Abstract
Objectives
To describe the clinical presentations and molecular diagnosis to aid the clinicians in early diagnosis and appropriate management of Prader-Willi syndrome (PWS).
Methods
Thirty-four clinically diagnosed PWS cases were enrolled after obtaining informed consent/assent. Demographic details, clinical data and anthropometry were recorded using structured proforma. The facial dysmorphology was evaluated. Appropriate genetic testing was performed to confirm the diagnosis.
Results
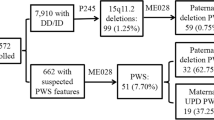
At diagnosis, the most common clinical features included obesity (59%) and short stature (53%). Distinct dysmorphic features were observed in 67%. Neonatal hypotonia with feeding difficulty, delayed development in infancy and childhood behavioral problems were reported in 94%, 94% and 74% respectively. Food seeking behavior and hyperphagia was reported in 67%. Seizures were reported in 47%. All children had underdeveloped external genitalia. Growth hormone (GH) deficiency and impaired glucose tolerance were found in 56% and 50% respectively. Sleep related problems were seen in 67%. Skin and rectal picking were reported in 67%. FISH confirmed micro-deletion was found in 64.7% and abnormal methylation in 35%, of which uniparental disomy was confirmed in 14.7%.
Conclusions
Clinical suspicion is vital for early detection of PWS. Confirmation of the diagnosis requires complex multi-tier molecular genetic testing.




Similar content being viewed by others
References
National Library of Medicine (US). Genetics Home Reference [Internet]. Bethesda (MD): The Library. Prader-Willi syndrome; [reviewed 2014 Jun]; Available at: https://ghr.nlm.nih.gov/condition/prader-willi-syndrome (2016). Accessed on 16 Apr 2016.
Butler MG. Prader-Willi syndrome: obesity due to genomic imprinting. Curr Genomics. 2011;12:204–15.
Angulo MA, Butler MG, Cataletto ME. Prader-Willi syndrome: a review of clinical, genetic, and endocrine findings. J Endocrinol Investig. 2015;38:1249–63.
McCann KL, Baserga SJ. Long noncoding RNAs (lncs) as sinks in Prader Willi syndrome. Mol Cell. 2012;48:155–7.
Khadilkar VV, Khadilkar AV, Cole TJ, Sayyad MG. Cross-sectional growth curves for height, weight and body mass index for affluent Indian children, 2007. Indian Pediatr. 2009;46:477–89.
Shawky RM, el Sedfy HH, Zaki OK, Mahmoud HM. Phenotypic expression of Egyptian patients with Prader-Willi syndrome. Egyptian J Med Hum Genet. 2001;2:55–66.
Gunay-Aygun M, Schwartz S, Heeger S, Riordan MAO, Cassidy SB. The changing purpose of Prader-Willi syndrome. Clinical diagnostic criteria and proposed revised criteria. Pediatrics [Internet]. 2001;108:1–5. Available at: http://pediatrics.aappublications.org/content/pediatrics/108/5/e92.full.pdf. Accessed on 16th April 2016.
Vendrame M, Maski KP, Chatterjee M, et al. Epilepsy in Prader-Willi syndrome: clinical characteristics and correlation to genotype. Epilepsy Behav. 2010;19:306–10.
Verrotti A, Soldani C, Laino D, d'Alonzo R, Grosso S. Epilepsy in Prader-Willi syndrome: clinical, diagnostic and treatment aspects. World J Pediatr. 2014;10:108–13.
Cassidy SB. Prader-Willi syndrome. J Med Genet. 1997;34:917–23.
Curfs LM, Fryns JP. Prader-Willi syndrome: a review with special attention to the cognitive and behavioral profile. Birth Defects Orig Artic Ser. 1992;28:99–104.
Purtell L, Sze L, Loughnan G, et al. In adults with Prader-Willi syndrome, elevated ghrelin levels are more consistent with hyperphagia than high PYY and GLP-1 levels. Neuropeptides. 2011;45:301–7.
Cummings DE, Clement K, Purnell JQ, et al. Elevated plasma ghrelin levels in Prader Willi syndrome. Nat Med. 2002;8:643–4.
Irizarry KA, Bain J, Butler MG, et al. Metabolic profiling in Prader-Willi syndrome and nonsyndromic obesity: sex differences and the role of growth hormone. Clin Endocrinol. 2015;83:797–805.
Deal CL, Tony M, Höybye C, Allen DB, Tauber M, Christiansen JS; 2011 Growth hormone in Prader-Willi syndrome clinical care guidelines workshop participants. Growth hormone research society workshop summary: consensus guidelines for recombinant human growth hormone therapy in Prader-Willi syndrome. J Clin Endocrinol Metab. 2013;98:E1072–87.
Acknowledgements
The authors wish to acknowledge Dr. Asha Benakappa, Director, IGICH for her support provided in the management of these children. They are thankful to the children and their parents who participated in this study.
Author information
Authors and Affiliations
Contributions
GNS was involved in the care of these children and also designing, data collection, analyzing and interpreting the data and writing the manuscript. MM was involved in the care of these children and designing and data collection. RKM was involved in the data processing and analysis. HK is involved in the care of these children and data collection. PSK and JSK were involved in the molecular testing and contributed in writing the manuscript. UH and EL were involved in the methylation testing and contributed in writing the manuscript. PR was involved in the care of these children and also designing, data collection, analyzing and interpreting the data and edited the manuscript. MB was involved in the care of these children and also designing, data collection, analyzing and interpreting the data and writing the manuscript. She has overseen the research and will also be the guarantor of the paper. All authors have reviewed the manuscript. GNS will act as guarantor for the paper.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Source of Funding
None.
Rights and permissions
About this article
Cite this article
Sanjeeva, G.N., Maganthi, M., Kodishala, H. et al. Clinical and Molecular Characterization of Prader-Willi Syndrome. Indian J Pediatr 84, 815–821 (2017). https://doi.org/10.1007/s12098-017-2386-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-017-2386-1