Abstract
Purpose
We analysed our initial experience with SBRT in liver metastasis from colorectal cancer at our institution.
Materials and methods
Between January/2014 and December/2017, 22 patients with 31 LMCCR were treated. Local control (LC) was assessed using the Kaplan–Meier and log-rank tests. We analysed potential prognostic factors for LC: sex, PTV size, number of LM and the radiation scheme.
Results
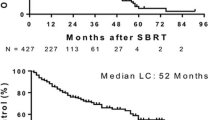
Median age: 69 years. Prior chemotherapy or local liver treatments: 81.8% and 63.6% of patients, respectively. SBRT consisted of 3 × 20 Gy (42.9%) and 3 × 15 Gy (31.4%). There were 88.5% responses (57.1% CR and 31.4% PR). Median follow-up was 30 months. LC per lesion at 12 and 24 months was 85.3% and 61.8%, respectively. Tumour volumes > 30 cc correlated with worsened 2-year-control rates (90% vs 34.5%) (p = 0.005). There was only a patient with CTC-grade 3 toxicity.
Conclusions
Liver SBRT is a safe and effective treatment that achieves high local control rates. We found a significant correlation between larger LMCRC and worse local control.



Similar content being viewed by others
References
Weiss L, Grundmann E, Torhorst J, Hartveit F, Moberg I, Eder M, et al. Haematogenous metastatic patterns in colonic carcinoma: an analysis of 1541 necropsies. J Pathol. 1986;150(3):195–203.
Bellier J, De Wolf J, Hebbar M, El Amrani M, Desauw C, Leteurtre E, Truant S. Repeated resections of hepatic and pulmonary metastases from colorectal cancer provide long-term survival. World J Surg. 2018;42:1171–9.
Quan D, Gallinger S, Nhan C, Auer RA, Biagi JJ, Fletcher GG, et al. The role of liver resection for colorectal cancer metastases in an era of multimodality treatment: a systematic review. Surgery. 2012;151:860–70.
Chow FC, Chok KS. Colorectal liver metastases: An update on multidisciplinary approach. World J Hepatol. 2019;11:150–72.
Gillams AR, Lees WR. Five-year survival in 309 patients with colorectal liver metastases treated with radiofrequency ablation. Eur Radiol. 2009;19:1206–13.
Cirocchi R, Trastulli S, Boselli C, Montedori A, Cavaliere D, Parisi A, et al. Radiofrequency ablation in the treatment of liver metastases from colorectal cancer. Cochrane Database Syst Rev. 2012;6:CD006317.
Dawood O, Mahadevan A, Goodman KA. Stereotactic body radiation therapy for liver metastases. Eur J Cancer. 2009;45(17):2947–59.
Rusthoven KE, Kavanagh BD, Cardenes H, Stieber VW, Burri SH, Feigenberg SJ, et al. Multi-institutional phase I/II trial of stereotactic body radiation therapy for liver metastases. J Clin Oncol. 2009;27:1572–8.
Habermehl D, Herfarth KK, Bermejo JL, Hof H, Rieken S, Kuhn S, et al. Single-dose radiosurgical treatment for hepatic metastases—therapeutic outcome of 138 treated lesions from a single institution. Radiat Oncol. 2013;8:175.
Berber B, Ibarra R, Snyder L, Yao M, Fabien J, Milano MT, et al. Multicentre results of stereotactic body radiotherapy for secondary liver tumours. HPB. 2013;15:851–7.
Timmerman RD. An overview of hypofractionation and introduction to this issue of seminars in radiation oncology. Radiat Oncol. 2008;18(4):215–22.
Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. U.S. Department of Health and Human Services. 2017. https://ctep.cancer.gov-docs/CTCAE_v5_Quick_Reference_5x7.
Schwartz LH, Litière S, de Vries E, Bogaerts J, Chen A, Dancey J, et al. RECIST 1.1-Update and clarification: from the RECIST committee. Eur J Cancer. 2016;62:132–7.
Khajornjiraphan N, Thu NA, Chow PK. Yttrium-90 microspheres: a review of its emerging clinical indications. Liver Cancer. 2015;4(1):6–15. https://doi.org/10.1159/000343876.
Schlick CJR, Merkow RP, Bentrem DJ. Nonresectional regional therapies for metastatic colorectal cancer to the liver. J Surg Oncol. 2019;119:636–41.
Chang DT, Swaminath A, Kozak M, Weintraub J, Koong AC, Kim J, et al. Stereotactic body radiotherapy for colorectal liver metastases: a pooled analysis. Cancer. 2011;117:4060–9.
Scorsetti M, Comito T, Tozzi A, Navarria P, Fogliata A, Clerici E, et al. Final results of a phase II trial for stereotactic body radiation therapy for patients with inoperable liver metastases from colorectal cancer. J Cancer Res Clin Oncol. 2015;141:543–53.
Van Der Pool AE, Mendez Romero A, Wunderink W, Heijmen BJ, Levendag PC, Verhoef C, et al. Stereotactic body radiation therapy for colorectal liver metastases. Br J Surg. 2010;97:377–82.
Hoyer M, Roed H, Traberg Hansen A, Ohlhuis L, Petersen J, Nellemann H, et al. Phase II study on stereotactic body radiotherapy of colorectal metastases. Acta Oncol. 2006;45:823–30.
Joo JH, Park J, Kim JC, Yu CS, Lim SB, Park IJ, et al. Radiación therapy for liver metastases from colorectal cancer. Int J Radiat Oncol Biol Phys. 2017;99(4):876–83.
Bujold A, Massey CA, Kim JJ, Brierley J, Cho C, Wong RK, et al. Sequential phase I and II trials of stereotactic body radiotherapy for locally advanced hepatocellular carcinoma. J Clin Oncol. 2013;31:1631–9.
Feng M, Suresh K, Schipper MJ, Bazzi L, Ben-Josef E, Matuszak MM, et al. Individualized adaptive stereotactic body radiotherapy for liver tumors in patients at high risk for liver damage: a phase 2 clinical trial. JAMA Oncol. 2018;4:40–7.
Moon DH, Wang AZ, Tepper JE. A prospective study of the safety and efficacy of liver stereotactic body radiotherapy in patients with and without prior liver-directed therapy. Radiother Oncol. 2018;126:527–33.
Takeda A, Sanuki N, Tsurugai Y, Iwabuchi S, Matsunaga K, Ebinuma H, et al. Phase 2 study of stereotactic body radiotherapy and optional transarterial chemoembolization for solitary hepatocellular carcinoma not amenable to resection and radiofrequency ablation. Cancer. 2016;122:2041–9.
Lasley FD, Mannina EM, Johnson CS, Perkins SM, Althouse S, Maluccio M, et al. Treatment variables related to liver toxicity in patients with hepatocellular carcinoma, Child-Pugh class A and B enrolled in a phase 1–2 trial of stereotactic body radiation therapy. Pract Radiat Oncol. 2015;5:e443–e44949.
Méndez Romero A, Wunderink W, Hussain SM, De Pooter JA, Heijmen BJ, Nowak PC, et al. Stereotactic body radiation therapy for primary and metastatic liver tumors: a single institution phase I–II study. Acta Oncol. 2006;45:831–7.
Stintzing S, Grothe A, Hendrich S, Hoffmann RT, Heinemann V, Rentsch M, et al. Percutaneous radiofrequency ablation (RFA) or robotic radiosurgery (RRS) for salvage treatment of colorectal liver metastases. Acta Oncol. 2013;52(5):971–7.
Jackson WC, Tao Y, Mendiratta-Lala M, Bazzi L, WahlDR SMJ, et al. Comparison of stereotactic body radiation therapy and radiofrequency ablation in the treatment of intrahepatic metastases. Int J Radiat Oncol Biol Phys. 2018;100(4):950–8.
Petrelli F, Comito T, Barni S, Pancera G, Scorsetti M, Ghidini A. Stereotactic body radiotherapy for colorectal cancer liver metastases: a systematic review. Radiother Oncol. 2018;129(3):427–34.
Joo JH, Park JH, Kim JC, Yu CS, Lim SB, Park IJ, et al. Local control outcomes using stereotactic body radiation therapy for liver metastases from colorectal cancer. Int J Radiat Oncol Biol Phys. 2017;99(4):876–83.
Méndez Romero A, Keskin-Cambay F, van Os RM, Nuyttens JJ, Heijmen BJM, Ijzermans JNM, et al. Institutional experience in the treatment of colorectal liver metastases with stereotactic body radiation therapy. Rep Pract Oncol Radiother. 2017;22(2):126–31.
Vera R, González-Flores E, Rubio C, Urbano J, Valero Camps M, Ciampi-Dopazo JJ. Multidisciplinary management of liver metastases in patients with colorectal cancer: a consensus of SEOM, AEC, SEOR, SERVEI, and SEMNIM. Clin Transl Oncol. 2020;22:647–62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Informed consent
All participants included in the study signed the informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Flamarique, S., Campo, M., Asín, G. et al. Stereotactic body radiation therapy for liver metastasis from colorectal cancer: size matters. Clin Transl Oncol 22, 2350–2356 (2020). https://doi.org/10.1007/s12094-020-02375-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02375-x