Abstract
Objectives
To assess the effect of the long-term use of inhaled corticosteroids (ICS) on the hypothalamic-pituitary-adrenal (HPA) axis.
Methods
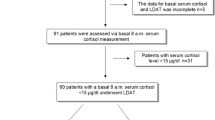
Children (5–18 y) diagnosed with asthma and on ICS therapy for ≥6 mo were included. In the first step, screening with fasting at 8 AM, cortisol level was measured; a value <15 mcg/dl was considered low. Children with low fasting cortisol levels were subjected to adreno-corticotropic hormone (ACTH) stimulation test in the second step. Post-ACTH stimulation, cortisol level <18 mcg/dl was considered to have HPA axis suppression.
Results
A total of 78 children (males 55, 70.5%) diagnosed with asthma, with a median age of 11.5 (8, 14) y, were enrolled. The median duration of ICS use was 12 (12–24) mo. The median value of post-ACTH stimulation cortisol level was 22.5 (20.6, 25.5) mcg/dl, and a value <18 mcg/dl was observed in 4 (5.1%; 95% CI 0.2–10%) children. There was statistically no significant correlation between low post-ACTH stimulation cortisol level with ICS dose (p = 0.23) and asthma control (p = 0.67). None of the children had clinical features of adrenal insufficiency.
Conclusions
In this study, a few children had low post-ACTH stimulation cortisol values; however, none had clinical evidence of HPA axis suppression. Therefore, ICS is a safe drug in children for treating asthma, even for long-term use.



Similar content being viewed by others
References
Asher MI, Rutter CE, Bissell K, et al; Global Asthma Network Phase I Study Group. Worldwide trends in the burden of asthma symptoms in school-aged children: global Asthma Network Phase I cross-sectional study. Lancet. 2021;398:1569–80.
Singh S, Sharma BB, Sharma SK, Sabir M, Singh V; ISAAC Collaborating Investigators. Prevalence and severity of asthma among indian school children aged between 6 and 14 years: Associations with parental smoking and traffic pollution. J Asthma. 2016;53:238–44.
Global Initiative for Asthma (GINA). GINA-2019-main-report-June-2019-wms.pdf. Available at: https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf. Accessed on 27th July 2022.
Roland NJ, Bhalla RK, Earis J. The local side effects of inhaled corticosteroids: current understanding and review of the literature. Chest. 2004;126:213–9.
Chmielewska M, Akst LM. Dysphonia associated with the use of inhaled corticosteroids. Curr Opin Otolaryngol Head Neck Surg. 2015;23:255–9.
Buhl R. Local oropharyngeal side effects of inhaled corticosteroids in patients with asthma. Allergy. 2006;61:518–26.
Pandya D, Puttanna A, Balagopal V. Systemic effects of inhaled corticosteroids: an overview. Open Respir Med J. 2014;8:59–65.
Lipworth BJ. Systemic adverse effects of inhaled corticosteroid therapy: a systematic review and meta-analysis. Arch Intern Med. 1999;159:941–55.
Kapadia CR, Nebesio TD, Myers SE, et al. Drugs and therapeutics Committee of the Pediatric Endocrine Society. Endocrine effects of inhaled corticosteroids in children. JAMA Pediatr. 2016;170:163–70.
Casale TB, Nelson HS, Stricker WE, Raff H, Newman KB. Suppression of hypothalamic-pituitary-adrenal axis activity with inhaled flunisolide and fluticasone propionate in adult asthma patients. Ann Allergy Asthma Immunol. 2001;87:379–85.
Jia CE, Zhang HP, Lv Y, et al. The Asthma Control Test and Asthma Control Questionnaire for assessing asthma control: systematic review and meta-analysis. J Allergy Clin Immunology. 2013;131:695–703.
Cavkaytar O, Vuralli D, Arik Yilmaz E, et al. Evidence of hypothalamic-pituitary-adrenal axis suppression during moderate-to-high-dose inhaled corticosteroid use. Eur J Pediatr. 2015;174:1421–31.
Atluri S, Sarathi V, Goel A, et al. Long-acting porcine sequence ACTH (Acton prolongatum) stimulation test is a reliable alternative test as compared to the gold standard insulin tolerance test for the diagnosis of adrenal insufficiency. Indian J Endocrinol Metab. 2022;26:38–43.
Atteih S, Ratner J. Endocrinology. In: Klienman K, Mcdaniel L, Molloy M, editors. The Harriet Lane Handbook; a Manual for Pediatric House officers. 22nd ed. Philadelphia: Elsevier; 2021. p. 316–57.
Choi IS, Sim D-W, Kim S-H, Wui J-W. Adrenal insufficiency associated with long-term use of inhaled steroid in asthma. Ann Allergy Asthma Immunol. 2017;118:66–72.e1.
Gundgurthi A, Garg MK, Dutta MK, Pakhetra R. Intramuscular ACTH stimulation test for assessment of adrenal function. J Assoc Physicians India. 2013;61:320–4.
Nair A, Jayakumari C, George GS, et al. Long acting porcine sequence ACTH in the diagnosis of adrenal insufficiency. Eur J Endocrinol. 2019;181:639–45.
Kwda A, Gldc P, Baui B, et al. Effect of long term inhaled corticosteroid therapy on adrenal suppression, growth and bone health in children with asthma. BMC Pediatr. 2019;19:411.
Smith RW, Downey K, Gordon M, et al. Prevalence of hypothalamic-pituitary-adrenal axis suppression in children treated for asthma with inhaled corticosteroid. Paediatr Child Health. 2012;17:e34–9.
Dorsey MJ, Cohen LE, Phipatanakul W, Denufrio D, Schneider LC. Assessment of adrenal suppression in children with asthma treated with inhaled corticosteroids: use of dehydroepiandrosterone sulfate as a screening test. Ann Allergy Asthma Immunol. 2006;97:182–6.
Rao Bondugulapati LN, Rees DA. Inhaled corticosteroids and HPA axis suppression: how important is it and how should it be managed? Clin Endocrinol. 2016;85:165–9.
Acknowledgements
The authors thank Professor Elizabeth Juniper, McMaster University, for permitting them to use Hindi version of the Asthma Control Questionnaire (ACQ).
Author information
Authors and Affiliations
Contributions
PK & JPG: Conceptualized the idea, conducted a literature search, and prepared the initial manuscript; LSH: Enrolled the patients, did data collection, did a literature search, and wrote the initial draft; VV & KS: Literature search and prepared the initial draft. The manuscript was critically reviewed and approved for publication by all authors. PK will act as a guarantor for this manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hema, L.S., Kumar, P., Goyal, J.P. et al. Effect of Long-term Inhaled Corticosteroids on the Hypothalamic-Pituitary-Adrenal Axis in Children with Asthma. Indian J Pediatr 91, 441–447 (2024). https://doi.org/10.1007/s12098-023-04706-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-023-04706-6