Abstract
Background
Changes in Caveolin-1 (CAV-1) expression are related to tumorigenesis. The aim of this study was to evaluate the role of CAV-1 in tumor progression in oral squamous cell carcinoma (SCC) tissue samples and the effect of CAV-1 silencing on two oral tongue SCC (OTSCC) cell lines (SCC-25, from a primary tumor, and HSC-3 from lymph node metastases).
Methods
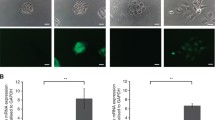
Mycroarray hybridization, mRNA expression, and immunohistochemistry were performed on OSCC tissue samples and corresponding non-tumoral margin tissues. The effects of CAV-1 silencing (siCAV-1) on cell viability, membrane fluidity, on the expression of epithelial to mesenchymal transition (EMT) markers and on cell migration and invasion capacity of OTSCC cell lines were evaluated.
Results
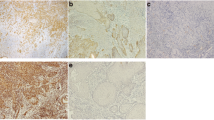
Microarray showed a greater CAV-1 expression (1.77-fold) in OSCC tumors than in non-tumoral tissues and 2.0-fold more in less aggressive OSCCs. However, significant differences in CAV-1 gene expression were not seen between tumors and non-tumoral margins nor CAV-1 with any clinicopathological parameters. CAV-1 protein was localized both in carcinoma and in spindle cells of the tumor microenvironment (TME), and CAV-1 positive TME cells were associated with smaller/more aggressive tumors, independent of the carcinoma cells’ expression. Silencing of CAV-1 increased cell viability only in SCC-25 cells. It also stimulated the invasion of HSC-3 cells and increased ECAD and BCAT mRNA in these cells; however, the protein levels of the EMT markers were not affected.
Conclusion
Decreased expression of CAV-1 by tumor cells in OSCC and an increase in the TME were associated with increased cell invasiveness and tumor aggressiveness.






Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Flavahan WA, Gaskell E, Bernstein BE (2017) Epigenetic plasticity and the hallmarks of cancer. Science. https://doi.org/10.1126/science.aal2380
Farah CS (2021) Molecular landscape of head and neck cancer and implications for therapy. Ann Transl Med. https://doi.org/10.21037/atm-20-6264
Campos A, Burgos-Ravanal R, González MF et al (2019) Cell intrinsic and extrinsic mechanisms of caveolin-1-enhanced metastasis. Biomolecules. https://doi.org/10.3390/biom9080314
Routray S (2014) Caveolin-1 in oral squamous cell carcinoma microenvironment: an overview. Tumor Biol 35:9487–9495. https://doi.org/10.1007/s13277-014-2482-z
Quest AFG, Gutierrez-Pajares JL, Torres VA (2008) Caveolin-1: an ambiguous partner in cell signalling and cancer. J Cell Mol Med 12:1130–1150. https://doi.org/10.1111/j.1582-4934.2008.00331.x
Hung K-F, Lin S-C, Liu C-J et al (2003) The biphasic differential expression of the cellular membrane protein, caveolin-1, in oral carcinogenesis. J Oral Pathol Med 32:461–467. https://doi.org/10.1034/j.1600-0714.2003.00185.x
Nakatani K, Wada T, Nakamura M et al (2005) Expression of caveolin-1 and its correlation with cisplatin sensitivity in oral squamous cell carcinoma. J Cancer Res Clin Oncol 131:445–452. https://doi.org/10.1007/s00432-004-0662-8
Zhang H, Su L, Müller S et al (2008) Restoration of caveolin-1 expression suppresses growth and metastasis of head and neck squamous cell carcinoma. Br J Cancer 99:1684–1694. https://doi.org/10.1038/sj.bjc.6604735
Vered M, Lehtonen M, Hotakainen L et al (2015) Caveolin-1 accumulation in the tongue cancer tumor microenvironment is significantly associated with poor prognosis: an in vivo and in-vitro study. BMC Cancer 15:25. https://doi.org/10.1186/s12885-015-1030-6
Auzair LBM, Vincent-Chong VK, Ghani WMN et al (2016) Caveolin 1 (Cav-1) and actin related protein 2/3 complex, subunit 1B (ARPC1B) expressions as prognostic indicators for oral squamous cell carcinoma (OSCC). Eur Arch Oto-Rhino-Laryngol 273:1885–1893. https://doi.org/10.1007/s00405-015-3703-9
Nohata N, Hanazawa T, Kikkawa N et al (2011) Caveolin-1 mediates tumor cell migration and invasion and its regulation by miR-133a in head and neck squamous cell carcinoma. Int J Oncol 38:209–217
de Morais EF, Rolim LSA et al (2020) Biological role of epithelial–mesenchymal-transition-inducing transcription factors in head and neck squamous cell carcinoma: a systematic review. Arch Oral Biol. https://doi.org/10.1016/j.archoralbio.2020.104904
Wahab A, Onkamo O, Pirinen M et al (2022) The budding and depth of invasion model in oral cancer: a systematic review and meta-analysis. Oral Dis 28:275–283. https://doi.org/10.1111/odi.13671
Beardsley A, Fang K, Mertz H et al (2005) Loss of caveolin-1 polarity impedes endothelial cell polarization and directional movement. J Biol Chem 280:3541–3547. https://doi.org/10.1074/jbc.M409040200
Grande-Garcia A, Echarri A, de Rooij J et al (2007) Caveolin-1 regulates cell polarization and directional migration through Src kinase and Rho GTPases. J Cell Biol 177:683–694. https://doi.org/10.1083/jcb.200701006
Xavier FCA, Rodini CO, Paiva KBS et al (2012) ORAOV1 is amplified in oral squamous cell carcinoma. J Oral Pathol Med 41:54–60. https://doi.org/10.1111/j.1600-0714.2011.01053.x
Severino P, Alvares AM, Michaluart P et al (2008) Global gene expression profiling of oral cavity cancers suggests molecular heterogeneity within anatomic subsites. BMC Res Notes 1:113. https://doi.org/10.1186/1756-0500-1-113
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:45e–445. https://doi.org/10.1093/nar/29.9.e45
Prosdócimi FC, Rodini CO, Sogayar MC et al (2014) Calcifying cystic odontogenic tumour: immunohistochemical expression of matrix metalloproteinases, their inhibitors (TIMPs and RECK) and inducer (EMMPRIN). J Oral Pathol Med 43:545–553. https://doi.org/10.1111/jop.12154
Koo JS, Park S, Il KS et al (2011) The impact of caveolin protein expression in tumor stroma on prognosis of breast cancer. Tumor Biol 32:787–799. https://doi.org/10.1007/s13277-011-0181-6
Salo T, Sutinen M, Hoque Apu E et al (2015) A novel human leiomyoma tissue derived matrix for cell culture studies. BMC Cancer 15:981. https://doi.org/10.1186/s12885-015-1944-z
Jaafari-Ashkavandi Z, Aslani E (2017) Caveolin-1 expression in oral lichen planus, dysplastic lesions and squamous cell carcinoma. Pathol - Res Pract 213:809–814. https://doi.org/10.1016/j.prp.2017.03.006
Jung AC, Ray A-M, Ramolu L et al (2015) Caveolin-1-negative head and neck squamous cell carcinoma primary tumors display increased epithelial to mesenchymal transition and prometastatic properties. Oncotarget 6:41884–41901. https://doi.org/10.18632/oncotarget.6099
Wahbi W, Naakka E, Tuomainen K et al (2020) The critical effects of matrices on cultured carcinoma cells: human tumor-derived matrix promotes cell invasive properties. Exp Cell Res. https://doi.org/10.1016/j.yexcr.2020.111885
Sohn J, Lin H, Fritch MR, Tuan RS (2018) Influence of cholesterol/caveolin-1/caveolae homeostasis on membrane properties and substrate adhesion characteristics of adult human mesenchymal stem cells. Stem Cell Res Ther 9:86. https://doi.org/10.1186/s13287-018-0830-4
Fu C, He J, Li C et al (2010) Cholesterol increases adhesion of monocytes to endothelium by moving adhesion molecules out of caveolae. Biochim Biophys Acta - Mol Cell Biol Lipids 1801:702–710. https://doi.org/10.1016/j.bbalip.2010.04.001
Acknowledgements
GENCAPO (Head and Neck Genome Project) Consortium for the sample collection and initial processing, clinical data collection, providing histopathological analysis of tissue samples, and the acquisition of informed consent from each patient. We declare the paper was reviewed for grammar and syntax by an English expert.
Funding
This work was supported by the National Council for Scientific and Technological Development (CNPq) [Grant No. 421249/2018–8] and Higher Education Personnel Improvement Coordination (RB Nascimento fellowship—CAPES).
Author information
Authors and Affiliations
Contributions
All authors have contributed to this article as follow: RBN performed all the in vitro experiments, analysis, data interpretation, and wrote the initial draft of the manuscript. MJ and TAS designed the in vitro experiments and data interpretation. LHSS performed IHC experiments. KBSP, MFSDR, COR, FCAX performed the microarray and qRT-PCR and their analysis of tumor samples. FCAX and FDN analysed IHC images and statistical analysis. RDC, RVML and FDN are members of GENCAPO and collected the tissue samples and clinical data. FCAX, KBSP and TAS were the advisors of RBN PhD thesis. All authors have reviewed and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
“All procedures performed in studies involving human participants were in accordance with the ethical standards of the Brazilian National Ethics Committee (Process #16491) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
For this type of study consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nascimento, R.B., Paiva, K.B.S., Risteli, M. et al. Loss of Caveolin-1 Expression in Tumor Cells is Associated with Increased Aggressiveness and Cell Invasion in Oral Squamous Cell Carcinoma. Head and Neck Pathol 17, 618–630 (2023). https://doi.org/10.1007/s12105-023-01562-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-023-01562-w