Abstract
Purpose
Glioblastoma multiforme (GBM) represents the most common and the most malignant type of brain tumor. Cell division cycle 6 (CDC6), a gene associated with DNA replication initiation, has been proven to be associated with the prognosis of multiple tumors. In this study, we aim to explore the association between CDC6 expression and GBM carcinogenesis and prognosis.
Methods
CDC6 expression in normal cells and GBM cells was explored by analyzing TCGA dataset, as well as by RT-PCR and western blot methods. Survival analysis was performed by the Kaplan–Meier method. Multivariate Cox-regression analysis was adopted to estimate the independence of CDC6 as a GBM prognostic factor.
Results and conclusions
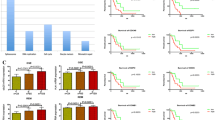
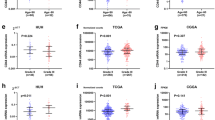
Elevated CDC6 levels in GBM tumor tissues compared with those in normal brain tissues were illustrated by analyzing the gene expression profiles from TCGA dataset, and confirmed by RT-PCR and western blot assays in GBM tumor and normal human astrocyte cell lines. Kaplan–Meier analysis indicated the negative influence of high CDC6 expression on GBM overall survival (OS) probability and days to progression (D2P) after initial treatment, but not on days to recurrence (D2R) after initial treatment. Multivariate Cox regression analysis showed CDC6 as an independent signature marker gene for GBM prognosis. In addition, the combination of CDC6 mRNA expression and CpG island methylator phenotype (CIMP) could sensitively predict 3-year OS and D2P. In conclusion, our study uncovered the role of CDC6 in GBM carcinogenesis and prognosis for the first time, which could shed new light on GBM diagnosis and treatment.




Similar content being viewed by others
References
Tyler B, DiMeco F, Grossman R, Pradilla G. Current and future novel treatments for glioblastoma multiforme. Biomed Res Int. 2014;2014:432195. https://doi.org/10.1155/2014/432195.
Kalkan R. Glioblastoma multiforme: the genetic perspective of the treatment planning. Crit Rev Eukaryot Gene Expr. 2015;25(4):281–5. https://doi.org/10.1615/critreveukaryotgeneexpr.2015014259.
Williams DS. Glioblastoma multiforme. J Insur Med. 2014;44(1):62–4.
Kotliarova S, Fine HA. SnapShot: glioblastoma multiforme. Cancer Cell. 2012;21(5):710-e1. https://doi.org/10.1016/j.ccr.2012.04.031.
Maiese K. “Micromanaging” glioblastoma multiforme: the potential of MicroRNAs, circular RNAs, and the hippo pathway as novel treatment strategies. Curr Neurovasc Res. 2018;15(3):173–4. https://doi.org/10.2174/1567202615999180711123907.
Khan IS, Ehtesham M. Targeting glioblastoma cancer stem cells: the next great hope? Neurosurg Focus. 2014;37(6):E7. https://doi.org/10.3171/2014.9.focus14509.
Kim GS, Lee I, Kim JH, Hwang DS. The replication protein Cdc6 suppresses centrosome over-duplication in a manner independent of its ATPase activity. Mol Cells. 2017;40(12):925–34. https://doi.org/10.14348/molcells.2017.0191.
Borlado LR, Mendez J. CDC6: from DNA replication to cell cycle checkpoints and oncogenesis. Carcinogenesis. 2008;29(2):237–43. https://doi.org/10.1093/carcin/bgm268.
Parker MW, Botchan MR, Berger JM. Mechanisms and regulation of DNA replication initiation in eukaryotes. Crit Rev Biochem Mol Biol. 2017;52(2):107–44. https://doi.org/10.1080/10409238.2016.1274717.
Teixeira LK, Reed SI. Cdc6: Skin in the carcinogenesis game. Cell Cycle. 2016;15(3):313. https://doi.org/10.1080/15384101.2015.1131528.
Kim YH, Byun YJ, Kim WT, Jeong P, Yan C, Kang HW, et al. CDC6 mRNA expression is associated with the aggressiveness of prostate cancer. J Korean Med Sci. 2018;33(47):e303. https://doi.org/10.3346/jkms.2018.33.e303.
Deng Y, Jiang L, Wang Y, Xi Q, Zhong J, Liu J, et al. High expression of CDC6 is associated with accelerated cell proliferation and poor prognosis of epithelial ovarian cancer. Pathol Res Pract. 2016;212(4):239–46. https://doi.org/10.1016/j.prp.2015.09.014.
Rossi E, Klersy C, Manca R, Zuffardi O, Solcia E. Correlation between genomic alterations assessed by array comparative genomic hybridization, prognostically informative histologic subtype, stage, and patient survival in gastric cancer. Hum Pathol. 2011;42(12):1937–45. https://doi.org/10.1016/j.humpath.2011.02.016.
Jiang W, Yu Y, Liu J, Zhao Q, Wang J, Zhang J, et al. Downregulation of Cdc6 inhibits tumorigenesis of osteosarcoma in vivo and in vitro. Biomed Pharmacother = Biomedecine Pharmacotherapie. 2019;115:108949. https://doi.org/10.1016/j.biopha.2019.108949.
Kong X, Duan Y, Sang Y, Li Y, Zhang H, Liang Y, et al. LncRNA-CDC6 promotes breast cancer progression and function as ceRNA to target CDC6 by sponging microRNA-215. J Cell Physiol. 2019;234(6):9105–17. https://doi.org/10.1002/jcp.27587.
Mahadevappa R, Neves H, Yuen SM, Bai Y, McCrudden CM, Yuen HF, et al. The prognostic significance of Cdc6 and Cdt1 in breast cancer. Sci Rep. 2017;7(1):985. https://doi.org/10.1038/s41598-017-00998-9.
Hu Y, Wang L, Li Z, Wan Z, Shao M, Wu S, et al. Potential prognostic and diagnostic values of CDC6, CDC45, ORC6 and SNHG7 in colorectal cancer. OncoTargets Ther. 2019;12:11609–21. https://doi.org/10.2147/OTT.S231941.
Yan X, Wan H, Hao X, Lan T, Li W, Xu L, et al. Importance of gene expression signatures in pancreatic cancer prognosis and the establishment of a prediction model. Cancer Manag Res. 2019;11:273–83. https://doi.org/10.2147/CMAR.S185205.
Sugimoto N, Fujita M. Molecular mechanism for chromatin regulation during MCM loading in mammalian cells. Adv Exp Med Biol. 2017;1042:61–78. https://doi.org/10.1007/978-981-10-6955-0_3.
Ohta S, Koide M, Tokuyama T, Yokota N, Nishizawa S, Namba H. Cdc6 expression as a marker of proliferative activity in brain tumors. Oncol Rep. 2001;8(5):1063–6. https://doi.org/10.3892/or.8.5.1063.
Stangeland B, Mughal AA, Grieg Z, Sandberg CJ, Joel M, Nygard S, et al. Combined expressional analysis, bioinformatics and targeted proteomics identify new potential therapeutic targets in glioblastoma stem cells. Oncotarget. 2015;6(28):26192–215. https://doi.org/10.18632/oncotarget.4613.
Young A, Berry R, Holloway AF, Blackburn NB, Dickinson JL, Skala M, et al. RNA-seq profiling of a radiation resistant and radiation sensitive prostate cancer cell line highlights opposing regulation of DNA repair and targets for radiosensitization. BMC Cancer. 2014;14:808. https://doi.org/10.1186/1471-2407-14-808.
Yu X, Liu Y, Yin L, Peng Y, Peng Y, Gao Y, et al. Radiation-promoted CDC6 protein stability contributes to radioresistance by regulating senescence and epithelial to mesenchymal transition. Oncogene. 2019;38(4):549–63. https://doi.org/10.1038/s41388-018-0460-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Research involving human participants and/or animals
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12094_2020_2449_MOESM1_ESM.pdf
Figure S1 CDC6 protein level is elevated in high-grade GBM tumor samples compared with that in low-grade GBM tumor samples. (PDF 11102 kb)
12094_2020_2449_MOESM2_ESM.pdf
Figure S2 Time-dependent ROC curves for 1-year OS and D2P (A) and 3-year OS and D2P (B) of GBM samples for estimating the performance of the combination of age, radiation, temozolomide chemotherapy, CDC6, and CIMP status in prognosis predicting. AUC values were provided. TP, True positive. FP, False positive. Rad, Radiation. TMZ, temozolomide chemotherapy. (PDF 432 kb)
Rights and permissions
About this article
Cite this article
Zhao, H., Zhou, X., Yuan, G. et al. CDC6 is up-regulated and a poor prognostic signature in glioblastoma multiforme. Clin Transl Oncol 23, 565–571 (2021). https://doi.org/10.1007/s12094-020-02449-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02449-w