Abstract
Background
HPV-associated oral cavity squamous cell carcinoma (SCC) is not well-characterized in the literature, and also has a clinical significance that is poorly understood.
Methods
We gathered a cohort of oral cavity (OC) SCC with nonkeratinizing morphology, either in the invasive or in situ carcinoma (or both), tested for p16 by immunohistochemistry and high risk HPV E6/E7 mRNA by RTPCR (reference standard for transcriptionally-active high risk HPV) and gathered detailed morphologic and clinicopathologic data.
Results
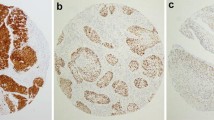
Thirteen patients from two institutions were proven to be HPV-associated by combined p16 and high risk HPV mRNA positivity. All 13 patients (100%) were males, all were heavy smokers (average 57 pack/year), and most were active drinkers (9/11 or 81.8%). All 13 (100%) involved the tongue and/or floor of mouth. All had nonkeratinizing features, but maturing squamous differentiation varied widely (0–90%; mean 37.3%). Nonkeratinizing areas had high N:C ratios and larger nests, frequently with pushing borders, and minimal (or no) stromal desmoplasia. The carcinoma in situ, when present, was Bowenoid/nonkeratinizing with cells with high N:C ratios, full thickness loss of maturation, and abundant apoptosis and mitosis. HPV was type 16 in 11 patients (84.6%) and type 33 in two (15.4%). Nine patients had treatment data available. These underwent primary surgical resection with tumors ranging from 1.6 to 5.2 cm. Most had bone invasion (6/9–66.7% were T4a tumors), and most (6/9–66.7%) had extensive SCC in situ with all 6 of these patients having final margins positive for in situ carcinoma.
Conclusions
HPV-associated OCSCC is an uncommon entity that shows certain distinct clinical and pathologic features. Recognition of these features may help pathologic diagnosis and could potentially help guide clinical management.





Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Ang KK, Harris J, Wheeler R, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363:24–35.
Fakhry C, Westra WH, Li S, et al. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst. 2008;100:261–9.
Gillison ML, Restighini C. Anticipation of the impact of human papillomavirus on clinical decision making for the head and neck cancer patient. Hematol/Oncol Clin North Am. 2015;29:1045–60.
Wagner S, Wittekindt C, Sharma SJ, et al. Human papillomavirus association is the most important predictor for surgically treated patients with oropharyngeal cancer. Br J Cancer. 2017;116:1604–11.
Chernock RD, El-Mofty SK, Thorstad WL, et al. HPV-related nonkeratinizing squamous cell carcinoma of the oropharynx: utility of microscopic features in predicting patient outcome. Head Neck Pathol. 2009;3:186–94.
Lewis JS Jr, Khan RA, Masand RP, et al. Recognition of nonkeratinizing morphology in oropharyngeal squamous cell carcinoma—a prospective cohort and interobserver variability study. Histopathology. 2012;60:427–36.
Gondim DD, Haynes W, Wang X, et al. Histologic typing in oropharyngeal squamous cell carcinoma: a 4-year prospective practice study with p16 and high-risk HPV mRNA testing correlation. Am J Surg Pathol. 2016;40:1117–24.
Bryant AK, Sojourner EJ, Vitzthum LK, et al. Prognostic role of p16 in nonoropharyngeal head and neck cancer. J Natl Cancer Inst. 2018;110:1393–9.
Chung CH, Zhang Q, Kong CS, et al. p16 Protein expression and human papillomavirus status as prognostic biomarkers of nonoropharyngeal head and neck squamous cell carcinoma. J Clin Oncol. 2014;32:3930–8.
Lingen MW, Xiao W, Schmitt A, et al. Low etiologic fraction for high-risk human papillomavirus in oral cavity squamous cell carcinomas. Oral Oncol. 2013;49:1–8.
Nauta IH, Heideman DAM, Brink A, et al. The unveiled reality of human papillomavirus as risk factor for oral cavity squamous cell carcinoma. Int J Cancer. 2021;149:420–30.
Rooper LM, Windon MJ, Hernandez T, et al. HPV-positive squamous cell carcinoma of the larynx, oral cavity, and hypopharynx: clinicopathologic characterization with recognition of a novel warty variant. Am J Surg Pathol. 2020;44:691–702.
Chernock RD. Morphologic features of conventional squamous cell carcinoma of the oropharynx: ‘keratinizing’ and ‘nonkeratinizing’ histologic types as the basis for a consistent classification system. Head Neck Pathol. 2012;6(Suppl 1):41–7.
Woo SB, Cashman EC, Lerman MA. Human papillomavirus-associated oral intraepithelial neoplasia. Mod Pathol. 2013;26:1288–97.
Lerman MA, Almazrooa S, Lindeman N, et al. HPV-16 in a distinct subset of oral epithelial dysplasia. Mod Pathol. 2017;30:1646–54.
Lewis JS Jr, Beadle B, Bishop JA, et al. Human papillomavirus testing in head and neck carcinomas: guideline from the College of American Pathologists. Arch Pathol Lab Med. 2018;142:559–97.
Gao G, Chernock RD, Gay HA, et al. A novel RT-PCR method for quantification of human papillomavirus transcripts in archived tissues and its application in oropharyngeal cancer prognosis. Int J Cancer. 2013;132:882–90.
WHO Classification of Tumours Editorial Board. Head and neck tumours. Lyon: International Agency for Research on Cancer; 2022.
McCord C, Bradley G. Histopathologic features of high risk HPV-associated oral epithelial dysplasia. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117:120–1.
McCord C, Xu J, Xu W, et al. Association of high-risk human papillomavirus infection with oral epithelial dysplasia. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;115:541–9.
Khanal S, Trainor PJ, Zahin M, et al. Histologic variation in high grade oral epithelial dysplasia when associated with high-risk human papillomavirus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2017;123:566–85.
Funding
Research was performed using discretionary funds from the Department of Pathology, Microbiology, and Immunology. The work also utilized the Translational Pathology Shared Resource (TPSR) at Vanderbilt University Medical Center which is supported by NCI/NIH Cancer Center Support Grant 5P30 CA68485-19 and the Shared Instrumentation Grant S10 OD023475. This work was supported by funds from the NIH Grant R01DE026471 (Wang). This work was also supported by the National Cancer Institute (NCI) K07CA218247 (PI: Krystle Kuhs); Vanderbilt Clinical Oncology Research Career Development Program (K12 CA090625); and the Vanderbilt Institute for Clinical and Translational Research (UL1 TR000445 from NCATS/NIH). This work was supported by funds from the NIH Grant U24 DK059637-16.
Author information
Authors and Affiliations
Contributions
All authors whose names appear on the submission made substantial contributions to the conception or design of the work, to the acquisition, analysis, or interpretation of data, and/or drafted the work or revised it critically for important intellectual content including approving the version to be published. They agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest as it relates to this work.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was performed with approval of the respective institutional review boards of Vanderbilt University Medical Center and the University of Kentucky and complies with required ethical standards. Given the retrospective nature of the study, with consultation with the institutional review boards, it was determined that the study did not need ethical approval. Patients were never contacted, and we did not require informed consent or specific consent to publish.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lewis, J.S., Smith, M.H., Wang, X. et al. Human Papillomavirus-Associated Oral Cavity Squamous Cell Carcinoma: An Entity with Distinct Morphologic and Clinical Features. Head and Neck Pathol 16, 1073–1081 (2022). https://doi.org/10.1007/s12105-022-01467-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-022-01467-0