Abstract
Purpose
To evaluate the efficacy and safety of capecitabine/cisplatin (XP) combined with intensity-modulated radiation therapy (IMRT) in patients with non-metastatic anal squamous cell carcinoma (ASCC).
Method and materials
All patients with ASCC who received radical concurrent chemoradiotherapy in the past 8 years were screened. Patients who received XP or mitomycin/5-fluorouracil (MF) were selected and analyzed retrospectively.
Results
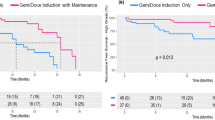
ASCC is an uncommon cancer, there were 36 patients were included in our study. The XP group and MF group included 18 patients each. The clinical complete response (cCR) rates in the XP group and the MF group were 94.4% and 88.9%, respectively (P = 1). The 2-year local control (LC), disease-free survival (DFS), and colostomy-free survival (CFS) rates were higher in the XP group than in the MF group (100% vs 93.3%, P = 0.32). Hematologic toxicities, especially grade ≥ 3 leukopenia (11.1% vs 44.4%, P = 0.06) and neutropenia (5.6% vs 61.1%, P = 0.001), were lower in the XP group than MF group. As a result of fewer side effects, fewer patients in the XP group demanded the dose reduction of chemotherapy (11.1% vs 50%, P = 0.03) and radiation interruption (55.6% vs 77.8%, P = 0.289). Delayed radiotherapy was shorter in the XP group (2.5 vs 6.5 days, P = 0.042) than in the MF group.
Conclusion
The XP regimen was as effective as the MF regimen in non-metastatic ASCC. Compared with the standard MF regimen, XP combined with IMRT showed higher treatment completion and lower toxicities. It could be considered a feasible alternative for patients with non-metastatic ASCC.


Similar content being viewed by others
Data availability
The data analysed during the current study are available from the corresponding author on reasonable request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. Cancer Stat Cancer J Clin. 2019;69(1):7–34. https://doi.org/10.3322/caac.21551.
Islami F, Ferlay J, Lortet-Tieulent J, Bray F, Jemal A. International trends in anal cancer incidence rates. Int J Epidemiol. 2017;46(3):924–38. https://doi.org/10.1093/ije/dyw276.
Siegel RL, Miller KD, Jemal A. Cancer statistics. Cancer J Clin. 2018. https://doi.org/10.3322/caac.21442.
Nigro ND, Vaitkevicius VK, Considine B Jr. Combined therapy for cancer of the anal canal: a preliminary report. Dis Colon Rectum. 1974;17(3):354–6. https://doi.org/10.1007/bf02586980.
Epidermoid anal cancer: results from the UKCCCR randomised trial of radiotherapy alone versus radiotherapy, 5-fluorouracil, and mitomycin. UKCCCR Anal Cancer Trial Working Party UK Co-ordinating Committee on Cancer Research. Lancet 348: 1049–1054
Bartelink H, Roelofsen F, Eschwege F, Rougier P, Bosset JF, Gonzalez DG, et al. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced anal cancer: results of a phase III randomized trial of the European organization for research and treatment of cancer radiotherapy and gastrointestinal cooperative groups. J Clin Oncol. 1997;15(5):2040–9. https://doi.org/10.1200/jco.1997.15.5.2040.
Ajani JA, Winter KA, Gunderson LL, Pedersen J, Benson AB 3rd, Thomas CR, et al. Fluorouracil, mitomycin, and radiotherapy vs fluorouracil, cisplatin, and radiotherapy for carcinoma of the anal canal: a randomized controlled trial. JAMA. 2008;299(16):1914–21. https://doi.org/10.1001/jama.299.16.1914.
James RD, Glynne-Jones R, Meadows HM, Cunningham D, Myint AS, Saunders MP, et al. Mitomycin or cisplatin chemoradiation with or without maintenance chemotherapy for treatment of squamous-cell carcinoma of the anus (ACT II): a randomised, phase 3, open-label, 2 × 2 factorial trial. Lancet Oncol. 2013;14(6):516–24. https://doi.org/10.1016/s1470-2045(13)70086-x.
Flam M, John M, Pajak TF, Petrelli N, Myerson R, Doggett S, et al. Role of mitomycin in combination with fluorouracil and radiotherapy, and of salvage chemoradiation in the definitive nonsurgical treatment of epidermoid carcinoma of the anal canal: results of a phase III randomized intergroup study. J Clin Oncol. 1996;14(9):2527–39. https://doi.org/10.1200/jco.1996.14.9.2527.
Gunderson LL, Winter KA, Ajani JA, Pedersen JE, Moughan J, Benson AB 3rd, et al. Long-term update of US GI intergroup RTOG 98–11 phase III trial for anal carcinoma: survival, relapse, and colostomy failure with concurrent chemoradiation involving fluorouracil/mitomycin versus fluorouracil/cisplatin. J Clin Oncol Off J Am Soc Clin Oncol. 2012;30(35):4344–51. https://doi.org/10.1200/jco.2012.43.8085.
Doci R, Zucali R, La Monica G, Meroni E, Kenda R, Eboli M, et al. Primary chemoradiation therapy with fluorouracil and cisplatin for cancer of the anus: results in 35 consecutive patients. J Clin Oncol Off J Am Soc Clin Oncol. 1996;14(12):3121–5. https://doi.org/10.1200/jco.1996.14.12.3121.
Peiffert D, Seitz JF, Rougier P, François E, Cvitkovic F, Mirabel X, et al. Preliminary results of a phase II study of high-dose radiation therapy and neoadjuvant plus concomitant 5-fluorouracil with CDDP chemotherapy for patients with anal canal cancer: a French cooperative study. Ann Oncology Off J Eur Soc Med Oncol. 1997;8(6):575–81. https://doi.org/10.1023/a:1008295119573.
Souza KT, Pereira AA, Araujo RL, Oliveira SC, Hoff PM, Riechelmann RP. Replacing 5-fluorouracil by capecitabine in localised squamous cell carcinoma of the anal canal: systematic review and meta-analysis. Ecancermedicalscience. 2016;10:699. https://doi.org/10.3332/ecancer.2016.699.
Oliveira SC, Moniz CM, Riechelmann R, Alex AK, Braghirolli MI, Bariani G, et al. Phase II Study of capecitabine in substitution of 5-FU in the chemoradiotherapy regimen for patients with localized squamous cell carcinoma of the anal canal. J Gastrointest Cancer. 2016;47(1):75–81. https://doi.org/10.1007/s12029-015-9790-4.
Meulendijks D, Dewit L, Tomasoa NB, van Tinteren H, Beijnen JH, Schellens JH, et al. Chemoradiotherapy with capecitabine for locally advanced anal carcinoma: an alternative treatment option. Br J Cancer. 2014;111(9):1726–33. https://doi.org/10.1038/bjc.2014.467.
Rotundo MS, Zampino MG, Ravenda PS, Bagnardi V, Peveri G, Dell’Acqua V, et al. Cisplatin plus capecitabine concomitant with intensity-modulated radiation therapy in non-metastatic anal squamous cell carcinoma: the experience of a single research cancer center. Therapeutic Adv in Med Oncol. 2020;12:1758835920940945. https://doi.org/10.1177/1758835920940945.
Li J, Xu H, Zou J, Wang X, Li Z, Shen Y. Cisplatin/capecitabine with intensity-modulated radiation therapy in anal squamous cell carcinoma: a preliminary study. Scand J Gastroenterol. 2021;56(4):432–6. https://doi.org/10.1080/00365521.2021.1879250.
Myerson RJ, Garofalo MC, El Naqa I, Abrams RA, Apte A, Bosch WR, et al. Elective clinical target volumes for conformal therapy in anorectal cancer: a radiation therapy oncology group consensus panel contouring atlas. Int J Radiat Oncol Biol Phys. 2009;74(3):824–30. https://doi.org/10.1016/j.ijrobp.2008.08.070.
Kachnic LA, Winter K, Myerson RJ, Goodyear MD, Willins J, Esthappan J, et al. RTOG 0529: a phase 2 evaluation of dose-painted intensity modulated radiation therapy in combination with 5-fluorouracil and mitomycin-C for the reduction of acute morbidity in carcinoma of the anal canal. Int J Radiat Oncol Biol Phys. 2013;86(1):27–33. https://doi.org/10.1016/j.ijrobp.2012.09.023.
Ng M, Leong T, Chander S, Chu J, Kneebone A, Carroll S, et al. Australasian Gastrointestinal Trials Group (AGITG) contouring atlas and planning guidelines for intensity-modulated radiotherapy in anal cancer. Int J Radiat Oncol Biol Phys. 2012;83(5):1455–62. https://doi.org/10.1016/j.ijrobp.2011.12.058.
Tchelebi LT, Eng C, Messick CA, Hong TS, Ludmir EB, Kachnic LA, et al. Current treatment and future directions in the management of anal cancer. CA Cancer J Clin. 2022;72(2):183–95. https://doi.org/10.3322/caac.21712.
Glynne-Jones R, Sebag-Montefiore D, Meadows HM, Cunningham D, Begum R, Adab F, et al. Best time to assess complete clinical response after chemoradiotherapy in squamous cell carcinoma of the anus (ACT II): a post-hoc analysis of randomised controlled phase 3 trial. Lancet Oncol. 2017;18(3):347–56. https://doi.org/10.1016/s1470-2045(17)30071-2.
Vieillot S, Fenoglietto P, Lemanski C, Moscardo CL, Gourgou S, Dubois JB, et al. IMRT for locally advanced anal cancer: clinical experience of the montpellier cancer center. Radiat Oncol. 2012;7:45. https://doi.org/10.1186/1748-717x-7-45.
Janssen S, Glanzmann C, Bauerfeind P, Stieb S, Studer G, Brown M, et al. Clinical experience of SIB-IMRT in anal cancer and selective literature review. Radiation Oncol. 2014;9:199. https://doi.org/10.1186/1748-717x-9-199.
Peixoto RD, Wan DD, Schellenberg D, Lim HJ. A comparison between 5-fluorouracil/mitomycin and capecitabine/mitomycin in combination with radiation for anal cancer. J Gastrointest Oncol. 2016;7(4):665–72. https://doi.org/10.21037/jgo.2016.06.04.
Ben-Josef E, Moughan J, Ajani JA, Flam M, Gunderson L, Pollock J, et al. Impact of overall treatment time on survival and local control in patients with anal cancer: a pooled data analysis of radiation therapy oncology group trials 87–04 and 98–11. J Clin Oncol. 2010;28(34):5061–6. https://doi.org/10.1200/jco.2010.29.1351.
Huang K, Haas-Kogan D, Weinberg V, Krieg R. Higher radiation dose with a shorter treatment duration improves outcome for locally advanced carcinoma of anal canal. World J Gastroenterol. 2007;13(6):895–900. https://doi.org/10.3748/wjg.v13.i6.895.
Roohipour R, Patil S, Goodman KA, Minsky BD, Wong WD, Guillem JG, et al. Squamous-cell carcinoma of the anal canal: predictors of treatment outcome. Dis Colon Rectum. 2008;51(2):147–53. https://doi.org/10.1007/s10350-007-9125-z.
Raphael MJ, Ko G, Booth CM, Brogly SB, Li W, Kalyvas M, et al. Factors associated with chemoradiation therapy interruption and noncompletion among patients with squamous cell anal carcinoma. JAMA Oncol. 2020;6(6):881–7. https://doi.org/10.1001/jamaoncol.2020.0809.
Almaazmi H, Taylor JP, Stem M, Yu D, Lo BD, Safar B, et al. Anal squamous cell carcinoma: radiation therapy alone must be avoided. J Surg Res. 2020;247:530–40. https://doi.org/10.1016/j.jss.2019.09.049.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
LJ and SY conceived the idea. WX, LZ and SY were responsible for the clinical care of patients. Data collection and analysis were performed by LJ and HC. The first draft of the manuscript was written by LJ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare they have no conflicts of interest. The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This study was approved by the Ethics Committee of West China hospital, Sichuan University China (No.20201000).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Huang, C., Wang, X. et al. Capecitabine/cisplatin combined with concurrent intensity-modulated radiation therapy: a feasible therapeutic strategy for anal squamous cell carcinoma. Clin Transl Oncol 26, 739–746 (2024). https://doi.org/10.1007/s12094-023-03296-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03296-1