Abstract
Background
Gastrointestinal stromal tumours (GISTs) located in the jejunum or ileum (JI-GIST) are considered worse prognosis compared to those of gastric (G-GIST) location. It has been suggested that this dogma should be revised. The aim of this study was to describe the characteristics of jejunoileal GISTs and its prognosis and to compare them with G-GISTs in the era of imatinib.
Methods
We retrospectively reviewed the clinical histories of all the patients diagnosed with GISTs between January 2000 and November 2016: Clinical and pathological data, as recurrence, metastatic state, disease-free survival (DFS) as well as overall survival (OS) rates of patients were reviewed.
Results
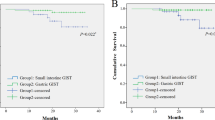
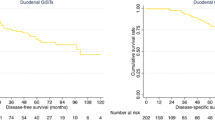
JI-GIST patients comprise 29 cases (37.7%). Compared to G-GIST, JI-GIST patients had undergone emergency surgery more frequently (37.9% vs. 10.4%, p = 0.007). According to the NIH-Fletcher classification, the low or very-low risk group represents 17.2% of JI-GISTs as opposed to 37.6% of G-GISTs (p < 0.005). When the AFIP-Miettinen system was used the low or very-low group represented 17.2% of JI-GISTs vs. 58.4% in the G-GISTs group (p < 0.001). Both local recurrence (24.1% vs. 12.5%, p < 0.05) and metastatic rate (34.5% vs. 22.9%, p < 0.05) were higher in the JI-GIST group than in G-GIST. 5- and 10-year DFS and 10-year OS rate were lower for JI-GIST (54.5% and 39.6% vs. 77.2% and 60.8%, and 57.9% vs. 65%, respectively, p < 0.05).
Conclusions
The observed differences between both groups in DFS and OS rates at long term could be attributed to the effect of imatinib.



Similar content being viewed by others
Abbreviations
- GIST:
-
Gastrointestinal stromal tumour
- JI-GIST:
-
Gastrointestinal stromal tumours located in the jejunum or ileum
- G-GIST:
-
Gastrointestinal stromal tumours with gastric location
- UGB:
-
Upper gastrointestinal bleeding
- KIT:
-
Gene located on chromosome 4q12
- PDGFRA:
-
Platelet derived growth factor receptor alpha
- HPF:
-
High-power field
- NIH:
-
National Institutes of Health
- AFIP:
-
Armed forces institute of pathology
- DFS:
-
Disease-free survival
- OS:
-
Overall survival
- MSKCCn:
-
Memorial Sloan Kettering Cancer Center nomogram
- NS:
-
Not statistically significant
References
Søreide K, Sandvik OM, Søreide JA, Giljaca V, Jureckova A, Bulusu VR. Global epidemiology of gastrointestinal stromal tumours (GIST): A systematic review of population-based cohort studies. Cancer Epidemiol. 2015;40:39–46.
Ma GL, Murphy JD, Martinez ME, Sicklick JK. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24(1):298–302.
Joensuu H, Vehtari A, Riihimäki J, Nishida T, Steigen SE, Brabec P, et al. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13(3):265–74.
Rutkowski P, Nowecki ZI, Michej W, Debiec-Rychter M, Woźniak A, Limon J, et al. Risk criteria and prognostic factors for predicting recurrences after resection of primary gastrointestinal stromal tumor. Ann Surg Oncol. 2007;14(7):2018–27.
Crosby JA, Catton CN, Davis A, Couture J, O’Sullivan B, Kandel R, et al. Malignant gastrointestinal stromal tumors of the small intestine: a review of 50 cases from a prospective database. Ann Surg Oncol. 2001;8(1):50–9.
Brainard JA, Goldblum JR. Stromal tumors of the jejunum and ileum: a clinicopathologic study of 39 cases. Am J Surg Pathol. 1997;21(4):407–16.
Wu T-J, Lee L-Y, Yeh C-N, Wu P-Y, Chao T-C, Hwang T-L, et al. Surgical treatment and prognostic analysis for gastrointestinal stromal tumors (GISTs) of the small intestine: before the era of imatinib mesylate. BMC Gastroenterol. 2006;6:29.
Liao C-H, Yeh C-N, Wang S-Y, Fu C-Y, Tsai C-Y, Liu Y-Y, et al. Surgical option for intestinal gastrointestinal stromal tumors perioperative and oncological outcomes of laparoscopic surgery. Anticancer Res. 2015;35(2):1033–40.
Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30(4):477–89.
Huang C-C, Yang C-Y, Lai I-R, Chen C-N, Lee P-H, Lin M-T. Gastrointestinal stromal tumor of the small intestine: a clinicopathologic study of 70 cases in the postimatinib era. World J Surg. 2009;33(4):828–34.
Han IW, Jang JY, Lee KB, Kang MJ, Kwon W, Park JW, et al. Clinicopathologic analysis of gastrointestinal stromal tumors in duodenum and small intestine. World J Surg. 2015;39(4):1026–33.
Sandvik OM, Søreide K, Gudlaugsson E, Søreide JA. Surgery for gastrointestinal stromal tumors (GISTs) of the stomach and small bowel: short and long-term outcomes over three decades. World J Surg. 2015;39(2):446–52.
Guller U, Tarantino I, Cerny T, Ulrich A, Schmied BM, Warschkow R. Revisiting a dogma: similar survival of patients with small bowel and gastric GIST. A population-based propensity score SEER analysis. Gastric Cancer. 2017;20(1):49–60.
Giuliano K, Nagarajan N, Canner J, Najafian A, Wolfgang C, Schneider E, et al. Gastric and small intestine gastrointestinal stromal tumors: do outcomes differ? J Surg Oncol. 2017;115(3):351–7.
Yang Z, Wang F, Liu S, Guan W. Comparative clinical features and short-term outcomes of gastric and small intestinal gastrointestinal stromal tumours: a retrospective study. Sci Rep. 2019;9(1):10033.
Boonstra PA, Steeghs N, Farag S, van Coevorden F, Gelderblom H, Grunhagen D, et al. Surgical and medical management of small bowel gastrointestinal stromal tumors: a report of the Dutch GIST registry. Eur J Surg Oncol. 2019;45(3):410–5.
Everett M, Gutman H. Surgical management of gastrointestinal stromal tumors: analysis of outcome with respect to surgical margins and technique. J Surg Oncol. 2008;98:588–93.
Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006;23(2):70–83.
Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33(5):459–65.
Patel S. Navigating risk stratification systems for the management of patients with GIST. Ann Surg Oncol. 2011;18(6):1698–704.
DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred Gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231(1):51–8.
Goh BK, Chow PK, Yap WM, Kesavan SM, Song IC, Paul PG, et al. Which is the optimal risk stratification system for surgically treated localized primary GIST? Comparison of three contemporary prognostic criteria in 171 tumors and a proposal for a modified Armed Forces Institute of Pathology risk criteria. Ann Surg Oncol. 2008;15(8):2153–63.
Gold JS, Gönen M, Gutierrez A, Broto JM, García-del Muro X, Smyrk TC, et al. Development and validation of a prognostic nomogram for recurrence-free survival after resection of localised primary gastrointestinal stromal tumor: a retrospective analysis. Lancet Oncol. 2009;10(11):1045–52.
Almaazmi H, Stem M, Lo BD, Taylor JP, Fang SH, Safar B, et al. The impact of imatinib on survival and treatment trends for small bowel and colorectal gastrointestinal stromal tumors. J Gastrointest Surg. 2020;24(1):98–108.
ESMO Guidelines Committee. Appendix 3: Soft tissue sarcoma: MCBS eUpdate published online 5 May 2017 (www.esmo.org/Guidelines/Sarcoma-and-GIST). Ann Oncol. 2017;28(suppl_4):iv147–48. https://doi.org/10.1093/annonc/mdx241.
Demetri GD, Benjamin RS, Blanke CD, Blay JY, Casali P, Choi H, et al. NCCN Task Force report: management of patients with gastrointestinal stromal tumor (GIST)–update of the NCCN clinical practice guidelines. J Natl Compr Canc Netw. 2007;5(Suppl 2):S1–30.
Poveda A, Martinez V, Serrano C, Sevilla I, Lecumberri MJ, de Beveridge RD, et al. SEOM Clinical Guideline for gastrointestinal sarcomas (GIST). Clin Transl Oncol. 2016;18(12):1221–8.
Casali PG, Le Cesne A, Poveda Velasco A, Kotasek D, Rutkowski P, Hohenberger P, et al. Time to definitive failure to the first tyrosine kinase inhibitor in localized gi stromal tumors treated with imatinib as an adjuvant: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Intergroup Randomized Trial in Collaboration With the Australasian Gastro-Intestinal Trials Group, UNICANCER, French Sarcoma Group, Italian Sarcoma Group, and Spanish Group for Research on Sarcomas. J Clin Oncol. 2015;33(36):4276–83.
Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schutte J, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307(12):1265–72.
Lin JX, Chen QF, Zheng CH, Li P, Xie JW, Wang JB, et al. Is 3-years duration of adjuvant imatinib mesylate treatment sufficient for patients with high-risk gastrointestinal stromal tumor? A study based on long-term follow-up. J Cancer Res Clin Oncol. 2017;143(4):727–34.
McDonnell MJ, Punnoose S, Viswanath YKS, Wadd NJ, Dhar A. Gastrointestinal stromal tumours (GISTs): an insight into clinical practice with review of literature. Front Gastroenterol. 2017;8(1):19–25.
Martínez-Marín V, Maki RG. Knowns and known unknowns of gastrointestinal stromal tumor adjuvant therapy. Gastroenterol Clin N Am. 2016;45(3):477–86.
Three Versus Five Years of Adjuvant Imatinib as Treatment of Patients With Operable GIST. ClinicalTrials.gov. Identifier: NCT02413736
Patel DJ, Lutfi W, Eguia E, Sweigert P, Knab L, Abood G, et al. Adjuvant systemic therapy for small bowel gastrointestinal stromal tumor (GIST): is there a survival benefit after R0 resection? Surgery. 2020.
Ye H, Xin H, Zheng Q, Shen Q, Dai W, Wu F, et al. Prognostic role of the primary tumour site in patients with operable small intestine and gastrointestinal stromal tumours: a large population-based analysis. Oncotarget. 2017;9(8):8147–54.
Giuliano K, Ejaz A, Reames BN, Choi W, Sham J, Gage M, et al. Comparing the long-term outcomes among patients with stomach and small intestine gastrointestinal stromal tumors: An analysis of the National Cancer Database. J Surg Oncol. 2018;118(3):486–92.
Author information
Authors and Affiliations
Contributions
All authors have contributed equally to the conception of the work, the acquisition, analysis, interpretation of data, drafting the work, and revising it critically for important intellectual content; final approval of the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this manuscript have no conflicts of interest to declare.
Ethical approval
All of the procedures carried out in studies with human participants were in accordance with the ethical standards of the institutional and/or national research committee and the Declaration of Helsinki of 1964 and its subsequent modifications or comparable ethical standards. The study was approved by the Ethics Committee of the “La Arrixaca” University Hospital in Murcia.
Informed consent
For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fernández, J.A., Ferreras, D., Ruiz-Manzanera, J.J. et al. Characteristics and prognosis of jejunoileal gastrointestinal stromal tumours (GISTs) in the era of imatinib: a comparative study with gastric GISTs. Clin Transl Oncol 23, 1368–1376 (2021). https://doi.org/10.1007/s12094-020-02528-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02528-y