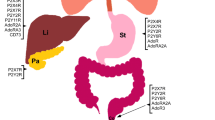
Abstract
Pancreatic cancer is one of the deadliest types of cancer, with a death rate nearly equal to the incidence. The P2X7 receptor (P2X7R) is a kind of extracellular adenosine triphosphate (ATP)-gated ion channel with special permeability, which exists in most tissues of human body and mediates inflammation-related signaling pathways and immune signal transduction after activation. P2X7R is also present on the surface of several tumor cells and is involved in tumor growth and progression. P2X7R expression in pancreatic cancer has also been identified in recent studies. Activation of P2X7R in pancreatic cancer can support the proliferation of pancreatic stellate cells, participate in protein interactions, and mediate ERK1/2, IL-6/STAT3, hCAP-18/LL-37, PI3K/AKT signaling pathways to promote pancreatic cancer progression. Inhibitors targeting P2X7R can inhibit the development of pancreatic cancer and are expected to be used in clinical therapy. Therefore, P2X7R is promising as a potential therapeutic target for pancreatic cancer. This article reviews the progress of research on P2X7R in pancreatic cancer.


Similar content being viewed by others
Data availability statement
Not applicable.
References
Ilic M, Ilic I. Epidemiology of pancreatic cancer. World J Gastroenterol. 2016;22:9694–705. https://doi.org/10.3748/wjg.v22.i44.9694.
Park W, Chawla A, O’Reilly EM. Pancreatic cancer: a review. JAMA. 2021;326:851–62. https://doi.org/10.1001/jama.2021.13027.
Mizrahi JD, Surana R, Valle JW, Shroff RT. Pancreatic cancer. Lancet. 2020;395:2008–20. https://doi.org/10.1016/S0140-6736(20)30974-0.
Vincent A, Herman J, Schulick R, Hruban RH, Goggins M. Pancreatic cancer. Lancet. 2011;378:607–20. https://doi.org/10.1016/S0140-6736(10)62307-0.
Burnstock G, Knight GE. Cellular distribution and functions of P2 receptor subtypes in different systems. Int Rev Cytol. 2004;240:31–304. https://doi.org/10.1016/s0074-7696(04)40002-3.
Cai X, Yao Y, Teng F, Li Y, Wu L, Yan W, et al. The role of P2X7 receptor in infection and metabolism: Based on inflammation and immunity. Int Immunopharmacol. 2021;101:108297. https://doi.org/10.1016/j.intimp.2021.108297.
Lara R, Adinolfi E, Harwood CA, Philpott M, Barden JA, Di Virgilio F, et al. P2X7 in cancer: from molecular mechanisms to therapeutics. Front Pharmacol. 2020;11:793. https://doi.org/10.3389/fphar.2020.00793.
Aria H, Rezaei M, Nazem S, Daraei A, Nikfar G, Mansoori B, et al. Purinergic receptors are a key bottleneck in tumor metabolic reprogramming: the prime suspect in cancer therapeutic resistance. Front Immunol. 2022;13:947885. https://doi.org/10.3389/fimmu.2022.947885.
Notomi S, Hisatomi T, Kanemaru T, Takeda A, Ikeda Y, Enaida H, et al. Critical involvement of extracellular ATP acting on P2RX7 purinergic receptors in photoreceptor cell death. Am J Pathol. 2011;179:2798–809. https://doi.org/10.1016/j.ajpath.2011.08.035.
Roger S, Pelegrin P. P2X7 receptor antagonism in the treatment of cancers. Expert Opin Investig Drugs. 2011;20:875–80. https://doi.org/10.1517/13543784.2011.583918.
Novak I, Yu H, Magni L, Deshar G. Purinergic signaling in pancreas-from physiology to therapeutic strategies in pancreatic cancer. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21228781.
Di Virgilio F, Dal Ben D, Sarti AC, Giuliani AL, Falzoni S. The P2X7 Receptor in Infection and Inflammation. Immunity. 2017;47:15–31. https://doi.org/10.1016/j.immuni.2017.06.020.
Giannuzzo A, Pedersen SF, Novak I. The P2X7 receptor regulates cell survival, migration and invasion of pancreatic ductal adenocarcinoma cells. Mol Cancer. 2015;14:203. https://doi.org/10.1186/s12943-015-0472-4.
Pothula SP, Pirola RC, Wilson JS, Apte MV. Pancreatic stellate cells: Aiding and abetting pancreatic cancer progression. Pancreatology. 2020;20:409–18. https://doi.org/10.1016/j.pan.2020.01.003.
Magni L, Bouazzi R, Heredero Olmedilla H, Petersen PSS, Tozzi M, Novak I. The P2X7 receptor stimulates IL-6 release from pancreatic stellate cells and tocilizumab prevents activation of STAT3 in pancreatic cancer cells. Cells. 2021. https://doi.org/10.3390/cells10081928.
Choi JH, Ji YG, Ko JJ, Cho HJ, Lee DH. Activating P2X7 Receptors Increases Proliferation of Human Pancreatic Cancer Cells via ERK1/2 and JNK. Pancreas. 2018;47:643–51. https://doi.org/10.1097/MPA.0000000000001055.
Sainz B Jr, Alcala S, Garcia E, Sanchez-Ripoll Y, Azevedo MM, Cioffi M, et al. Microenvironmental hCAP-18/LL-37 promotes pancreatic ductal adenocarcinoma by activating its cancer stem cell compartment. Gut. 2015;64:1921–35. https://doi.org/10.1136/gutjnl-2014-308935.
Campos-Contreras ADR, Diaz-Munoz M, Vazquez-Cuevas FG. Purinergic signaling in the hallmarks of cancer. Cells. 2020. https://doi.org/10.3390/cells9071612.
von Muecke-Heim IA, Ries C, Urbina L, Deussing JM. P2X7R antagonists in chronic stress-based depression models: a review. Eur Arch Psychiatry Clin Neurosci. 2021;271:1343–58. https://doi.org/10.1007/s00406-021-01306-3.
Stagg J, Smyth MJ. Extracellular adenosine triphosphate and adenosine in cancer. Oncogene. 2010;29:5346–58. https://doi.org/10.1038/onc.2010.292.
Jacobson KA, Paoletta S, Katritch V, Wu B, Gao ZG, Zhao Q, et al. Nucleotides acting at P2Y receptors: connecting structure and function. Mol Pharmacol. 2015;88:220–30. https://doi.org/10.1124/mol.114.095711.
Bartlett R, Stokes L, Sluyter R. The P2X7 receptor channel: recent developments and the use of P2X7 antagonists in models of disease. Pharmacol Rev. 2014;66:638–75. https://doi.org/10.1124/pr.113.008003.
Di Virgilio F, Schmalzing G, Markwardt F. The elusive P2X7 macropore. Trends Cell Biol. 2018;28:392–404. https://doi.org/10.1016/j.tcb.2018.01.005.
Di Virgilio F, Jiang LH, Roger S, Falzoni S, Sarti AC, Vultaggio-Poma V, et al. Structure, function and techniques of investigation of the P2X7 receptor (P2X7R) in mammalian cells. Methods Enzymol. 2019;629:115–50. https://doi.org/10.1016/bs.mie.2019.07.043.
De Salis SKF, Li L, Chen Z, Lam KW, Skarratt KK, Balle T, et al. Alternatively spliced isoforms of the P2X7 receptor: structure, function and disease associations. Int J Mol Sci. 2022. https://doi.org/10.3390/ijms23158174.
Denlinger LC, Fisette PL, Sommer JA, Watters JJ, Prabhu U, Dubyak GR, et al. Cutting edge: the nucleotide receptor P2X7 contains multiple protein- and lipid-interaction motifs including a potential binding site for bacterial lipopolysaccharide. J Immunol (Baltimore Md: 1950). 2001;167(2001):1871–6. https://doi.org/10.4049/jimmunol.167.4.1871.
Cunha SR, Mohler PJ. Ankyrin protein networks in membrane formation and stabilization. J Cell Mol Med. 2009;13:4364–76. https://doi.org/10.1111/j.1582-4934.2009.00943.x.
Smart ML, Gu B, Panchal RG, Wiley J, Cromer B, Williams DA, et al. P2X7 receptor cell surface expression and cytolytic pore formation are regulated by a distal C-terminal region. J Biol Chem. 2003;278:8853–60. https://doi.org/10.1074/jbc.M211094200.
Surprenant A, Rassendren F, Kawashima E, North RA, Buell G. The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science (New York, NY.). 1996;272:735–8. https://doi.org/10.1126/science.272.5262.735.
Hsu H, Huang J, Shu HB, Baichwal V, Goeddel DV. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity. 1996;4:387–96. https://doi.org/10.1016/s1074-7613(00)80252-6.
Wittmann I, Schönefeld M, Aichele D, Groer G, Gessner A, Schnare M. Murine bactericidal/permeability-increasing protein inhibits the endotoxic activity of lipopolysaccharide and gram-negative bacteria. J Immunol (Baltimore Md: 1950). 2008;180(2008):7546–52. https://doi.org/10.4049/jimmunol.180.11.7546.
Roger S, Jelassi B, Couillin I, Pelegrin P, Besson P, Jiang LH. Understanding the roles of the P2X7 receptor in solid tumour progression and therapeutic perspectives. Biochim Biophys Acta. 2015;1848:2584–602. https://doi.org/10.1016/j.bbamem.2014.10.029.
Illes P, Muller CE, Jacobson KA, Grutter T, Nicke A, Fountain SJ, et al. Update of P2X receptor properties and their pharmacology: IUPHAR Review 30. Br J Pharmacol. 2021;178:489–514. https://doi.org/10.1111/bph.15299.
Gu BJ, Saunders BM, Jursik C, Wiley JS. The P2X7-nonmuscle myosin membrane complex regulates phagocytosis of nonopsonized particles and bacteria by a pathway attenuated by extracellular ATP. Blood. 2010;115:1621–31. https://doi.org/10.1182/blood-2009-11-251744.
Pelegrin P, Surprenant A. Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. EMBO J. 2006;25:5071–82. https://doi.org/10.1038/sj.emboj.7601378.
Ugur M, Ugur O. A mechanism-based approach to P2X7 receptor action. Mol Pharmacol. 2019;95:442–50. https://doi.org/10.1124/mol.118.115022.
Kopp R, Krautloher A, Ramirez-Fernandez A, Nicke A. P2X7 interactions and signaling—making head or tail of it. Front Mol Neurosci. 2019;12:183. https://doi.org/10.3389/fnmol.2019.00183.
Adinolfi E, Giuliani AL, De Marchi E, Pegoraro A, Orioli E, Di Virgilio F. The P2X7 receptor: a main player in inflammation. Biochem Pharmacol. 2018;151:234–44. https://doi.org/10.1016/j.bcp.2017.12.021.
Adinolfi E, Raffaghello L, Giuliani AL, Cavazzini L, Capece M, Chiozzi P, et al. Expression of P2X7 receptor increases in vivo tumor growth. Cancer Res. 2012;72:2957–69. https://doi.org/10.1158/0008-5472.CAN-11-1947.
Grassi F, De Ponte Conti B. The P2X7 receptor in tumor immunity. Front Cell Dev Biol. 2021;9:694831. https://doi.org/10.3389/fcell.2021.694831.
Di Virgilio F, Sarti AC, Falzoni S, De Marchi E, Adinolfi E. Extracellular ATP and P2 purinergic signalling in the tumour microenvironment. Nat Rev Cancer. 2018;18:601–18. https://doi.org/10.1038/s41568-018-0037-0.
Park M, Kim J, Phuong NTT, Park JG, Park JH, Kim YC, et al. Involvement of the P2X7 receptor in the migration and metastasis of tamoxifen-resistant breast cancer: effects on small extracellular vesicles production. Sci Rep. 2019;9:11587. https://doi.org/10.1038/s41598-019-47734-z.
Solini A, Cuccato S, Ferrari D, Santini E, Gulinelli S, Callegari MG, et al. Increased P2X7 receptor expression and function in thyroid papillary cancer: a new potential marker of the disease? Endocrinology. 2008;149:389–96. https://doi.org/10.1210/en.2007-1223.
Vazquez-Cuevas FG, Martinez-Ramirez AS, Robles-Martinez L, Garay E, Garcia-Carranca A, Perez-Montiel D, et al. Paracrine stimulation of P2X7 receptor by ATP activates a proliferative pathway in ovarian carcinoma cells. J Cell Biochem. 2014;115:1955–66. https://doi.org/10.1002/jcb.24867.
Zhang Y, Ding J, Wang L. The role of P2X7 receptor in prognosis and metastasis of colorectal cancer. Adv Med Sci. 2019;64:388–94. https://doi.org/10.1016/j.advms.2019.05.002.
Boldrini L, Giordano M, Alì G, Servadio A, Pelliccioni S, Niccoli C, et al. P2X7 protein expression and polymorphism in non-small cell lung cancer (NSCLC). J Negat Results Biomed. 2014;13:16. https://doi.org/10.1186/1477-5751-13-16.
Pegoraro A, Adinolfi E. The ATP/P2X7 axis is a crucial regulator of leukemic initiating cells proliferation and homing and an emerging therapeutic target in acute myeloid leukemia. Purinergic Signal. 2021;17:319–21. https://doi.org/10.1007/s11302-021-09789-4.
Pegoraro A, De Marchi E, Ferracin M, Orioli E, Zanoni M, Bassi C, et al. P2X7 promotes metastatic spreading and triggers release of miRNA-containing exosomes and microvesicles from melanoma cells. Cell Death Dis. 2021;12:1088. https://doi.org/10.1038/s41419-021-04378-0.
Slater M, Danieletto S, Gidley-Baird A, Teh LC, Barden JA. Early prostate cancer detected using expression of non-functional cytolytic P2X7 receptors. Histopathology. 2004;44:206–15. https://doi.org/10.1111/j.0309-0167.2004.01798.x.
Sun SH. Roles of P2X7 receptor in glial and neuroblastoma cells: the therapeutic potential of P2X7 receptor antagonists. Mol Neurobiol. 2010;41:351–5. https://doi.org/10.1007/s12035-010-8120-x.
Liu H, Liu W, Liu Z, Liu Y, Zhang W, Xu L, et al. Prognostic value of purinergic P2X7 receptor expression in patients with hepatocellular carcinoma after curative resection. Tumour Biol. 2015;36:5039–49. https://doi.org/10.1007/s13277-015-3155-2.
Li X, Zhou L, Feng YH, Abdul-Karim FW, Gorodeski GI. The P2X7 receptor: a novel biomarker of uterine epithelial cancers. Cancer Epidemiol Biomarkers Prev. 2006;15:1906–13. https://doi.org/10.1158/1055-9965.EPI-06-0407.
Li X, Qi X, Zhou L, Catera D, Rote NS, Potashkin J, et al. Decreased expression of P2X7 in endometrial epithelial pre-cancerous and cancer cells. Gynecol Oncol. 2007;106:233–43. https://doi.org/10.1016/j.ygyno.2007.03.032.
Balkwill FR, Capasso M, Hagemann T. The tumor microenvironment at a glance. J Cell Sci. 2012;125:5591–6. https://doi.org/10.1242/jcs.116392.
Burnstock G, Knight GE. The potential of P2X7 receptors as a therapeutic target, including inflammation and tumour progression. Purinergic Signal. 2018;14:1–18. https://doi.org/10.1007/s11302-017-9593-0.
Adinolfi E, Callegari MG, Ferrari D, Bolognesi C, Minelli M, Wieckowski MR, et al. Basal activation of the P2X7 ATP receptor elevates mitochondrial calcium and potential, increases cellular ATP levels, and promotes serum-independent growth. Mol Biol Cell. 2005;16:3260–72. https://doi.org/10.1091/mbc.e04-11-1025.
de Andrade Mello P, Bian S, Savio LEB, Zhang H, Zhang J, Junger W, et al. Hyperthermia and associated changes in membrane fluidity potentiate P2X7 activation to promote tumor cell death. Oncotarget. 2017;8:67254–68. https://doi.org/10.18632/oncotarget.18595.
Feng YH, Li X, Wang L, Zhou L, Gorodeski GI. A truncated P2X7 receptor variant (P2X7-j) endogenously expressed in cervical cancer cells antagonizes the full-length P2X7 receptor through hetero-oligomerization. J Biol Chem. 2006;281:17228–37. https://doi.org/10.1074/jbc.M602999200.
Pegoraro A, De Marchi E, Adinolfi E. P2X7 variants in oncogenesis. Cells. 2021. https://doi.org/10.3390/cells10010189.
Giuliani AL, Colognesi D, Ricco T, Roncato C, Capece M, Amoroso F, et al. Trophic activity of human P2X7 receptor isoforms A and B in osteosarcoma. PLoS ONE. 2014;9:e107224. https://doi.org/10.1371/journal.pone.0107224.
Gilbert SM, Gidley Baird A, Glazer S, Barden JA, Glazer A, Teh LC, et al. A phase I clinical trial demonstrates that nfP2X(7) -targeted antibodies provide a novel, safe and tolerable topical therapy for basal cell carcinoma. Br J Dermatol. 2017;177:117–24. https://doi.org/10.1111/bjd.15364.
Qiao C, Tang Y, Li Q, Zhu X, Peng X, Zhao R. ATP-gated P2X7 receptor as a potential target for prostate cancer. Hum Cell. 2022;35:1346–54. https://doi.org/10.1007/s13577-022-00729-x.
Qian F, Xiao J, Hu B, Sun N, Yin W, Zhu J. High expression of P2X7R is an independent postoperative indicator of poor prognosis in colorectal cancer. Hum Pathol. 2017;64:61–8. https://doi.org/10.1016/j.humpath.2017.03.019.
Zhang Y, Cheng H, Li W, Wu H, Yang Y. Highly-expressed P2X7 receptor promotes growth and metastasis of human HOS/MNNG osteosarcoma cells via PI3K/Akt/GSK3beta/beta-catenin and mTOR/HIF1alpha/VEGF signaling. Int J Cancer. 2019;145:1068–82. https://doi.org/10.1002/ijc.32207.
Tan C, Han LI, Zou L, Luo C, Liu A, Sheng X, et al. Expression of P2X7R in breast cancer tissue and the induction of apoptosis by the gene-specific shRNA in MCF-7 cells. Exp Ther Med. 2015;10:1472–8. https://doi.org/10.3892/etm.2015.2705.
Qin J, Zhang X, Tan B, Zhang S, Yin C, Xue Q, et al. Blocking P2X7-mediated macrophage polarization overcomes treatment resistance in lung cancer. Cancer Immunol Res. 2020;8:1426–39. https://doi.org/10.1158/2326-6066.CIR-20-0123.
Ghalali A, Wiklund F, Zheng H, Stenius U, Hogberg J. Atorvastatin prevents ATP-driven invasiveness via P2X7 and EHBP1 signaling in PTEN-expressing prostate cancer cells. Carcinogenesis. 2014;35:1547–55. https://doi.org/10.1093/carcin/bgu019.
Zhang Y, Li F, Wang L, Lou Y. A438079 affects colorectal cancer cell proliferation, migration, apoptosis, and pyroptosis by inhibiting the P2X7 receptor. Biochem Biophys Res Commun. 2021;558:147–53. https://doi.org/10.1016/j.bbrc.2021.04.076.
de Andrade Mello P, Coutinho-Silva R, Savio LEB. Multifaceted effects of extracellular adenosine triphosphate and adenosine in the tumor-host interaction and therapeutic perspectives. Front Immunol. 2017;8:1526. https://doi.org/10.3389/fimmu.2017.01526.
He X, Wan J, Yang X, Zhang X, Huang D, Li X, et al. Bone marrow niche ATP levels determine leukemia-initiating cell activity via P2X7 in leukemic models. J Clin Invest. 2021. https://doi.org/10.1172/JCI140242.
Künzli BM, Berberat PO, Giese T, Csizmadia E, Kaczmarek E, Baker C, et al. Upregulation of CD39/NTPDases and P2 receptors in human pancreatic disease. Am J Physiol Gastrointest Liver Physiol. 2007;292:G223–230. https://doi.org/10.1152/ajpgi.00259.2006.
Mohammed A, Janakiram NB, Madka V, Pathuri G, Li Q, Ritchie R, et al. Lack of chemopreventive effects of P2X7R inhibitors against pancreatic cancer. Oncotarget. 2017;8:97822–34. https://doi.org/10.18632/oncotarget.22085.
Zhang GX, Wang MX, Nie W, Liu DW, Zhang Y, Liu HB. P2X7R blockade prevents NLRP3 inflammasome activation and pancreatic fibrosis in a mouse model of chronic pancreatitis. Pancreas. 2017;46:1327–35. https://doi.org/10.1097/MPA.0000000000000928.
Apte M, Pirola RC, Wilson JS. Pancreatic stellate cell: physiologic role, role in fibrosis and cancer. Curr Opin Gastroenterol. 2015;31:416–23. https://doi.org/10.1097/MOG.0000000000000196.
Masamune A, Watanabe T, Kikuta K, Shimosegawa T. Roles of pancreatic stellate cells in pancreatic inflammation and fibrosis. Clin Gastroenterol Hepatol. 2009;7:S48-54. https://doi.org/10.1016/j.cgh.2009.07.038.
Künzli BM, Nuhn P, Enjyoji K, Banz Y, Smith RN, Csizmadia E, et al. Disordered pancreatic inflammatory responses and inhibition of fibrosis in CD39-null mice. Gastroenterology. 2008;134:292–305. https://doi.org/10.1053/j.gastro.2007.10.030.
Haanes KA, Schwab A, Novak I. The P2X7 receptor supports both life and death in fibrogenic pancreatic stellate cells. PLoS ONE. 2012;7:e51164. https://doi.org/10.1371/journal.pone.0051164.
Fallah G, Romer T, Detro-Dassen S, Braam U, Markwardt F, Schmalzing G. TMEM16A(a)/anoctamin-1 shares a homodimeric architecture with CLC chloride channels. Mol Cell Proteomics. 2011;10:M110 004697. https://doi.org/10.1074/mcp.M110.004697.
Stolz M, Klapperstuck M, Kendzierski T, Detro-Dassen S, Panning A, Schmalzing G, et al. Homodimeric anoctamin-1, but not homodimeric anoctamin-6, is activated by calcium increases mediated by the P2Y1 and P2X7 receptors. Pflugers Arch. 2015;467:2121–40. https://doi.org/10.1007/s00424-015-1687-3.
Mazzone A, Eisenman ST, Strege PR, Yao Z, Ordog T, Gibbons SJ, et al. Inhibition of cell proliferation by a selective inhibitor of the Ca(2+)-activated Cl(-) channel, Ano1. Biochem Biophys Res Commun. 2012;427:248–53. https://doi.org/10.1016/j.bbrc.2012.09.022.
Wang B, Zhu XX, Pan LY, Chen HF, Shen XY. PP4C facilitates lung cancer proliferation and inhibits apoptosis via activating MAPK/ERK pathway. Pathol Res Pract. 2020;216:152910. https://doi.org/10.1016/j.prp.2020.152910.
Zhang Q, Li T, Wang Z, Kuang X, Shao N, Lin Y. lncRNA NR2F1-AS1 promotes breast cancer angiogenesis through activating IGF-1/IGF-1R/ERK pathway. J Cell Mol Med. 2020;24:8236–47. https://doi.org/10.1111/jcmm.15499.
Jin C, Chen Z, Shi W, Lian Q. Tropomodulin 3 promotes liver cancer progression by activating the MAPK/ERK signaling pathway. Oncol Rep. 2019;41:3060–8. https://doi.org/10.3892/or.2019.7052.
Yan Z, Ohuchida K, Fei S, Zheng B, Guan W, Feng H, et al. Inhibition of ERK1/2 in cancer-associated pancreatic stellate cells suppresses cancer-stromal interaction and metastasis. J Exp Clin Cancer Res. 2019;38:221. https://doi.org/10.1186/s13046-019-1226-8.
Tan G, Huang C, Chen J, Zhi F. HMGB1 released from GSDME-mediated pyroptotic epithelial cells participates in the tumorigenesis of colitis-associated colorectal cancer through the ERK1/2 pathway. J Hematol Oncol. 2020;13:149. https://doi.org/10.1186/s13045-020-00985-0.
Fresno Vara JA, Casado E, de Castro J, Cejas P, Belda-Iniesta C, Gonzalez-Baron M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004;30:193–204. https://doi.org/10.1016/j.ctrv.2003.07.007.
Hsieh AC, Truitt ML, Ruggero D. Oncogenic AKTivation of translation as a therapeutic target. Br J Cancer. 2011;105:329–36. https://doi.org/10.1038/bjc.2011.241.
Mistafa O, Stenius U. Statins inhibit Akt/PKB signaling via P2X7 receptor in pancreatic cancer cells. Biochem Pharmacol. 2009;78:1115–26. https://doi.org/10.1016/j.bcp.2009.06.016.
Giannuzzo A, Saccomano M, Napp J, Ellegaard M, Alves F, Novak I. Targeting of the P2X7 receptor in pancreatic cancer and stellate cells. Int J Cancer. 2016;139:2540–52. https://doi.org/10.1002/ijc.30380.
Sperb N, Tsesmelis M, Wirth T. Crosstalk between tumor and stromal cells in pancreatic ductal adenocarcinoma. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21155486.
Mohammed A, Qian L, Janakiram NB, Lightfoot S, Steele VE, Rao CV. Atorvastatin delays progression of pancreatic lesions to carcinoma by regulating PI3/AKT signaling in p48Cre/+ LSL-KrasG12D/+ mice. Int J Cancer. 2012;131:1951–62. https://doi.org/10.1002/ijc.27456.
Dong X, Fu J, Yin X, Cao S, Li X, Lin L, et al. Emodin: a review of its pharmacology, toxicity and pharmacokinetics. Phytother Res. 2016;30:1207–18. https://doi.org/10.1002/ptr.5631.
Liu L, Zou J, Liu X, Jiang LH, Li J. Inhibition of ATP-induced macrophage death by emodin via antagonizing P2X7 receptor. Eur J Pharmacol. 2010;640:15–9. https://doi.org/10.1016/j.ejphar.2010.04.036.
Zhu T, Zhang L, Ling S, Duan J, Qian F, Li Y, et al. Scropolioside B inhibits IL-1beta and cytokines expression through NF-kappaB and inflammasome NLRP3 pathways. Mediators Inflamm. 2014. https://doi.org/10.1155/2014/819053.
Rodgers MA, Bowman JW, Fujita H, Orazio N, Shi M, Liang Q, et al. The linear ubiquitin assembly complex (LUBAC) is essential for NLRP3 inflammasome activation. J Exp Med. 2014;211:1333–47. https://doi.org/10.1084/jem.20132486.
Zhang Q, Hu F, Guo F, Zhou Q, Xiang H, Shang D. Emodin attenuates adenosine triphosphate-induced pancreatic ductal cell injury in vitro via the inhibition of the P2X7/NLRP3 signaling pathway. Oncol Rep. 2019;42:1589–97. https://doi.org/10.3892/or.2019.7270.
Zhang Q, Tao X, Xia S, Qu J, Song H, Liu J, et al. Emodin attenuated severe acute pancreatitis via the P2X ligand-gated ion channel 7/NOD-like receptor protein 3 signaling pathway. Oncol Rep. 2019;41:270–8. https://doi.org/10.3892/or.2018.6844.
Funding
This work was supported by Shandong Provincial Natural Science Foundation (No. ZR2021MH306) and the National Natural Science Foundation of China (no. 81770915).
Author information
Authors and Affiliations
Contributions
QY and XW: literature searching, manuscript writing and figure preparing. XL and XB: literature searching and manuscript revision. XP and RZ: conception and supervised the current review. All of the authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Q., Wang, X., Li, X. et al. Purinergic P2X7R as a potential target for pancreatic cancer. Clin Transl Oncol 25, 2297–2305 (2023). https://doi.org/10.1007/s12094-023-03123-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03123-7