Abstract
Background
Doublecortin-like kinase 2 (DCLK2) is a microtubule-associated protein kinase that participates in neural development and maturation; however, whether it is involved in tumour progression remains unclear.
Methods
DCLK2 overexpression and knockdown clones were established by lentivirus transfection. Western blot, PCR assays and bioinformatics analyses were conducted to observe the expression of DCLK2. CCK8, colony formation, scratch migration and Transwell assays were used to detect cell proliferation, migration and invasion, respectively. Tumour metastasis was evaluated in vivo using a tail vein metastasis model. Bioinformatics analyses were performed to analyse the expression correlation between DCLK2 and TCF4, or EMT markers in breast cancer.
Results
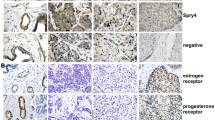
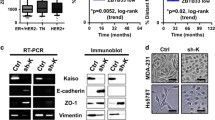
Our data indicate that DCLK2 is highly expressed in breast cancer cells and is associated with poor prognosis. Silencing DCLK2 does not affect the proliferation rate of tumour cells, but significantly suppresses migration and invasion as well as lung metastasis processes. Overexpression of DCLK2 can enhance the migratory and invasive abilities of normal breast epithelial cells. Moreover, TCF4/β-catenin inhibitor LF3 downregulates the expression of DCLK2 and inhibits the migration and invasion of breast cancer cells. Furthermore, we found that the downregulation of DCLK2 blocks the epithelial–mesenchymal transition (EMT) process.
Conclusion
Our study indicates that DCLK2 plays an important role in EMT, cell invasion and metastasis, suggesting that DCLK2 is a potential target for the treatment of metastatic breast cancer.





Similar content being viewed by others
Data availability statement
All data supporting the findings of this study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49.
Alarcón CR, Tavazoie SF. Cancer: endothelial-cell killing promotes metastasis. Nature. 2016;536(7615):154–5.
Seyfried TN, Huysentruyt LC. On the origin of cancer metastasis. Crit Rev Oncog. 2013;18(1–2):43–73.
Guise TA. Breast cancer bone metastases: it’s all about the neighborhood. Cell. 2013;154(5):957–9.
Yates LR, Knappskog S, Wedge D, Farmery JHR, Gonzalez S, Martincorena I, et al. Genomic evolution of breast cancer metastasis and relapse. Cancer Cell. 2017;32(2):169–84.
Chen Q, Boire A, Jin X, Valiente M, Emrah Er E, Lopez-Soto A, et al. Corrigendum: carcinoma-astrocyte gap junctions promote brain metastasis by cGAMP transfer. Nature. 2017;544(7648):124.
Reiner O, Coquelle FM, Peter B, Levy T, Kaplan A, Sapir T, et al. The evolving doublecortin (DCX) superfamily. BMC Genom. 2006;7:188.
Westphalen CB, Quante M, Wang TC. Functional implication of Dclk1 and Dclk1-expressing cells in cancer. Small GTPases. 2017;8(3):164–71.
Cheng L, Huang S, Chen L, Dong X, Zhang L, Wu C, et al. Research progress of DCLK1 inhibitors as cancer therapeutics. Curr Med Chem. 2022;29(13):2261–73.
Shin E, Kashiwagi Y, Kuriu T, Iwasaki H, Tanaka T, Koizumi H, et al. Doublecortin-like kinase enhances dendritic remodelling and negatively regulates synapse maturation. Nat Commun. 2013;4:1440.
Edelman AM, Kim WY, Higgins D, Goldstein EG, Oberdoerster M, Sigurdson W. Doublecortin kinase-2, a novel doublecortin-related protein kinase associated with terminal segments of axons and dendrites. J Biol Chem. 2005;280(9):8531–43.
Vreugdenhil E, Kolk SM, Boekhoorn K, Fitzsimons CP, Schaaf M, Schouten T, et al. Doublecortin-like, a microtubule-associated protein expressed in radial glia, is crucial for neuronal precursor division and radial process stability. Eur J Neurosci. 2007;25(3):635–48.
Koizumi H, Tanaka T, Gleeson JG. Doublecortin-like kinase functions with doublecortin to mediate fiber tract decussation and neuronal migration. Neuron. 2006;49(1):55–66.
Nawabi H, Belin S, Cartoni R, Williams PR, Wang C, Latremolière A, et al. Doublecortin-like kinases promote neuronal survival and induce growth cone reformation via distinct mechanisms. Neuron. 2015;88(4):704–19.
Deuel TA, Liu JS, Corbo JC, Yoo SY, Rorke-Adams LB, Walsh CA. Genetic interactions between doublecortin and doublecortin-like kinase in neuronal migration and axon outgrowth. Neuron. 2006;49(1):41–53.
Friocourt G, Liu JS, Antypa M, Rakic S, Walsh CA, Parnavelas JG. Both doublecortin and doublecortin-like kinase play a role in cortical interneuron migration. J Neurosci. 2007;27(14):3875–83.
Kerjan G, Koizumi H, Han EB, Dubé CM, Djakovic SN, Patrick GN, et al. Mice lacking doublecortin and doublecortin like kinase 2 display altered hippocampal neuronal maturation and spontaneous seizures. Proc Natl Acad Sci USA. 2009;106(16):6766–71.
Barrow TM, Wong Doo N, Milne RL, Giles GG, Willmore E, Strathdee G, et al. Analysis of retrotransposon subfamily DNA methylation reveals novel early epigenetic changes in chronic lymphocytic leukemia. Haematologica. 2021;106(1):98–110.
Chaffer CL, San Juan BP, Lim E, Weinberg RA. EMT, cell plasticity and metastasis. Cancer Metastasis Rev. 2016;35(4):645–54.
Brabletz T, Kalluri R, Nieto MA, Weinberg RA. EMT in cancer. Nat Rev Cancer. 2018;18(2):128–34.
Brueffer C, Vallon-Christersson J, Grabau D, Ehinger A, Häkkinen J, Hegardt C, et al. Clinical value of RNA sequencing-based classifiers for prediction of the five conventional breast cancer biomarkers: a report from the population-based multicenter Sweden cancerome analysis network-breast initiative. JCO Precis Oncol. 2018. https://doi.org/10.1200/PO.17.00135.
Clevers H, Nusse R. Wnt/β-catenin signaling and disease. Cell. 2012;149(6):1192–205.
Kohn AD, Moon RT. Wnt and calcium signaling: beta-catenin-independent pathways. Cell Calcium. 2005;38(3–4):439–46.
Fang L, Zhu Q, Neuenschwander M, Specker E, Wulf-Goldenberg A, Weis WI, et al. A small-molecule antagonist of the β-Catenin/TCF4 interaction blocks the self-renewal of cancer stem cells and suppresses tumorigenesis. Cancer Res. 2016;76(4):891–901.
Nieto MA, Huang RY, Jackson RA, Thiery JP. EMT: 2016. Cell. 2016;166(1):21–45.
Gao T, Wang M, Xu L, Wen T, Liu J, An G. DCLK1 is up-regulated and associated with metastasis and prognosis in colorectal cancer. J Cancer Res Clin Oncol. 2016;142(10):2131–40.
Mohammadi Y, Tavangar SM, Saidijam M, Amini R, Etemadi K, Karimi Dermani F, et al. DCLK1 plays an important role in colorectal cancer tumorgenesis through the regulation of miR-200c. Biomed Pharmacother. 2018;103:301–7.
Eubelen M, Bostaille N, Cabochette P, Gauquier A, Tebabi P, Dumitru AC, et al. A molecular mechanism for Wnt ligand-specific signaling. Science. 2018. https://doi.org/10.1126/science.aat1178.
Tammela T, Sanchez-Rivera FJ, Cetinbas NM, Wu K, Joshi NS, Helenius K, et al. A Wnt-producing niche drives proliferative potential and progression in lung adenocarcinoma. Nature. 2017;545(7654):355–9.
McCracken KW, Aihara E, Martin B, Crawford CM, Broda T, Treguier J, et al. Erratum: Wnt/β-catenin promotes gastric fundus specification in mice and humans. Nature. 2017;543(7643):136.
Yan KS, Janda CY, Chang J, Zheng GXY, Larkin KA, Luca VC, et al. Non-equivalence of Wnt and R-spondin ligands during Lgr5(+) intestinal stem-cell self-renewal. Nature. 2017;545(7653):238–42.
Boj SF, van Es JH, Huch M, Li VS, José A, Hatzis P, et al. Diabetes risk gene and Wnt effector Tcf7l2/TCF4 controls hepatic response to perinatal and adult metabolic demand. Cell. 2012;151(7):1595–607.
Sánchez-Tilló E, de Barrios O, Siles L, Cuatrecasas M, Castells A, Postigo A. β-catenin/TCF4 complex induces the epithelial-to-mesenchymal transition (EMT)-activator ZEB1 to regulate tumor invasiveness. Proc Natl Acad Sci USA. 2011;108(48):19204–9.
Loh CY, Chai JY, Tang TF, Wong WF, Sethi G, Shanmugam MK, et al. The E-Cadherin and N-Cadherin switch in epithelial-to-mesenchymal transition: signaling, therapeutic implications, and challenges. Cells. 2019;8(10):1118.
Cano A, Pérez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, et al. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol. 2000;2(2):76–83.
Funding
This research was supported by National Natural Science Foundation of China (81672629, 81872168 and 82072925) to JS.
Author information
Authors and Affiliations
Contributions
JS conceived and supervised the study, wrote the manuscript; YH, XD, SL and XZ performed experiments; KG and KS performed bioinformatics analyses; YH, XD and SL analysed data.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This work has been approved by the Ethics Committee of Southern Medical University and Guangdong Provincial People’s Hospital.
Informed consent
All patients gave informed consent for participation.
Consent for publication
This article has been read and approved in the present form for submission by all authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, Y., Dai, X., Li, S. et al. Doublecortin-like kinase 2 promotes breast cancer cell invasion and metastasis. Clin Transl Oncol 25, 1102–1113 (2023). https://doi.org/10.1007/s12094-022-03018-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-022-03018-z