Abstract
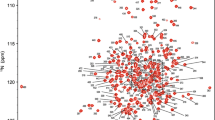
MarH is an essential epimerase that catalyzes the isomerization of 3R-β-methyl-indolepyruvate (β-MeInPy) to 3S-β-MeInPy, which is the important precursor for biosynthesis of Maremycins. Biophysical study of the structure of MarH would be informative for better understanding of its catalytic mechanism and feasible application of the enzyme in isomerization reaction. Here, we report the backbone and side-chain NMR chemical shift assignments of MarH, which lays a foundation for further structural and mechanical study of the enzyme.


Similar content being viewed by others
References
Balkbindseil W, Helmke E, Weyland H, Laatsch H (1995) Maremycin A and B, new diketopiperazines from a marine Streptomyces sp. Liebigs Ann 7:1291–1294
Chan TH, Hill RK (1970) The absolute configuration of indolmycin. J Org Chem 35:3519–3521
Chavez FA, Banerjee A, Sljivic B (2011) Modeling the metal binding site in cupin proteins. In: Pramatarova L (ed) On biomimetics. InTech, Rijeka. https://doi.org/10.5772/18262
Delaglio F, Grzesiek S, Vuister GW, Zhu G, Pfeifer J, Bax A (1995) NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J Biomol NMR 6:277–293
Dunwell JM, Gane PJ (1998) Microbial relatives of seed storage proteins: conservation of motifs in a functionally diverse superfamily of enzymes. J Mol Evol 46:147–154
Dunwell JM, Culham A, Carter CE, Sosaaguirre CR, Goodenough PW (2001) Evolution of functional diversity in the cupin superfamily. Trends Biochem Sci 26:740–746
Dunwell JM, Purvis A, Khuri S (2004) Cupins: the most functionally diverse protein superfamily? Phytochemistry 65:7–17
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ (2015) The Phyre2 web portal for protein modelling, prediction and analysis. Nat Protoc 10:845–858
Lee W, Tonelli M, Markley JL (2015) NMRFAM-SPARKY: enhanced software for biomolecular NMR spectroscopy. Bioinformatics 31:1325–1327
Sheehan JC, Mania D, Nakamura S, Stock JA, Maeda K (1968) The structure of telomycin. J Am Chem Soc 90:462–470
Ueda T, Inada M, Okamoto I, Morita N, Tamura O (2008) Synthesis of maremycins A and D1 via cycloaddition of a nitrone with (E)-3-ethylidene-1-methylindolin-2-one. Org Lett 10:2043–2046
Wilkinson B, Micklefield J (2009) Biosynthesis of nonribosomal peptide precursors. Methods Enzymol 458:353–378
Wishart DS et al (1995) 1H, 13C and 15N chemical shift referencing in biomolecular NMR. J Biomol NMR 6:135–140
Yang S, Bax A (2015) Protein structural information derived from NMR chemical shift with the neural network program TALOS-N. Methods Mol Biol 1260:17–32
Zou Y et al (2013) Stereospecific biosynthesis of β-methyltryptophan from L-tryptophan features a stereochemical switch. Angew Chem Int Ed 52:12951–12955
Acknowledgements
This work was supported by the National Key Research and Development Program of China (No. 2017YFC0906900), the National Natural Science Foundation of China (No. 21573258) and Yunnan Provincial Science and Technology Department (No. 2015FA028).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, B., Fang, S., Ma, X. et al. Backbone and side-chain chemical shift assignments of MarH, a critical intermediary epimerase for biosynthesis of Maremycins in Streptomyces. Biomol NMR Assign 12, 335–338 (2018). https://doi.org/10.1007/s12104-018-9835-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-018-9835-2