Abstract
Objective
To validate the Catalan minimum basic data set (MBDS) of hospital discharges as an information source for detecting incident breast (BC) and colorectal cancer (CRC), against the Hospital del Mar Cancer Registry (RTHMar) in Barcelona (Spain) as the gold standard.
Methods
Using ASEDAT software (Analysis, Selection and Extraction of Tumour Data), we identified Catalan public hospital discharge abstracts in patients with a first-time diagnosis of BC and CRC in the years 2005, 2008, and 2011, aggregated by unique patient identifiers and sorted by date. Once merged with the RTHMar database and anonymized, tumour-specific algorithms were validated to extract data on incident cases, tumour stage, surgical treatment, and date of incidence.
Results
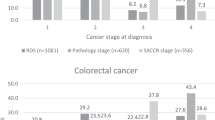
MBDS had a respective sensitivity and positive predictive value (PPV) of 78.0% (564/723) and 90.5% (564/623) for BC case detection; and 83.9% (387/461) and 94.9% (387/408) for CRC case detection. The staging algorithms overestimated the proportion of local-stage cases and underestimated the regional-stage cases in both cancers. When loco-regional stage and surgery were combined, sensitivity and PPV reached 98.3% and 99.8%, respectively, for BC and 96.4% and 98.4% for CRC. The differences between dates of incidence between RTHMar and MBDS were greater for BC cases without initial surgery, whereas they were generally smaller and homogeneous for CRC cases.
Conclusions
The MBDS is a valid and efficient instrument to improve the completeness of a hospital-based cancer registry (HBCR), particularly in BC and CRC, which require hospitalization and are predominantly surgical.

Similar content being viewed by others
Abbreviations
- MBDS:
-
Catalan minimum basic data set of hospital discharge
- BC:
-
Breast cancer
- CRC:
-
Colorectal cancer
- RTHMar:
-
Hospital del Mar Cancer Registry
- ASEDAT:
-
Analysis, Selection and Extraction of Tumour data
- PPV:
-
Positive predictive value
- HBCR:
-
Hospital-based cancer registry
- ICD-9-CM:
-
International Classification of Diseases, 9th revision, Clinical Modification
- ICD-10-CM/PCS:
-
International Statistical Classification of Disease and Related Health Problems, 10th revision
- HMar:
-
Hospital Parc de Salut MAR
- ICD-O-1:
-
International Classification of Diseases for Oncology, 1st edition
- CI:
-
Confidence interval
- FP:
-
False positive
- FN:
-
False negative
- PBCR:
-
Population-based cancer registry
References
Bernal Delgado E, Martos C, Martínez N, Chirlaque MD, Márquez M, Navarro C, et al. Is hospital discharge administrative data an appropriate source of information for cancer registries purposes? Some insights from four Spanish registries. BMC Health Serv Res. 2010;10:9. https://doi.org/10.1186/1472-6963-10-9.
Abraha I, Orso M, Grilli P, Cozzolino F, Eusebi P, Casucci P, et al. The current state of validation of administrative healthcare databases in Italy: a systematic review. Int J Stat Med Res. 2014;3:309–20.
Penberthy L, McClish D, Pugh A, Smith W, Manning C, Retchin S. Using hospital discharge files to enhance cancer surveillance. Am J Epidemiol. 2003;158:27–34.
Creighton N, Walton R, Roder D, Aranda S, Currow D. Validation of administrative hospital data for identifying incident pancreatic and periampullary cancer cases: a population-based study using linked cancer registry and administrative hospital data in New South Wales, Australia. BMJ Open. 2016;6:e011161. https://doi.org/10.1136/bmjopen-2016-011161.
Fernández Navarro P, López Abente G, Salido Campos C, Sanz Anquela JM. The minimum basic data set (MBDS) as a tool for cancer epidemiological surveillance. Eur J Int Med. 2016;34:94–7.
Abraha I, Serraino D, Montedori A, Fusco M, Giovaninni G, Casucci P, et al. Sensitivity and specificity of breast cancer ICD-9-CM codes in three Italian administrative healthcare databases: a diagnostic accuracy study. BMJ Open. 2018;8:e020627. https://doi.org/10.1136/bmjopen-2017-020627.
Abraha I, Montedori A, Serraino D, Orso M, Giovaninni G, Scotti V, et al. Accuracy of administrative databases in detecting primary breast cancer diagnoses: a systematic review. BMJ Open. 2018;8:e019264. https://doi.org/10.1136/bmjopen-2017-019264.
Cozzolino F, Bidoli E, Abraha I, Fusco M, Giovannini G, Casucci P, et al. Accuracy of colorectal cancer ICD-9-CM codes in Italian administrative healthcare databases: a cross-sectional diagnostic study. BMJ Open. 2018;8:e020630. https://doi.org/10.1136/bmjopen-2017-020630.
Goldsbury D, Weber M, Yap S, Banks E, O’Connell DL, Canfell K. Identifying incident colorectal and lung cancer cases in health service utilization databases in Australia: a validation study. BMC Med Inform Decis Mak. 2017;17:23. https://doi.org/10.1186/s12911-017-0417-5.
Goldsbury DE, Armstrong K, Simonella L, Armstrong BK, 0′Connell DL. Using administrative health data to describe colorectal and lung cancer care in New South Wales, Australia: a validation study. BMC Health Serv Res. 2012;12:387–95.
Kemp A, Preen DB, Saunders C, D’Arcy C, Holman J, Bulsara M. Ascertaining invasive breast cancer cases; the validity of administrative and self-reported data sources in Australia. BMC Med Res Methodol. 2013;13:17. https://doi.org/10.1186/1471-2288-13-17.
Diop M, Strumpf EC, Datta GD. Measuring colorectal cancer incidence: the performance of an algorithm using administrative health data. BMC Med Res Methodol. 2018;18:38. https://doi.org/10.1186/s12874-018-0494-x.
Benchimol EI, Manuel DG, To T, Griffiths AM, Rabeneck L, Guttmann A. Development and use of reporting guidelines for assessing the quality of validation studies of health administrative data. J Clin Epidemiol. 2011;64:821–9.
Korevaar D, Cohen JF, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, et al. Updating standards for reporting diagnostic accuracy: the development of STARD 2015. Res Integr Peer Rev. 2016;1:7. https://doi.org/10.1186/s41073-016-0014-7.
Smith GL, Shih YT, Giordano SH, Smith BD, Buchholz TA. A method to predict breast cancer stage using medicare claims. Epidemiol Perspect Innov. 2010;7:1–9.
Yuen E, Louis D, Cisbani L, Rabinowitz C, De Palma R, Maio V, et al. Using administrative data to identify and stage breast cancer cases: implications for assessing quality of care. Tumori. 2011;97:428–35.
Thompson B, Lumley J, Coory M. Hospital morbidity data for determining spread of disease at diagnosis for colorectal cancer: a validation study. Asia Pac J Clin Oncol. 2012;8:e17–22. https://doi.org/10.1111/j.1743-7563-2012.01537.x.
Chawla N, Yabroff KR, Mariotto A, McNeel TS, Schrag D, Warren JL. Limited validity of diagnosis codes in medicare claims for identifying cancer metastases and inferring stage. Ann Epidemiol. 2014;24:666–72.
Nordstrom BL, Whyte JL, Stolar M, Mercaldi C, Kallich JD. Identification of metastatic cancer in claims data. Pharmacoepidemiol Drug Saf. 2012;21(S2):21–8. https://doi.org/10.1002/pds.3247.
White JL, Engel-Nitz NM, Teitelbaum A, Gomez Rey G, Kallich JD. An evaluation of algorithms for identifying metastatic breast, lung, or colorectal cancer in administrative claims data. Med Care. 2015;53:e49–57.
Ribes J, Gálvez J, Melià A, Clèries R, Messeguer X, Bosch FX. Automatización de un registro hospitalario de tumores. Gac Sanit. 2005;19:221–8.
Escribà JM, Pareja L, Esteban L, Gálvez J, Melià A, Roca L, et al. Trends in the surgical procedures of women with incident breast cancer in Catalonia, Spain, over a 7-year period (2005–2011). BMC Res Notes. 2014;7:587. https://doi.org/10.1186/1756-0500-7-587.
Posso M, Macià F. Informe 2018 del Registro de Tumores del Hospital del Mar (RTHMar), Barcelona. Barcelona: Servei d'Epidemiologia i Avaluació, Parc de Salut Mar; septiembre 2018.
Baldi I, Vicari P, di Cuonzo D, Zanetti R, Pagano E, Rosato R. A high positive predictive value algorithm using hospital administrative data identified incident cancer cases. J Clin Epidemiol. 2008;61:373–9.
Couris CM, Polazzi S, Olive F, Remontet L, Bossard N, Gomez F, et al. Breast cancer incidence using administrative data: correction with sensitivity and specificity. J Clin Epidemiol. 2009;62:660–6.
Quantin C, Benzenine E, Fassa M, Hägi M, Fournier E, Gentil J, et al. Evaluation of the interest of using discharge abstract databases to estimate breast cancer incidence in two French departments. Stat J IAOS. 2012;28:73–85. https://doi.org/10.3233/SJI-2012-0742.
Quantin C, Benzenine E, Hägi M, Auverlot B, Abrahamowicz M, Cottenet J, et al. Estimation of national colorectal-cancer incidence using claims databases. J Cancer Epidemiol. 2012;2012:298369. https://doi.org/10.1155/2102/298369.
Dregan A, Moller H, Murray-Thomas T, Gulliford MC. Validity of cancer diagnosis in a primary care database compared with linked cancer registrations in England. Population-based cohort study. Cancer Epidemiol. 2012;36:425–9.
Brackley ME, Penning MJ, Lesperance ML. In the absence of cancer registry data, is it sensible to assess incidence using hospital separation records? Int J Equity Health. 2006;5:12. https://doi.org/10.1186/1475-9276-5-12.
Ganry O, Taleb A, Peng J, Raverdy N, Dubreuil A. Evaluation of an algorithm to identify incident breast cancer using DRGS data. Eur J Cancer Prev. 2003;12:295–9.
Márquez M, Valera I, Chirlaque MD, Tortosa J, Párraga E, Navarro C. Validación de los códigos diagnósticos de cáncer de colon y recto del Conjunto Mínimo Básico de Datos. Gac Sanit. 2006;20:266–72.
Helqvist L, Erichsen R, Gammelager H, Johansen MB, Sorensen HT. Quality of ICD-10 colorectal cancer diagnosis codes in the Danish National Registry of Patients. Eur J Cancer Care. 2012;21:722–7.
Schifano P, Papini P, Agabiti N, Scarinci M, Borgia P, Perucci CA. Indicators of breast cancer severity and appropriateness of surgery based on hospital administrative data in The Lazio Region, Italy. BMC Public Health. 2006;6:25. https://doi.org/10.1186/1471-2458-6-25.
Li X, King C, deGara C, White J, Winget M. Validation of colorectal cancer surgery data from administrative data sources. BMC Med Res Method. 2012;12:97–103.
Gil M, Rodríguez A, Montoya H, González R, Alvárez A, Rodriguez S, et al. Validation study of colorectal cancer diagnosis in the Spanish primary care database, BIFAP. Pharmacoepidemiol Drug Saf. 2019;28:209–16.
Acknowledgements
We thank A. Melià, L. Roca, and P. Rodríguez from the Catalan Cancer Planning Directorate, and C. Hernández from the RTHMar, for reviewing the information of cases included in this study. We would also like to thank M. Bustins from the Evaluation Unit of the Health Department of the Government of Catalonia for providing the Catalan minimum basic data set of hospital discharge. This study was partially funded by the Agency for Management of University and Research Grants (AGAUR), Government of Catalonia, Grant Number 2017SGR00450 (http://agaur.gencat.cat), under the authority of the Secretariat of Universities and Research, Ministry of Enterprise and Knowledge of the Government of Catalonia. Meggan Harris for editing and improving this manuscript.
Author information
Authors and Affiliations
Contributions
JME conceived and designed the study, collected and analysed data, and wrote the article. MB designed the study, collected and analysed data, and reviewed and approved the final version. FM designed the study, interpreted data and reviewed and approved the final version. JG and XS collected and prepared the data. RC, LE, and LP performed the statistical data analyses. JR designed the study, analysed the data, wrote the article, and approved the final version. XC and JMB critically reviewed and approved the final version. All authors approved the final version.The number of authors is justified as it is a collaborative project in which different health professionals have participated, including medical epidemiologists, technical computer engineers, biostatisticians, and trained documentation specialists for the review of cases between two institutions.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest for the present study. All the authors have carefully read the manuscript and fully approved it. The manuscript is original, and it has not been submitted to other journals.
Ethical approval
The current study has been performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was not required, because we used administrative databases with anonymized information to the researchers.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Escribà, J.M., Banqué, M., Macià, F. et al. Detection of incident breast and colorectal cancer cases from an administrative healthcare database in Catalonia, Spain. Clin Transl Oncol 22, 943–952 (2020). https://doi.org/10.1007/s12094-019-02219-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02219-3