Abstract
Objective
To analyse response to growth hormone therapy on Indian patients with short stature.
Methods
Data were collected on 71 patients of short stature on GHT. All patients underwent clinical and hormonal evaluation. GHD was diagnosed in the presence of short stature (height SDS < 2) and peak GH levels < 10 ng/ml. Bone age was estimated using Tanner Whitehouse 3 method (TW3).
Results
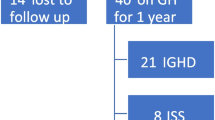
Primary GHD (73%) was the commonest diagnosis among patients on GHT, followed by organic GHD (12.6%), genetic syndromes (8.4%) and systemic diseases (5.4%). Mean chronological age at presentation was 10.07±3.26 years (median-11 years, range 3–15 years), mean height age was 6.98±2.82 years (median 7.5 years, range 1–13 years) and mean bone age (available for 55 patients) was 7.19±3.1 years (median 8.2 years, range 1.3–13 years). Patients with systemic diseases (6.75±3.5 years) presented earlier, compared to patients with GHD (10.27±3.16 years) and genetic syndromes (10.18±3.20 years) (p=0.349). Most of the patients on GHT were in the age group 9–15 years (60.6%). Mean height gain with GHT was 8.7±2.7 cm (median 8.3 cm, range 3.0–13cm) during 1st year then decreased to 6.9±2.4 cm (median 7.0 cm, range 3.0–12.5 cm) in the second year, and was maintained through the third year (mean 7.1±3.0 cm, median 7.0, range 3.0–13 cm). Among patients with GHD, those with primary deficiency had significantly better response to GHT in 1st year than secondary deficiency (9.0±2.65 vs 6.8±3.03 cm, p = 0.026). Response to GHT was negatively correlated with CA (r-0.27, p = 0.05), HA (r-0.47, p = 0.027) and BA (r-0.31, p=0.022) at presentation. Four patients (5.6%) developed hypothyroidism and one patient each developed disseminated tuberculosis and rickets. One patient of Turner’s syndrome died of adrenal carcinoma. Short follow up and absence of measurement of IGF-1 and IGFBP3 were major limitations of this study.
Conclusions
Response to GHT in Indian patients is comparable to western counterparts. Maximum height gain on GHT is during the first year than decreases in second year, but is maintained through third year. Patients with primary GHD had better response than secondary GHD. Response to GHT is negatively correlated with chronological, height and bone age at presentation.
Similar content being viewed by others
References
B Bakker, J Frane, M Anhall, B Lippe, Rosenfeld RG. Height velocity from the National cooperative growth study for first year growth hormone responses in short children. J Clin Endocrinol Metab 2008; 93: 352–357.
Quigley CA. Growth hormone treatment of Non Growth Hormone deficient Growth disorders. Endocrinol Metab Clin N Am 2007; 36: 131–186.
Kannan V, Usharani K. Human Growth Hormone Therapy: Long term responses in 30 children with Growth Hormone deficiency. Indian J Pediatr (Suppl) 1991; 58: 65–69.
Khadilkar VV, Khadilkar AV, Nandy M, Maskati GB. Multicentric study of Efficacy and Safety of Growth Hormone use in Growth Hormone Deficient Children in India. Indian J Pediatr 2007; 74: 51–54.
Bajpai A, Kabra M, Gupta AK, Menon PSN. Growth pattern and Skeletal maturation following growth hormone Therapy in growth hormone deficiency: factors influencing Outcome. Indian Pediatr 2006; 43: 593–599.
Khadilkar VV, khadilkar AV, Nandy M, Maskati GB. Growth hormone in Turner Syndrome. Indian Pediatr 2006;43: 236–240.
Menon PSN, Virmani A, Sethi AK. Biosynthetic growth hormone therapy in children with growth hormone deficiency: Experience at AIIMS, New Delhi. Indian J Pediatr (Suppl) 1991; 58: 71–77.
Consensus guidelines for the diagnosis and treatment of Growth hormone deficiency in childhood and adolescence: Summary statement of the GH research society. J Clin Endocrinol Metab 2000; 85: 3990–3993.
Tanner JM, Whitehouse RH, Cameron N, Marshall WA, Healy MJR, Goldstein H.A. Assessment of skeletal maturity and prediction of adult height(TW3 method). Pub WB Saunders, 3rd ed. 2001; 1–107.
Khadilkar VV, Khadilkar AV, Choudhury P, Agarwal KN, Ugra D, Shah NK. IAP growth monitoring guidelines for children from birth to 18 years. Indian Pediatr 2007;44: 187–197.
Wilton P, Wallstrom A. An overview of the diagnosis in the Kabi Pharmacia International Growth Study. Acta Paediatr 2008; 80: 93–98.
Price DA, Johnston DI, Bettr PR, Buckler JMH, Donaldson MDC. Biosynthetic human growth hormone treatment in the UK. An audit of current practice (Kabi Pharmacia International Growth Study) Arch Dis Child 1994; 71: 266–271.
Berghe GVD, Zegher FD, Veldhuis JD et al. The Somatotropic Axis in Critical Illness: Effect of Continuous Growth Hormone (GH)-Releasing Hormone and GH-Releasing Peptide-2 Infusion. J Clin Endocrinol Metab 1997; 82: 590–599.
Marwaha RK, Tandon N, Reddy D. Vitamin D and bone mineral density status of healthy school children in Northern India. Am J Clin Nutrition 2005; 82: 477–482.
Czernichow P, Albertson-Wikland K, Tuvemo T, Gunnarson R. Growth hormone treatment and diabetes: Survey of Kabi Pharmacia International Growth Study. Acta Pediatrica 2008; 80: 104–107.
Bolar K, Hoffman AR, Maneatis T, Lipe B. Long term safety of recombinant human growth hormone in Turner’s syndrome. J Clin Endocrinol Metab 2008; 93: 344–351.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garg, M.K., Pakhetra, R., Dutta, M.K. et al. Response to growth hormone therapy in Indian patients. Indian J Pediatr 77, 639–642 (2010). https://doi.org/10.1007/s12098-010-0090-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-010-0090-5