Abstract
Objective
To investigate the relationship between surfactant replacement therapy and the development of a haemodynamically significant ductus arteriosus.
Methods
All premature infants at 28–32 wk gestation with a clinical diagnosis of respiratory distress syndrome were retrospectively reviewed and subdivided into two groups; intubated, mechanically ventilated and received surfactant (Group 1); and received nasal continuous positive airway pressure alone (Group 2). The relationship between groups and characteristics of the hemodynamically significant ductus arteriosus was analyzed.
Results
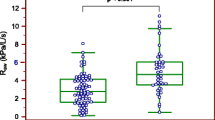
Seventy babies were identified of whom 35 (50%) received surfactant. Babies in group I and II were comparable for gestational age, birthweight, antenatal steroids, gender and fluid intake in first week of life. Babies treated with surfactant therapy were found to be more likely to have a haemodynamically significant ductus arteriosus (p<0.01), larger transductal diameter (p=0.01) and increased rate of therapeutic interventions to close the ductus (p<0.01). Ventilation parameters (mean airway pressure and fractional inspired oxygen) were higher in group I.
Conclusion
Neonates with respiratory distress syndrome (RDS) who were treated with surfactant replacement are at increased risk of a hemodynamically significant ductus arteriosus that requires therapeutic intervention. Whether the relationship reflects their underlying lung disease or is a direct effect of surfactant requires prospective evaluation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Vidyasagar D, Maeta H, Raju TNK, John E, Bhat R, Go M. Bovine surfactant therapy in immature baboons with hyaline membrane disease. Pediatrics 1985; 75: 1132–1142.
Soll RF. Prophylactic synthetic surfactant for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 1998; 2: CD001079. DOI: 10.1002/14651858.CD001079.
Soll RF. Prophylactic natural surfactant extract for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 1997; 4: CD000511. DOI: 10.1002/14651858.CD000511.
Stevens TP, Blennow M, Soll RF. Early surfactant administration with brief ventilation vs selective surfactant and continued mechanical ventilation for preterm infants with or at risk for respiratory distress syndrome. Cochrane Database Syst Rev 2004; 3: CD003063
Hailey GC, Stenson BJ, Laing IA, McIntosh M. Acute blood pressure response to surfactant administration. Arch Dis Child 1995; 73: 197.
Kappa P, Seppanen M, Kero P, Saraste M. Pulmonary hemodynamics after synthetic surfactant replacement in neonatal respiratory distress syndrome. J Pediatr 1993; 123: 115–119.
Clyman RI, Jobe A, Heymann M, Ikegami M, Roman C, Payne B, Mauray F. Increased shunt through the patent ductus arteriosus after surfactant replacement therapy. J Pediatr 1982; 100: 101–107.
Halliday HL, McCord FB, McClure BG, Reid M McC. Acute effects of instillation of surfactant in severe respiratory distress syndrome. Arch Dis Child 1989; 64: 13–16.
Bloom BT, Clark RH for the Infasurf Survanta Clinical Trial Group. Comparison of Infasurf (Calfactant) and Survanta (Beractant) in the Prevention and Treatment of Respiratory Distress Syndrome. Pediatrics 2005; 116: 392–399.
Stevens TP, Sinkin RA. Surfactant Replacement Therapy. Chest 2007; 131:1577–1582.
Smallhorn JF, Gow R, Olley PM, Freedom RM, Swyer PR, Perlman M, Rowe RD. Combined noninvasive assessment of the patent ductus arteriosus in the preterm infant before and after indomethacin treatment. Am J Cardiol 1984; 54: 1300–1304.
Sehgal A, McNamara PJ. Does point of care functional echocardiography enhance cardiovascular care in the NICU? J Perinatol 2008; 28: 729–735.
Fujiwara T, Chida S, Watabe Y, Maeta H, Morita T, Abe T. Artificial surfactant therapy in hyaline-membrane disease. Lancet 1980; 1: 55–59.
Jorch G, Rabe H, Garbe M, Michel E, Gortner L. Acute and protracted effects of intratracheal surfactant application on internal carotid blood flow velocity, blood pressure and carbon dioxide tension in very low birth weight infants. Eur J Pediatr 1989; 148: 770–773.
Sehgal A, Mak W, Dunn M, Kelly E, Redington A, Whyte HE et al. Impact of prophylactic surfactant on the ductus arteriosus and hemodynamics. Coronary artery perfusion is associated with impaired diastolic dysfunction in preterm infants after PDA ligation. Paediatric Academic Societies Annual Meeting 2008. Abstract 5842.23.1.
Van Bel F, de Winter PJ, Wijnands HBG, van de Bor M, Egberts J. Cerebral and aortic blood flow velocity patterns in preterm infants receiving prophylactic surfactant treatment. Acta Paediatr 1992; 81: 504–510.
Heldt GP, Pesonen E, Merritt A, Elias W, Shan DJ. Closure of the ductus arteriosus and mechanics of breathing in preterm infants after surfactant replacement therapy. Pediatr Res 1989; 25: 305–310.
Gonzalez A, Sosenko IRS, Chandar J, Hummler H, Claure N, Bancalari E. Influence of infection on patent ductus arteriosus and chronic lung disease in premature infants weighing 1000 grams or less. J Pediatr 1996; 128: 470–478.
McCurnin D, Seidner S, Chang LY, Waleh N, Ikegami M, Petershack J et al. Ibuprofen-Induced Patent Ductus Arteriosus Closure: Physiologic, Histologic, and Biochemical Effects on the Premature Lung. Pediatrics 2008; 121: 945–956.
Reller MD, Ziegler ML, Rice MJ, Solin RC, McDonald RW. Duration of ductal shunting in healthy preterm infants: An echocardiographic color flow Doppler study. J Pediatr 1988; 112: 441–446.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Lakkundi, A., McNamara, P.J. et al. Surfactant and patent ductus arteriosus. Indian J Pediatr 77, 51–55 (2010). https://doi.org/10.1007/s12098-009-0299-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-009-0299-3