Abstract
Background
Patients with pancreatic cancer have a dismal prognosis due to tumor cell infiltration and metastasis. Many reports have documented that EMT and PI3K–AKT–mTOR axis control pancreatic cancer cell infiltration and metastasis. Chloroxine is an artificially synthesized antibacterial compound that demonstrated anti-pancreatic cancer effects in our previous drug-screening trial. We have explored the impact of chloroxine on pancreatic cancer growth, infiltration, migration, and apoptosis.
Methods
The proliferation of pancreatic cancer cell lines (PCCs) treated with chloroxine was assessed through real-time cell analysis (RTCA), colony formation assay, CCK-8 assay, as well as immunofluorescence. Chloroxine effects on the infiltrative and migratory capacities of PCCs were assessed via Transwell invasion and scratch experiments. To assess the contents of EMT- and apoptosis-associated proteins in tumor cells, we adopted Western immunoblotting as well as immunofluorescence assays, and flow cytometry to determine chloroxine effects on PCCs apoptosis. The in vivo chloroxine antineoplastic effects were explored in nude mice xenografts.
Results
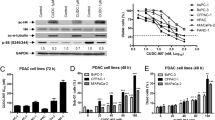
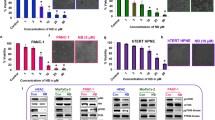
Chloroxine repressed pancreatic cancer cell growth, migration, and infiltration in vitro, as well as in vivo, and stimulated apoptosis of the PCCs. Chloroxine appeared to inhibit PCC growth by Ki67 downregulation; this targeted and inhibited aberrant stimulation of the PI3K–AKT–mTOR signaling cascade, triggered apoptosis in PCC via mitochondria-dependent apoptosis, and modulated the EMT to inhibit PCC infiltration and migration.
Conclusions
Chloroxine targeted and inhibited the PI3K–AKT–mTOR cascade to repress PCCs growth, migration, as well as invasion, and triggered cellular apoptosis. Therefore, chloroxine may constitute a potential antineoplastic drug for the treatment of pancreatic cancer.






Similar content being viewed by others
Data availability
Available from the corresponding author upon reasonable request.
Abbreviations
- AKT:
-
Protein kinase B
- AVMA:
-
American Veterinary Medical Association
- BCL2:
-
B cell lymphoma-2
- BAX:
-
BCL2-associated X
- CCK-8:
-
Cell counting Kit-8
- DAPI:
-
4′6-Diamidino-2-phenylindole
- DMEM:
-
Dulbecco modified Eagle medium
- ECM:
-
Extracellular matrix
- EMT:
-
Epithelial–mesenchymal transition
- FBS:
-
Fetal bovine serum
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- JAK2:
-
Janus kinase 2
- KEAP1:
-
Kelch-like ECH-associated protein 1
- MMP-2:
-
Matrix metalloproteinase 2
- MMP-9:
-
Matrix metalloproteinase 9
- mTOR:
-
Mammalian target of rapamycin
- PBS:
-
Phosphate-buffered saline
- PCCs:
-
Pancreatic cancer cell lines
- PI3K:
-
Phosphatidylinositol 3 kinase
- RTCA:
-
Real-time cell analysis
- SHH:
-
Sonic Hedgehog
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Escorcia FE, Houghton JL, Abdel-Atti D, Pereira PR, Cho A, Gutsche NT, et al. ImmunoPET predicts response to met-targeted radioligand therapy in models of pancreatic cancer resistant to met kinase inhibitors. Theranostics. 2020;10:151–65.
Su D, Guo X, Huang L, Ye H, Li Z, Lin L, et al. Tumor-neuroglia interaction promotes pancreatic cancer metastasis. Theranostics. 2020;10:5029–47.
Wang W, Zhan L, Guo D, Xiang Y, Zhang Y, Tian M, et al. Transcriptome analysis of pancreatic cancer cell response to treatment with grape seed proanthocyanidins. Oncol Lett. 2019;17:1741–9.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30.
McGuigan A, Kelly P, Turkington RC, Jones C, Coleman HG, McCain RS. Pancreatic cancer: a review of clinical diagnosis, epidemiology, treatment and outcomes. World J Gastroenterol. 2018;24:4846–61.
Melisi D, Garcia-Carbonero R, Macarulla T, Pezet D, Deplanque G, Fuchs M, et al. Galunisertib plus gemcitabine vs gemcitabine for first-line treatment of patients with unresectable pancreatic cancer. Br J Cancer. 2018;119:1208–14.
Murphy JE, Wo JY, Ryan DP, Jiang W, Yeap BY, Drapek LC, et al. Total neoadjuvant therapy with FOLFIRINOX followed by individualized chemoradiotherapy for borderline resectable pancreatic adenocarcinoma: a phase 2 clinical trial. JAMA Oncol. 2018;4:963–9.
Tu M, Li H, Lv N, Xi C, Lu Z, Wei J, et al. Vasohibin 2 reduces chemosensitivity to gemcitabine in pancreatic cancer cells via Jun proto-oncogene dependent transactivation of ribonucleotide reductase regulatory subunit M2. Mol Cancer. 2017;16:66.
Chinese Pancreatic Surgery Association CSoSCMA. Guidelines for the diagnosis and treatment of pancreatic cancer in China (2021). Zhonghua Wai Ke Za Zhi. 2021;59:561–77.
Dhir M, Zenati MS, Hamad A, Singhi AD, Bahary N, Hogg ME, et al. FOLFIRINOX versus gemcitabine/nab-paclitaxel for neoadjuvant treatment of resectable and borderline resectable pancreatic head adenocarcinoma. Ann Surg Oncol. 2018;25:1896–903.
Macedo FI, Ryon E, Maithel SK, Lee RM, Kooby DA, Fields RC, et al. Survival outcomes associated with clinical and pathological response following neoadjuvant FOLFIRINOX or gemcitabine/nab-paclitaxel chemotherapy in resected pancreatic cancer. Ann Surg. 2019;270:400–13.
Weng CC, Hsieh MJ, Wu CC, Lin YC, Shan YS, Hung WC, et al. Loss of the transcriptional repressor TGIF1 results in enhanced Kras-driven development of pancreatic cancer. Mol Cancer. 2019;18:96.
Zhou P, Li B, Liu F, Zhang M, Wang Q, Liu Y, et al. The epithelial to mesenchymal transition (EMT) and cancer stem cells: implication for treatment resistance in pancreatic cancer. Mol Cancer. 2017;16:52.
Zeisberg M, Neilson EG. Biomarkers for epithelial-mesenchymal transitions. J Clin Invest. 2009;119:1429–37.
Sisto M, Ribatti D, Lisi S. ADAM 17 and epithelial-to-mesenchymal transition: the evolving story and its link to fibrosis and cancer. J Clin Med. 2021;10:3373.
Sun H, Gao D. Propofol suppresses growth, migration and invasion of A549 cells by down-regulation of miR-372. BMC Cancer. 2018;18:1252.
Hua H, Kong Q, Zhang H, Wang J, Luo T, Jiang Y. Targeting mTOR for cancer therapy. J Hematol Oncol. 2019;12:71.
Mossmann D, Park S, Hall MN. mTOR signalling and cellular metabolism are mutual determinants in cancer. Nat Rev Cancer. 2018;18:744–57.
Guo Y, Zhu H, Xiao Y, Guo H, Lin M, Yuan Z, et al. The anthelmintic drug niclosamide induces GSK-beta-mediated beta-catenin degradation to potentiate gemcitabine activity, reduce immune evasion ability and suppress pancreatic cancer progression. Cell Death Dis. 2022;13:112.
Guo Y, Xiao Y, Guo H, Zhu H, Chen D, Wang J, et al. The anti-dysenteric drug fraxetin enhances anti-tumor efficacy of gemcitabine and suppresses pancreatic cancer development by antagonizing STAT3 activation. Aging (Albany NY). 2021;13:18545–63.
Silva VL, Saxena J, Nicolini F, Hoare JI, Metcalf S, Martin SA, et al. Chloroxine overrides DNA damage tolerance to restore platinum sensitivity in high-grade serous ovarian cancer. Cell Death Dis. 2021;12:395.
Paulis YW, Soetekouw PM, Verheul HM, Tjan-Heijnen VC, Griffioen AW. Signalling pathways in vasculogenic mimicry. Biochim Biophys Acta. 2010;1806:18–28.
Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525–30.
Mittal V. Epithelial mesenchymal transition in tumor metastasis. Annu Rev Pathol. 2018;13:395–412.
Boice A, Bouchier-Hayes L. Targeting apoptotic caspases in cancer. Biochim Biophys Acta Mol Cell Res. 2020;1867: 118688.
Keller N, Ozmadenci D, Ichim G, Stupack D. Caspase-8 function, and phosphorylation, in cell migration. Semin Cell Dev Biol. 2018;82:105–17.
Vincent A, Herman J, Schulick R, Hruban RH, Goggins M. Pancreatic cancer. Lancet. 2011;378:607–20.
Georgiadou D, Sergentanis TN, Sakellariou S, Vlachodimitropoulos D, Psaltopoulou T, Lazaris AC, et al. Prognostic role of sex steroid receptors in pancreatic adenocarcinoma. Pathol Res Pract. 2016;212:38–43.
Ramacciato G, Nigri G, Petrucciani N, Pinna AD, Ravaioli M, Jovine E, et al. Pancreatectomy with mesenteric and portal vein resection for borderline resectable pancreatic cancer: multicenter study of 406 patients. Ann Surg Oncol. 2016;23:2028–37.
Slapak EJ, Duitman J, Tekin C, Bijlsma M, Spek CA. Matrix metalloproteases in pancreatic ductal adenocarcinoma: key drivers of disease progression? Biology (Basel). 2020;9:80.
Solano-Galvez SG, Abadi-Chiriti J, Gutierrez-Velez L, Rodriguez-Puente E, Konstat-Korzenny E, Alvarez-Hernandez D, et al. Apoptosis: activation and inhibition in health and disease. Med Sci (Basel). 2018;6:54.
Bratton SB, MacFarlane M, Cain K, Cohen GM. Protein complexes activate distinct caspase cascades in death receptor and stress-triggered apoptosis. Exp Cell Res. 2000;256:27–33.
Li P, Nijhawan D, Wang X. Mitochondrial activation of apoptosis. Cell. 2004;116:S57-59 (52 p following S59).
Cain K, Bratton SB, Cohen GM. The Apaf-1 apoptosome: a large caspase-activating complex. Biochimie. 2002;84:203–14.
Zhang D, Zhang Q, Zheng Y, Lu J. Anti-breast cancer and toxicity studies of total secondary saponin from anemone raddeana rhizome on MCF-7 cells via ROS generation and PI3K/AKT/mTOR inactivation. J Ethnopharmacol. 2020;259: 112984.
Pungsrinont T, Kallenbach J, Baniahmad A. Role of PI3K-AKT-mTOR pathway as a pro-survival signaling and resistance-mediating mechanism to therapy of prostate cancer. Int J Mol Sci. 2021;22:11088.
Karimi Roshan M, Soltani A, Soleimani A, Rezaie Kahkhaie K, Afshari AR, Soukhtanloo M. Role of AKT and mTOR signaling pathways in the induction of epithelial-mesenchymal transition (EMT) process. Biochimie. 2019;165:229–34.
Mohan CD, Srinivasa V, Rangappa S, Mervin L, Mohan S, Paricharak S, et al. Trisubstituted-imidazoles induce apoptosis in human breast cancer cells by targeting the oncogenic PI3K/Akt/mTOR signaling pathway. PLoS ONE. 2016;11: e0153155.
Chen X, Li S, Li D, Li M, Su Z, Lai X, et al. Brucea javanica ethanol extract of Brucea javanica seed inhibit triple-negative breast cancer by restraining autophagy via PI3K/Akt/mTOR pathway. Front Pharmacol. 2020;11:606.
Yardena S, Wang Z, Alberto B, Natalie S, Jnine P, Steve S, et al. High frequency of mutations of the PIK3CA gene in human cancers. Science. 2004;304(5670):554.
Liu P, Cheng H, Roberts T, Zhao J. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov. 2009;8(8):627–44.
Filip J, Jennifer J, Aung N, Gerald S, David S, Vanda M, et al. PIK3CA mutation H1047R is associated with response to PI3K/AKT/mTOR signaling pathway inhibitors in early-phase clinical trials. Cancer Res. 2013;73(1):276–84.
Funding
This study was supported by the National Natural Science Foundation of China (Grant No. 81470874).
Author information
Authors and Affiliations
Contributions
QZ and ZC designed the research. ML, YX, YD, and YM performed the experiments, analyzed the data and drafted the manuscript. ZC and ML edited the manuscript. QZ, YX, and LX contributed to the discussion and review of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest in this study.
Ethical approval and consent to participate
The animal study was approved by the Laboratory Animal Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University, China (approval No. WYYY-AEC-2021-320). Animal experiments were performed according to all regulatory and institutional guidelines for animal welfare (National Institutes of Health Publications, NIH Publications No. 80-23).
Consent for publication
All authors agreed with submission of the manuscript for publication and agree to be accountable for all aspect of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, M., Xiao, Y., Dai, Y. et al. Chloroxine inhibits pancreatic cancer progression through targeted antagonization of the PI3K/AKT/mTOR signaling pathway. Clin Transl Oncol 26, 951–965 (2024). https://doi.org/10.1007/s12094-023-03328-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03328-w