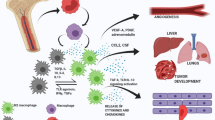
Abstract
Cancer is a health issue causing utmost concern and continuing to be one of the leading causes of mortality worldwide. Effective tumor eradication methods that will improve the prognosis and prolong human life are an important topic in modern medicine. Increasing amounts of evidence indicate that the tumor microenvironment plays a significant role in tumor development and migration. Macrophages are important immune cells that commonly infiltrate the tumor microenvironment. Several studies found that macrophages play different roles in the process of cancer development. This article focuses on the tumor microenvironment and the generation, classification, and function of tumor-associated macrophages as well as their significance for tumor immunotherapy and other aspects, it summarizes nearly 10 years of tumor microenvironment and tumor-associated macrophage research, providing a novel insight for tumor immunotherapy.




Similar content being viewed by others
Abbreviations
- TAMs:
-
Tumor-associated macrophage
- ECM:
-
Extracellular matrix
- α-SMA:
-
Alpha-smooth muscle actin
- MMPS:
-
Matrix metalloproteinases
- CAFs:
-
Cancer-associated fibroblasts
- TEMs:
-
Tumor endothelial markers
- MDSCs:
-
Myeloid-derived suppressor cells
References
Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3(6):453–8. https://doi.org/10.1038/nrc1098.
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16(9):582–98. https://doi.org/10.1038/nrc.2016.73.
Polyak K, Haviv I, Campbell IG. Co-evolution of tumor cells and their microenvironment. Trends Genet. 2009;25(1):30–8. https://doi.org/10.1016/j.tig.2008.10.012.
Hede K. Environmental protection: studies highlight importance of tumor microenvironment. J Natl Cancer I. 2004;96(15):1120–1. https://doi.org/10.1093/jnci/96.15.1120.
Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008;27(45):5904–12. https://doi.org/10.1038/onc.2008.271.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. https://doi.org/10.1016/j.cell.2011.02.013.
De Wever O, Van Bockstal M, Mareel M, Hendrix A, Bracke M. Carcinoma-associated fibroblasts provide operational flexibility in metastasis. Semin Cancer Biol. 2014;25:33–46. https://doi.org/10.1016/j.semcancer.2013.12.009.
Bergamaschi A, Tagliabue E, Srlie T, Naume B, Triulzi T, Orlandi R, Russnes HG, Nesland JM, Tammi R, Auvinen P, Kosma VM, Ménard S, Børresen-Dale AL. Extracellular matrix signature identifies breast cancer subgroups with different clinical outcome. J Pathol. 2008;214(3):357–67. https://doi.org/10.1002/path.2278.
Ma XJ, Dahiya S, Richardson E, Erlander M, Sgroi DC. Gene expression profiling of the tumor microenvironment during breast cancer progression. Breast Cancer Res. 2009;11(1):R7. https://doi.org/10.1186/bcr2222.
Dvorak HF. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. New Engl J Med. 1986;315(26):1650–9. https://doi.org/10.1158/2326-6066.CIR-14-0209.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860–7. https://doi.org/10.1038/nature01322.
De Wever O, Mareel M. Role of tissue stroma in cancer cell invasion. J Pathol. 2003;200(4):429–47. https://doi.org/10.1002/path.1398.
Nazareth MR, Broerick L, Simpson-Abelson MR, Kelleher RJ Jr, Yokota SJ, Bankert RB. Charaterization of human lung tumor-associated fibroblasts and their ability to modulate the activation of tumor-associated T cells. J Immunol. 2007;178(9):5552–62. https://doi.org/10.4049/jimmunol.178.9.5552.
Gonda TA, Varro A, Wang TC, Tycko B. Molecular biology of cancer-associated fibroblasts: can these cells be targeted in anti-cancer therapy? Semin Cell Dev Biol. 2010;21(1):2–10. https://doi.org/10.1016/j.semcdb.2009.10.001.
Martinez-Outschoorn UE, Lisanti MP, Sotgia F. Catabolic cancer-associated fibroblasts transfer energy and biomass to anabolic cancer cells, fueling tumor growth. Semin Cancer Biol. 2014;25(2):47–60. https://doi.org/10.1016/j.semcancer.2014.01.005.
Junttila MR, de Sauvage FJ. Influence of tumour microenvironment heterogeneity on therapeutic response. Nature. 2013;501(7467):346–54. https://doi.org/10.1038/nature12626.
Pena C, Cespedes MV, Lindh MB, Kiflemariam S, Mezheyeuski A, Edqvist PH, Hägglöf C, Birgisson H, Bojmar L, Jirström K, Sandström P, Olsson E, Veerla S, Gallardo A, Sjöblom T, Chang AC, Reddel RR, Mangues R, Augsten M, Ostman A. STC1 expression by cancer associated fibroblasts drives metastasis of colorectal cancer. Cancer Res. 2013;73(4):1287–97. https://doi.org/10.1158/0008-5472.CAN-12-1875.
Augsten M, Hagglof C, Olsson E, Stolz C, Tsagozis P, Levchenko T, Frederick M, Borg Å, Micke P, Egevad L, Östman A. CXCL14 is an autocrine growth factor for fbroblasts and acts as a multi-modal stimulator of prostate tumor growth. Proc Natl Acad Sci USA. 2009;106(9):3414–9. https://doi.org/10.1073/pnas.0813144106.
Augsten M, Sjoberg E, Frings O, Vorrink SU, Frijhoff J, Olsson E, Borg Å, Östman A. Cancer-associated fibroblasts expressing CXCL14 rely upon NOS1-derived nitric oxide signaling for their tumor-supporting properties. Cancer Res. 2014;74(11):2999–3010. https://doi.org/10.1158/0008-5472.CAN-13-2740.
Ribatti D, Nico B, Crivellato E, Roccaro AM, Vacca A. The history of angiogenic switch concept. Leukemia. 2007;21(1):44–52. https://doi.org/10.1038/sj.leu.2404402.
St Croix B, Rago C, Velculescu V, Traverso G, Romans KE, Montgomery E, Lal A, Riggins GJ, Lengauer C, Vogelstein B, Kinzler KW. Genes expressed in human tumor endothelium. Science. 2000;289(5482):1197–2002. https://doi.org/10.1126/science.289.5482.1197.
Dirat B, Bochet L, Dabek M, Daviaud D, Dauvillier S, Majed B, Wang YY, Meulle A, Salles B, Le Gonidec S, Garrido I, Escourrou G, Valet P, Muller C. Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Res. 2011;71(7):2455–65. https://doi.org/10.1158/0008-5472.CAN-10-3323.
Gajewski TF, Schreiber H, Fu YX. Innate and adaptive immune cells in the tumor microenvironment. Nat immunol. 2013;14(10):1014–22. https://doi.org/10.1038/ni.2703.
Talmadge JE, Gabrilovich DI. History of myeloid-derived suppressor cells. Nat Rev Cancer. 2013;13(10):739–52. https://doi.org/10.1038/nrc3581.
Trikha P, Carson EW III. Signaling pathways involved in MDSC regulation. Biochim Biophys Acta. 2014;1846(1):55–65. https://doi.org/10.1016/j.bbcan.2014.04.003.
Mao Y, Sarhan D, Steven A, Seliger B, Kiessling R, Lundqvist A. Inhibition of tumor-derived prostaglandin-e2 blocks the induction of myeloid-derived suppressor cells and recovers natural killer cell activity. Clin Cancer Res. 2014;20(15):4096–106. https://doi.org/10.1158/1078-0432.CCR-14-0635.
Yu F, Shi Y, Wang J, Li J, Fan DP, Ai WD. Deficiency of Kruppel-like factor KLF4 in mammary tumor cells inhibits tumor growth and pulmonary metastasis and is accompanied by compromised recruitment of myeloid-derived suppressor cells. Int J Cancer. 2013;133(12):2872–83. https://doi.org/10.1002/ijc.28302.
Wang J, Yu F, Jia X, Iwanowycz S, Wang Y, Huang S, Ai W, Fan D. MicroRNA-155 deficiency enhances the recruitment and functions of myeloid-derived suppressor cells in tumor microenvironment and promotes solid tumor growth. Int J Cancer. 2015;136(6):602–13. https://doi.org/10.1002/ijc.29151.
Liu J, Zhang Y, Zhao J, Yang Z, Li D, Katirai F, Huang B. Mast cell: insight into remodeling a tumor microenvironment. Cancer Metastasis Rev. 2011;30(2):177–84. https://doi.org/10.1007/s10555-011-9276-1.
Saleem SJ, Martin RK, Morales JK, Sturgill JL, Gibb DR, Graham L, Bear HD, Manjili MH, Ryan JJ, Conrad DH. Cutting edge: mast cells critically augment myeloid-derived suppressor cell activity. J Immunol. 2012;189(2):511–5. https://doi.org/10.4049/jimmunol.1200647.
Spano D, Zollo M. Tumor microenvironment: a main actor in the metastasis process. Clin Exp Metastasis. 2012;29(4):381–95. https://doi.org/10.1007/s10585-012-9457-5.
Gillies ER, Goodmin AP, Frechet JMJ. Acetals as pH-sensitive linkages for drug delivery. Bioconjugate chem. 2004;15(16):1254–63. https://doi.org/10.1016/S1350-4487(03)00156-2.
Felber AE, Dufresne MH, Leroux JC. pH-sensitive Vesicles, polymeric micelles, and nanospheres prepared with polycarboxylates. Adv Drug Deliver Rev. 2011;64(11):979–92. https://doi.org/10.1016/j.addr.2011.09.006.
Lisanti MP, Martinez-Outschoorn UE, Sotgia F. Oncogenes induce the cancer-associated fibroblast phenotype metabolic symbiosis and “fibroblast addiction” are new therapeutic targets for drug discovery. Cell Cycle. 2013;12(17):2723–32. https://doi.org/10.4161/cc.25695.
Gordon S, Martinez FO. Alternative activation of macrophages: mechanism and functions. Immunity. 2010;32(5):593–604. https://doi.org/10.1016/j.immuni.2010.05.007.
Gordon S, Plüddemann A, Martinez EF. Macrophage heterogeneity in tissues: phenotypic diversity and functions. Immunol Rev. 2015;262(1):36–55. https://doi.org/10.1111/imr.12223.
Stein M, Keshav S, Harris N, Gordon S. Interleukin 4 potently enhances murine macrophage mannose receptor activity: a marker of alternative immunologic macrophage activation. J Exp Med. 1992;176(1):287–92. https://doi.org/10.1084/jem.176.1.287.
Gordon S, Mantovani A. Diversity and plasticity of mononuclear phagocytes. Eur J Immunol. 2011;41(9):2470–2. https://doi.org/10.1002/eji.201141988.
Mantovani A, Biswas SK, Galdiero MR, Sica A, Locati M. Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol. 2013;229(2):176–85. https://doi.org/10.1002/path.4133.
Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11(10):889–96. https://doi.org/10.1038/ni.1937.
Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8(12):958–69. https://doi.org/10.1038/nri2448.
Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436–44. https://doi.org/10.1038/nature07205.
Gordon S, Taylor PR. Monocyte and macrophage heterogeneity. Nat Rev Immunol. 2005;5(12):953–64. https://doi.org/10.1038/nri1733.
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23(11):549–55. https://doi.org/10.1016/S1471-4906(02)02302-5.
Hume DA. The many alternative faces of macrophage activation. Front Immunol. 2015;6:370. https://doi.org/10.3389/fimmu.2015.00370.
Gordon S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3:23–35. https://doi.org/10.1038/nri978.
Pollard JW. Trophic macrophages in development and disease. Nat Rev Immunol. 2009;9(4):259–70. https://doi.org/10.1038/nri2528.
Eubank TD, Roberts RD, Khan M, Curry JM, Nuovo GJ, Kuppusamy P, Marsh CB. Granulocyte macrophage colony-stimulating factor inhibits breast cancer growth and metastasis by invoking an anti-angiogenic program in tumor-educated macrophages. Cancer Res. 2009;69(5):2133–40. https://doi.org/10.1158/0008-5472.CAN-08-1405.
Mantovani A, Sica A. Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr Opin Immunol. 2010;22(2):231–7. https://doi.org/10.1016/j.coi.2010.01.009.
Ryder M, Ghossein RA, Ricarte-Filho JC, Knauf JA, Fagin JA. Increased density of tumor-associated macrophages is associated with decreased survival in advanced thyroid cancer. Endocr-Relat Cancer. 2008;15(4):1069–74. https://doi.org/10.1677/ERC-08-0036.
Muraille E, Leo O, Moser M. TH1/TH2 paradigm extended: macrophage polarization as an unappreciated pathogen-driven escape mechanism? Front Immunol. 2014;5:603. https://doi.org/10.3389/fimmu.2014.00603.
Caronni N, Savino B, Bonecchi R. Myeloid cells in cancer-related inflammation. Immunobiology. 2015;220(2):249–53. https://doi.org/10.1016/j.imbio.2014.10.001.
Mantovani A, Locati M. Tumor-associated macrophages as a paradigm of macrophage plasticity, diversity, and polarization: lessons and open questions. Arterioscl Throm Vas. 2013;33(7):1478–83. https://doi.org/10.1161/atvbaha.113.300168.
Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25(12):677–86. https://doi.org/10.1016/j.it.2004.09.015.
Chen S, Yang X, Feng W, Yang F, Wang R, Chen C, Wang L, Lin Y, Ren Q, Zheng G. Characterization of peritoneal leukemia-associated macrophages in Notch 1 induced mouse T cell acute lymphoblastic leukemia. Mol Immunol. 2017;81:35–41. https://doi.org/10.1016/j.molimm.2016.11.014.
Balkwill FR, Mantovani A. Cancer-related inflammation: common themes and therapeutic opportunities. Semin Cancer Biol. 2012;22(1):33–40. https://doi.org/10.1016/j.semcancer.2011.12.005.
Allavena P, Sica A, Garlanda C, Mantovani A. The Yin-Yang of tumor-associated macrophages in neoplastic progression and immune surveillance. Immunol Rev. 2008;222(1):155–61. https://doi.org/10.1111/j.1600-065X.2008.00607.x.
Liu Y, Cao X. The origin and function of tumor-associated macrophages. Cell Mol Immunol. 2015;12(1):1–4. https://doi.org/10.1038/cmi.2014.83.
Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012;122(3):787–95. https://doi.org/10.1172/JCI59643.
Wang B, Li Q, Qin L, Zhao S, Wang J, Chen X. Transition of tumor-associated macrophages from MHC class II(hi) to MHC class II(low) mediates tumor progression in mice. BMC Immunol. 2011;12(1):43. https://doi.org/10.1186/1471-2172-12-43.
Riabov V, Gudima A, Wang N, Mickley A, Orekhov A, Kzhyshkowska J. Role of tumor associated macrophages in tumor angiogenesis and lymphangiogenesis. Front Physiol. 2014;5:75. https://doi.org/10.3389/fphys.2014.00075.
Lin EY, Li JF, Gnatovskiy L, Deng Y, Zhu L, Grzesik DA, Qian H, Xue XN, Pollard JW. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. 2006;66(23):11238–46. https://doi.org/10.1158/0008-5472.CAN-06-1278.
Chen P, Huang Y, Bong R, Ding Y, Song N, Wang X, Song X, Luo Y. Tumor-associated macrophages promote angiogenesis and melanoma growth via adrenomedullin in a paracrine and autocrine manner. Clin Cancer Res. 2011;17(23):7230–9. https://doi.org/10.1158/1078-0432.CCR-11-1354.
Bingle L, Lewis CE, Corke KP, Reed MWR, Brown NJ. Macrophages promote angiogenesis in human breast tumour spheroids in vivo. Brit J Cancer. 2006;94(1):101–7. https://doi.org/10.1038/sj.bjc.6602901.
Huang S, Van Arsdall M, Tedjarati S, McCarty M, Wu W, Langley R, Fidler IJ. Contributions of stromal metalloproteinase-9 to angiogenesis and growth of human ovarian carcinoma in mice. J Natl Cancer I. 2002;94(15):1134–42. https://doi.org/10.1093/jnci/94.15.1134.
Guiet R, Van Goethem E, Gugoule C, Balor S, Valette A, Al Saati T, Lowell CA, Le Cabec V, Maridonneau-Parini I. The process of macrophage migration promotes matrix metalloproteinase-independent invasion by tumor cells. J Immunol. 2011;187(7):3806–14. https://doi.org/10.4049/jimmunol.1101245.
Wyckoff JB, Wang Y, Lin EY, Li JF, Goswami S, Stanley ER, Segall JE, Pollard JW, Condeelis J. Direct visualization of macrophage assisted tumor cell intravasation in mammary tumors. Cancer Res. 2007;67(6):2649–56. https://doi.org/10.1158/0008-5472.CAN-06-1823.
Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, Stanley ER, Graf T, Pollard JW, Segall J, Condellis J. A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res. 2004;64(19):7022–9. https://doi.org/10.1158/0008-5472.CAN-04-1449.
Kim S, Takahashi H, Lin WW, Descargues P, Grivennikov S, Kim Y, Luo JL, Karin M. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature. 2009;457(7225):102–6. https://doi.org/10.1038/nature07623.
Gil-Bernabé AM, Ferjancic S, Tlalka M, Zhao L, Allen PD, Im JH, Watson K, Hill SA, Amirkhosravi A, Francis JL, Pollard JW, Ruf W, Muschel RJ. Recruitment of monocytes/macrophages by tissue factor-mediated coagulation is essential for metastatic cell survival and premetastatic niche establishment in mice. Blood. 2012;119(13):3164–75. https://doi.org/10.1182/blood-2011-08-376426.
Erler JT, Bennewith KL, Cox TR, Lang G, Bird D, Koong A, Le QT, Giaccia AJ. Hypoxia-induced lysyl-oxidase is a critical mediator of bone marrow cell recruitment to form the pre-metastatic niche. Cancer Cell. 2009;15(1):35–44. https://doi.org/10.1016/j.ccr.2008.11.012.
Parker KH, Sinha P, Horn LA, Clements VK, Yang H, Li J, Tracey KJ, Ostrand-Rosenberg S. HMGB1 enhances immune suppression by facilitating the differentiation and suppressive activity of myeloid-derived suppressor cells. Cancer Res. 2014;74(20):5723–33. https://doi.org/10.1158/0008-5472.CAN-13-2347.
Kuang DM, Zhao Q, Peng C, Xu J, Zhang JP, Wu C, Zheng L. Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J Exp Med. 2009;206(6):1327–37. https://doi.org/10.1084/jem.20082173.
Van Esch EM, van Poelgeest MI, Trimbos JB, Fleuren GJ, Jordanova ES, van der Burg SH. Intraepithelial macrophage infiltration is related to a high number of regulatory T cells and promotes a progressive course of HPV-induced vulvar neoplasia. Int J Cancer. 2015;136(4):E85–E94. https://doi.org/10.1002/ijc.29173.
Eruslanov E, Stoffs T, Kim WJ, Daurkin I, Gilbert SM, Su LM, Vieweg J, Daaka Y, Kusmartsev S. Expansion of CCR8+ inflammatory myeloid cells in cancer patients with urothelial and renal carcinomas. Clin Cancer Res. 2013;19(7):1670–80. https://doi.org/10.1158/1078-0432.CCR-12-2091.
Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006;5(6):471–84. https://doi.org/10.1038/nrd2059.
Green DR, Ferguson T, Zitvogel L, Kroemer G. Immunogenic and tolerogenic cell death. Nat Rev Immunol. 2009;9(5):353–63. https://doi.org/10.1038/nri2545.
Acknowledgements
We thank all the cited authors for providing the research results for our reference.
Funding
This research was supported by Youth Science Foundation of Guangxi Medical University (GXMUYSF201612), the National Natural Science Foundation of China (Grant No. 81560446) and the National Natural Science Foundation of Guangxi (Grant No. 2016GXNSFBA380157).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval (research involving human participants and/or animals)
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, W., Huang, X., Carlos, C.J.J. et al. Research progress of tumor microenvironment and tumor-associated macrophages. Clin Transl Oncol 22, 2141–2152 (2020). https://doi.org/10.1007/s12094-020-02367-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02367-x