doi.org/10.1007/s12127-016-0210-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12127-016-0210-7