Abstract
Introduction
Cancer of unknown primary (CUP) is a challenging malignancy. The purpose of this study was to investigate the clinical characteristics and prognosis of bone metastatic CUP using the population-based Surveillance, Epidemiology, and End Results (SEER) database.
Methods
From the SEER database, we identified 1908 patients with bone metastatic CUP at initial presentation between 2010 and 2018. Histology was subdivided following International Classification of Diseases for Oncology codes as Adenocarcinoma, Squamous cell, Neuroendocrine, or Carcinoma not otherwise specified (NOS). Cox proportional hazard modeling was applied using factors of age, sex, ethnicity, histological subtype, and therapeutic intervention.
Results
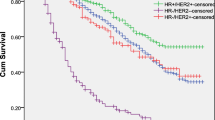
Among the 1908 patients, histology was Neuroendocrine in 240 patients, Squamous cell in 201 patients, Adenocarcinoma in 810 patients and NOS in 657 patients. In each subtype, patients tended to be predominantly male and white. Chemotherapy was introduced for 28% of patients and radiation for 34% in the entire cohort. Survival in patients with bone metastatic CUP was unfavorable, with a median survival of 2 months. Among the histological subtypes, Adenocarcinoma showed shorter survival than the other groups. In addition, treatment interventions such as chemotherapy and radiation therapy prolonged survival, particularly for Squamous cell, Adenocarcinoma and NOS, but not for Neuroendocrine.
Discussion
Bone metastatic CUP showed extremely poor prognosis, but treatment interventions such as chemotherapy and radiation generally offered survival benefits. Further randomized clinical research is needed to confirm the present results.


Similar content being viewed by others
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Ettinger DS, Handorf CR, Agulnik M, Bowles DW, Cates JM, Cristea M, et al. Occult primary, version 3.2014. J Natl Compr Canc Netw. 2014;12(7):969–74. https://doi.org/10.6004/jnccn.2014.0093.
Muir C. Cancer of unknown primary site. Cancer. 1995;75(1 Suppl):353–6. https://doi.org/10.1002/1097-0142(19950101)75:1+%3c353::aid-cncr2820751317%3e3.0.co;2-p.
Abbruzzese JL, Abbruzzese MC, Hess KR, Raber MN, Lenzi R, Frost P. Unknown primary carcinoma: natural history and prognostic factors in 657 consecutive patients. J Clin Oncol. 1994;12(6):1272–80. https://doi.org/10.1200/JCO.1994.12.6.1272.
Hainsworth JD, Greco FA. Treatment of patients with cancer of an unknown primary site. N Engl J Med. 1993;329(4):257–63. https://doi.org/10.1056/NEJM199307223290407.
Nystrom JS, Weiner JM, Heffelfinger-Juttner J, Irwin LE, Bateman JR, Wolf RM. Metastatic and histologic presentations in unknown primary cancer. Semin Oncol. 1977;4(1):53–8.
Mayordomo JI, Guerra JM, Guijarro C, Garcia-Prats MD, Gomez A, Lopez-Brea M, et al. Neoplasms of unknown primary site: a clinicopathological study of autopsied patients. Tumori. 1993;79(5):321–4. https://doi.org/10.1177/030089169307900507.
Abbruzzese JL, Abbruzzese MC, Lenzi R, Hess KR, Raber MN. Analysis of a diagnostic strategy for patients with suspected tumors of unknown origin. J Clin Oncol. 1995;13(8):2094–103. https://doi.org/10.1200/JCO.1995.13.8.2094.
Seve P, Billotey C, Broussolle C, Dumontet C, Mackey JR. The role of 2-deoxy-2-[F-18]fluoro-D-glucose positron emission tomography in disseminated carcinoma of unknown primary site. Cancer. 2007;109(2):292–9. https://doi.org/10.1002/cncr.22410.
Pavlidis N, Briasoulis E, Hainsworth J, Greco FA. Diagnostic and therapeutic management of cancer of an unknown primary. Eur J Cancer. 2003;39(14):1990–2005. https://doi.org/10.1016/s0959-8049(03)00547-1.
Fizazi K, Greco FA, Pavlidis N, Daugaard G, Oien K, Pentheroudakis G, et al. Cancers of unknown primary site: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(Suppl 5):v133–8. https://doi.org/10.1093/annonc/mdv305.
Varadhachary GR, Raber MN. Cancer of unknown primary site. N Engl J Med. 2014;371(8):757–65. https://doi.org/10.1056/NEJMra1303917.
Briasoulis E, Tolis C, Bergh J, Pavlidis N, Force EGT. ESMO Minimum Clinical Recommendations for diagnosis, treatment and follow-up of cancers of unknown primary site (CUP). Ann Oncol. 2005;16(Suppl 1):i75–6. https://doi.org/10.1093/annonc/mdi804.
Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379(9824):1428–35. https://doi.org/10.1016/S0140-6736(11)61178-1.
Dennis JL, Hvidsten TR, Wit EC, Komorowski J, Bell AK, Downie I, et al. Markers of adenocarcinoma characteristic of the site of origin: development of a diagnostic algorithm. Clin Cancer Res. 2005;11(10):3766–72. https://doi.org/10.1158/1078-0432.CCR-04-2236.
Yoon HH, Foster NR, Meyers JP, Steen PD, Visscher DW, Pillai R, et al. Gene expression profiling identifies responsive patients with cancer of unknown primary treated with carboplatin, paclitaxel, and everolimus: NCCTG N0871 (alliance). Ann Oncol. 2016;27(2):339–44. https://doi.org/10.1093/annonc/mdv543.
Urban D, Rao A, Bressel M, Lawrence YR, Mileshkin L. Cancer of unknown primary: a population-based analysis of temporal change and socioeconomic disparities. Br J Cancer. 2013;109(5):1318–24. https://doi.org/10.1038/bjc.2013.386.
Scoazec JY, Couvelard A, Monges G, Guyetant S, Bisot-Locard S, Parot X, et al. Professional practices and diagnostic issues in neuroendocrine tumour pathology: results of a prospective one-year survey among french pathologists (the PRONET Study). Neuroendocrinology. 2017;105(1):67–76. https://doi.org/10.1159/000448431.
Wang SC, Parekh JR, Zuraek MB, Venook AP, Bergsland EK, Warren RS, et al. Identification of unknown primary tumors in patients with neuroendocrine liver metastases. Arch Surg. 2010;145(3):276–80. https://doi.org/10.1001/archsurg.2010.10.
Rinke A, Muller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27(28):4656–63. https://doi.org/10.1200/JCO.2009.22.8510.
Caplin ME, Pavel M, Cwikla JB, Phan AT, Raderer M, Sedlackova E, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371(3):224–33. https://doi.org/10.1056/NEJMoa1316158.
Moertel CG, Kvols LK, O'Connell MJ, Rubin J. (1991) Treatment of neuroendocrine carcinomas with combined etoposide and cisplatin. Evidence of major therapeutic activity in the anaplastic variants of these neoplasms. Cancer 68(2):227–32. https://doi.org/10.1002/1097-0142(19910715)68:2<227::aid-cncr2820680202>3.0.co;2-i.
Mitry E, Baudin E, Ducreux M, Sabourin JC, Rufie P, Aparicio T, et al. Treatment of poorly differentiated neuroendocrine tumours with etoposide and cisplatin. Br J Cancer. 1999;81(8):1351–5. https://doi.org/10.1038/sj.bjc.6690325.
Chen KW, Liu CJ, Lu HJ, Tzeng CH, Liu JH, Chiou TJ, et al. Evaluation of prognostic factors and the role of chemotherapy in unfavorable carcinoma of unknown primary site: a 10-year cohort study. BMC Res Notes. 2012;5:70. https://doi.org/10.1186/1756-0500-5-70.
Colletier PJ, Garden AS, Morrison WH, Goepfert H, Geara F, Ang KK. Postoperative radiation for squamous cell carcinoma metastatic to cervical lymph nodes from an unknown primary site: outcomes and patterns of failure. Head Neck. 1998;20(8):674–81. https://doi.org/10.1002/(sici)1097-0347(199812)20:8%3c674::aid-hed3%3e3.0.co;2-h.
Aslani M, Sultanem K, Voung T, Hier M, Niazi T, Shenouda G. Metastatic carcinoma to the cervical nodes from an unknown head and neck primary site: Is there a need for neck dissection? Head Neck. 2007;29(6):585–90. https://doi.org/10.1002/hed.20581.
Guarischi A, Keane TJ, Elhakim T. Metastatic inguinal nodes from an unknown primary neoplasm. A review of 56 cases. Cancer. 1987;59(3):572–7.
Joseph K, Sawyer MB, Amanie J, Jones Thachuthara J, Ghosh S, Tai P. Carcinoma of unknown primary in the inguinal lymph node region of squamous cell origin: a case series. Pract Radiat Oncol. 2014;4(6):404–8. https://doi.org/10.1016/j.prro.2013.12.007.
Rosen LS, Gordon D, Tchekmedyian S, Yanagihara R, Hirsh V, Krzakowski M, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: a phase III, double-blind, randomized trial–the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21(16):3150–7. https://doi.org/10.1200/JCO.2003.04.105.
Lipton A, Fizazi K, Stopeck AT, Henry DH, Brown JE, Yardley DA, et al. Superiority of denosumab to zoledronic acid for prevention of skeletal-related events: a combined analysis of 3 pivotal, randomised, phase 3 trials. Eur J Cancer. 2012;48(16):3082–92. https://doi.org/10.1016/j.ejca.2012.08.002.
Argentiero A, Solimando AG, Brunetti O, Calabrese A, Pantano F, Iuliani M, et al. Skeletal metastases of unknown primary: biological landscape and clinical overview. Cancers (Basel). 2019. https://doi.org/10.3390/cancers11091270.
Funding
We have received no specific funding from any funding bodies to carry out this work.
Author information
Authors and Affiliations
Contributions
MM was involved in the design of the study, performed the clinical assessment, analysis, and interpretation of data, and drafted and revised the manuscript. TO and IY assisted with data interpretation and revised the manuscript for important intellectual content. KI, YS, HM, EK, and NI were involved in data acquisition and revised the manuscript critically for important intellectual content. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
IY reports grants from KAKENHI, AMED, and Health, Labour and Welfare Policy Research Grants, research funds from Nihon Medi-Physics, and speaking fees from Chugai Pharmaceutical Co. and AstraZeneca, outside the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matsuoka, M., Onodera, T., Yokota, I. et al. Bone metastatic cancer of unknown primary at initial presentation. Clin Transl Oncol 26, 461–467 (2024). https://doi.org/10.1007/s12094-023-03267-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03267-6