Abstract
Objective
Larynx preservation is the current standard for locally advanced (LA) laryngeal/hypopharyngeal tumors, but not all patients respond as expected. TALK score model measures four variables (T-staging, albumin levels, liquor consumption and Karnofsky score) to determine which cases are best suited to preservation treatment scheme. We aimed to validate this prognostic model in a Southern European population.
Methods
We retrospectively evaluated 175 patients diagnosed from July 2008 to December 2015 with LA laryngeal/hypopharyngeal carcinoma and treated with a laryngeal preservation scheme comprising induction chemotherapy followed by concomitant chemotherapy and radiotherapy. We applied the TALK score model to predict larynx preservation rate.
Results
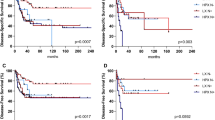
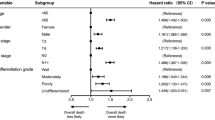
Of the 175 patients evaluated, 96.6% were men, 98.3% were smokers and 77.1% misused alcohol. Tumors were laryngeal 66.3% vs 33.7% in hypopharynx, and all were either stage III (37.7%) or stage IV (62.3%). TALK prognostic subgroups were: good risk 40.0%; intermediate risk 52.5%; and poor risk 7.5%. With a median follow-up of 40.1 months, larynx preservation rate, laryngectomy-free survival and overall survival at 3 years was 84.5%, 63.7% and 68.2%, respectively. Although TALK score was not predictive of 3-year larynx preservation rate (good risk 85.5%; intermediate risk 83.1%; poor risk 91.6%), it was predictive of 3-year overall survival (good risk 81.9%; intermediate risk 62.9%; poor risk 33.5%) and 3-year laryngectomy-free survival (good risk 75.6%; intermediate risk 59.6%; poor risk 30.7%).
Conclusion
TALK model could predict OS and laryngectomy-free survival, helping clinicians to decide which patients should avoid laryngeal preservation and undergo laryngectomy after diagnosis.




Similar content being viewed by others
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Globocan—Global Cancer Observatory 2020. Accessed on 14 Jul 2022. https://gco.iarc.fr
Yamazaki H, Suzuki G, Nakamura S, Hirano S, Yoshida K, Konishi K, et al. Radiotherapy for locally advanced resectable T3–T4 laryngeal cancer-does laryngeal preservation strategy compromise survival? J Radiat Res. 2018;59(1):77–90.
Forastiere AA, Ismaila N, Lewin JS, Nathan CA, Adelstein DJ, Eisbruch A, et al. Use of larynx-preservation strategies in the treatment of laryngeal cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2018;36(11):1143–69.
Garneau JC, Bakst RL, Miles BA. Hypopharyngeal cancer: a state of the art review. Oral Oncol. 2018;86:244–50.
Petersen JF, Timmermans AJ, Van Dijk CBA, Overbeek LIH, Smit LA, Hilgers FJM, et al. Trends in treatment, incidence and survival of hypopharynx cancer: a 20-year population-based study in the Netherlands. Eur Arch Otorhinolaryngol. 2018;275:181–9.
Megwalu UC, Sikora AG. Survival outcomes in advanced laryngeal cancer. JAMA Otolaryngol Head Neck Surg. 2014;140:855–60.
Soo KC, Tan EH, Wee J, Lim D, Tai B-C, Khoo M-L, et al. Surgery and adjuvant RT vs concurrent chemoradiotherapy in stage III/IV non metastatic squamous cell head and neck cancer: a randomized comparison. Br J Cancer. 2005;93:279–86.
Aldestein DJ, Lavertu P, Saxton JP, Secic M, Wood BG, Wanamaker JR, et al. Mature results of a phase III randomized trial comparing concurrent chemoradiotherapy with radiation therapy alone in patients with stage III and IV squamous cell carcinoma of the head and neck. Cancer. 2000;88:876–83.
Wolf GT, Fisher SG, Hong WK, Hillman R, Spaulding M, Laramore GE, et al. Induction chemotherapy plus radiation compared with surgery plus radiation in patients with advanced laryngeal cancer. N Engl J Med. 1991;324:1685–90.
Forastiere AA, Goepfert H, Maor M, Pajak TF, Weber R, Morrison W, et al. Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med. 2003;349:2091–8.
Fuller CD, Mohamed AS, Garden AS, Gunn GB, Mulcahy CF, Zafereo M, et al. Long-term outcomes after multidisciplinary management of T3 laryngeal squamous cell carcinomas: improved functional outcomes and survival with modern therapeutic approaches. Head Neck. 2016;38:1739–51.
Arribas L, Hurtós L, Sendrós MJ, Peiró I, Salleras N, Fort E, et al. NUTRISCORE: a new nutritional screening tool for oncological outpatients. Nutrition. 2017;33:297–303.
Lefebvre JL, Andry G, Chevalier D, Luboinski B, Collette L, Traissac L, et al. Laryngeal preservation with induction chemotherapy for hypopharyngeal squamous cell carcinoma: 10-year results of EORTC trial 24891. Ann Oncol. 2012;23:2708–14.
Lefebvre JL, Rolland F, Tesselaar M, Bardet E, Leemans CR, Geoffrois L, et al. Phase 3 randomized trial on larynx preservation comparing sequential vs alternating chemotherapy and radiotherapy. J Natl Cancer Inst. 2009;101:142–52.
Henriques De Figueiredo B, Fortpied C, Menis J, Lefebvre JL, Barzan L, de Raucourt D, et al. Long-term update of the 24954 EORTC phase III trial on larynx preservation. Eur J Cancer. 2016;65:109–12.
Pointreau Y, Garaud P, Chapet S, Sire C, Tuchais C, Tortochaux J, et al. Randomized trial of induction chemotherapy with cisplatin and 5-fluorouracil with or without docetaxel for larynx preservation. J Natl Cancer Inst. 2009;101:498–506.
Janoray G, Pointreau Y, Garaud P, Chapet S, Alfonsi M, Sire C et al. Long term results of a multicenter randomized phase III trial of induction chemotherapy with cisplatin, 5-fluorouracil, docetaxel for larynx preservation. J Natl Cancer Inst 2016;108(4):djv368.
Vermorken JB, Remenar E, van Herpen C, Gorlia T, Mesia R, Degardin M, et al. Cisplatin, fluorouracil, and docetaxel in unresectable head and neck cancer. N Engl J Med. 2007;357:1695–704.
Posner MR, Hershock DM, Blajman CR, Mickiewicz E, Winquist E, Gorbounova V, et al. Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl JMed. 2007;357:1705–15.
Hitt R, López-Pousa A, Martínez-Trufero J, Escrig V, Carles J, Rizo A, et al. Phase III study comparing cisplatin plus fluorouracil to paclitaxel, cisplatin, and fluorouracil induction chemotherapy followed by chemoradiotherapy in locally advanced head and neck cancer. J Clin Oncol. 2005;23:8636–45.
Bonner J, Giralt J, Harari P, Spencer S, Schulten J. Hossain A, et al. Cetuximab and radiotherapy in laryngeal preservation for cancers of the larynx and hypopharynx: a secondary analysis of a randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2016;142:842–9.
Lefebvre JL, Pointreau Y, Rolland F, Alfonsi M, Baudoux A, Sire C, et al. Induction chemotherapy followed by either chemoradiotherapy or bioradiotherapy for larynx preservation: the TREMPLIN randomized phase II study. J Clin Oncol. 2013;31:853–9.
Posner MR, Norris CM, Wirth LJ, Shin DM, Cullen KJ, Winquist EW, et al. Sequential therapy for the locally advanced larynx and hypopharynx cancer subgroup in TAX 324: survival, surgery, and organ preservation. Ann Oncol. 2009;20:921–7.
Forastiere AA, Zhang Q, Weber RS, Maor MH, Goepfert H, Pajak TF, et al. Long-term results of RTOG 91–11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. JCO. 2013;31(7):845–52.
Sherman EJ, Fisher SG, Kraus DH, Zelefsky MJ, Seshan VE, Singh B, et al. TALK score: development and validation of a prognostic model for predicting larynx preservation outcome. Laryngoscope. 2012;122:1043–50.
ICO-ICS praxis para el tratamiento médico y con irradiación del cáncer de orofaringe, hipofaringe y nasofaringe v2.2; noviembre 2017.
TNM classification of malignant tumors, 7th edition. August 2011.
Miller AB, Hoogstraten B, Statquet M, Winkler A, et al. Reporting results of cancer treatment. Cancer. 1981;47:207–14.
Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, Cohen RB, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006;354:567–78.
Pignon JP, le Maître A, Maillard E, Bourhis J. MACH-NC Collaborative group: Meta-Analysis of Chemotherapy in Head and Neck Cancer (MACH-NC): an update on 93 randomized trials and 17,346 patients. Radiother Oncol. 2009;92:4–14.
Weinstein GS, El-Sawy MM, Ruiz C, Dooley P, Chalian A, El-Sayed MM, et al. Laryngeal preservation with supracricoid partial laryngectomy results in improved quality of life when compared with total laryngectomy. Laryngoscope. 2001;111:191–9.
Weber RS, Berkey BA, Forastiere A, Cooper J, Maor M, Goepfert H, et al. Outcome of salvage total laryngectomy following organ preservation therapy: the Radiation Therapy Oncology Group trial 91–11. Arch Otolaryngol Head Neck Surg. 2003;129:44–9.
Eisbruch A, Schwartz M, Rasch C, Vineberg K, Damen E, Van As CJ, et al. Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected, and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004;60:1425–39.
Otsuki N, Matsui M, Takahashi M, Sasaki R, Kiyota N, Usami M, et al. Modified TALK score for Japanese patients with laryngeal and hypopharyngeal cancers to predict the possibility of laryngeal preservation by concurrent chemoradiotherapy. Kobe J Med Sci. 2017;63(num4):113–22.
Johnson M, Porterfield JZ and Kejner AE. Assessing the applicability of the TALK score: a modification for concurrent tobacco use during treatment. American Academy of Otolaryngology Head and Neck surgery foundation. 2022;166(2):282–288.
Funding
The authors received no specific funding for their participation in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Ethical approval and informed consent
Ethics committee was consulted and approved this retrospective review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marruecos-Querol, J., Rubió-Casadevall, J., Lozano, A. et al. Validation of a prognostic model for predicting larynx preservation outcome (TALK score) in a Southern European population. Clin Transl Oncol 25, 2384–2392 (2023). https://doi.org/10.1007/s12094-023-03121-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03121-9