Abstract
Purpose
The aim of the study was to evaluate the effectiveness of fentanyl pectin nasal spray (FPNS) in controlling procedural breakthrough cancer pain (BTCP) in advanced cancer patients undergoing radiotherapy.
Materials and methods
This study involved 62 advanced cancer patients, with well-controlled background pain, who presented BTCP associated to routine radiotherapy procedures, treated with FPNS according to our protocol of administration. The BPE intensity was measured using a visual analog scale (VAS).
Results
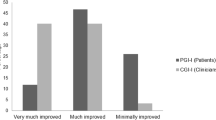
The BTCP was triggered during the computed tomography simulation (79.3%) or treatment delivery (20.7%). Patients indicated a mean VAS of 8.8 (range 7–10) when attempting the procedure. After 4.5 min (range 2–10) of the first FPNS dose, the majority of patients (85.5%) indicated a VAS of 4.3 (range 2–6). 15.5% of the patients did not respond after 15 min; requiring a second dose. All these patients responded, reporting a mean VAS of 4.2 (range 4–6) after 3.0 min (range 2–5) of the second dose. None of the patients required a third dose, nor reported an AE after the administration of FPNS.
Conclusions
In our knowledge, our study is the one of highest recruitment, and with the fastest response of BTCP treated with FPNS reported in advanced cancer patients undergoing radiotherapy. FPNS has proven to be highly effective in reducing the intensity of procedural BTCP in a very short period of time.

Similar content being viewed by others
References
Bruera E, Kim HN. Cancer pain. JAMA. 2003;290:2476–9.
Breivik H, Cherny N, Collett B, et al. Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes. Ann Oncol. 2009;20:1420–33.
Portenoy RK. Treatment of cancer pain. Lancet. 2011;377:2237–47.
Bell BC, Butler EB. Management of predictable pain using fentanyl pectin nasal spray in patients undergoing radiotherapy. J Pain Res. 2013;6:843–8.
Caraceni A, Hanks G, Kaasa S, et al. Use of opioid analgesics in the treatment of cancer pain: evidence-based recommendations from the EAPC. Lancet Oncol. 2012;13:58–68.
Davies AN, Dickman A, Reid C, et al. The management of cancer-related breakthrough pain: recommendations of a task group of the Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. Eur J Pain. 2009;13:331–8.
Caraceni A, Martini C, Zecca E, et al. Breakthrough pain characteristics and syndromes in patients with cancer pain. An international survey. Palliat Med. 2004;18:177–83.
Hjermstad MJ, Kaasa S, Caraceni A, et al. Characteristics of breakthrough cancer pain and its influence on quality of life in an international cohort of patients with cancer. BMJ Support Palliat Care. 2016;6:344–52.
Deandrea S, Corli O, Consonni D, et al. Prevalence of breakthrough cancer pain: a systematic review and a pooled analysis of published literature. J Pain Symptom Manag. 2014;47:57–76.
Davies A. Introduction. In: Davies A, editor. Cancer-related breakthrough pain. Oxford: Oxford University Press; 2006. p. 1–11.
Yaromina A, Krause M, Baumann M. Individualization of cancer treatment from radiotherapy perspective. Mol Oncol. 2012;6:211–21.
Mercadante S, Marchetti P, Cuomo A, et al. Breakthrough cancer pain: preliminary data of the Italian Oncologic Pain Multisetting Multicentric Survey (IOPS-MS). Adv Ther. 2017;34(1):120–35.
Caraceni A, Davies A, Poulain P, et al. Guidelines for the management of breakthrough pain in patients with cancer. J Natl Compr Cancer Netw. 2013;11:S29–36.
Taylor DR, Gabrail N. Fentanyl pectin nasal spray for breakthrough cancer pain. Future Oncol. 2012;8:121–30.
European Medicines Agency. PecFent (Fentanyl). http://www.ema.europa.eu/docs/es_ES/document_library/EPAR_-_Product_Information/human/001164/WC500096493.pdf. Accessed 16 Mar 18.
Watts P, Smith A. PecSys: in situ gelling system for optimised nasal drug delivery. Expert Opin Drug Deliv. 2009;6:543–52.
Portenoy RK, Raffaeli W, Torres LM, et al. Long-term safety, tolerability, and consistency of effect of fentanyl pectin nasal spray for breakthrough cancer pain in opioid-tolerant patients. J Opioid Manag. 2010;6:319–28.
Fallon M, Reale C, Davies A, et al. Efficacy, safety and patient acceptability of fentanyl pectin nasal spray compared with immediate-release morphine sulphate tablets in the treatment of breakthrough cancer pain: multicenter, double-blind, double-dummy, multiple crossover study. J Support Oncol. 2011;9:224–31.
Davies A, Sitte T, Elsner F, et al. Consistency of efficacy, patient acceptability, and nasal tolerability of fentanyl pectin nasal spray compared with immediate-release morphine sulfate in breakthrough cancer pain. J Pain Symptom Manag. 2011;4:358–66.
Zeppetella G, Davies A, Eijgelshoven I, et al. A network meta-analysis of the efficacy of opioid analgesics for the management of breakthrough cancer pain episodes. J Pain Symptom Manag. 2014;47:772–85.e5.
Magnani C, Giannarelli D, Casale G. Procedural pain in palliative care: is it breakthrough pain? A multicenter national prospective study to assess prevalence, intensity, and treatment of procedure-related pain in patients with advanced disease. Clin J Pain. 2017;33:707–14.
Mercadante S, Marchetti P, Cuomo A, et al. Factors influencing the clinical presentation of breakthrough pain in cancer patients. Cancers. 2018;10:175. https://doi.org/10.3390/cancers10060175.
Escobar Álvarez Y, Biete i Solà A, Camba Rodríguez M, et al. Diagnosis and treatment of breakthrough cancer pain: consensus recommendations. Rev Soc Esp Dolor. 2013;20:61–8 (article in Spanish).
Zeppetella G, Davies AN. Opioids for the management of breakthrough pain in cancer patients. In: Cochrane database of systematic reviews. Hoboken: Wiley-Blackwell; 2013.
Bossi P, Locati L, Bergamini C, et al. Fentanyl pectin vassal spray as treatment for incident predictable breakthrough pain in oral mucositis induced by chemoradiotherapy in head and neck cancer. Oral Oncol. 2014;50(9):884–7. https://doi.org/10.1016/j.oraloncology.2014.06.013(Epub 2014 Jul 4).
Caravatta L, Ramella S, Melano A, et al. Breakthrough pain management in patients undergoing radiotherapy: a national survey on behalf of the Palliative and Supportive Care Study Group. Tumori. 2015;101(6):603–8. https://doi.org/10.5301/tj.5000308(Epub 2015 May 16).
Prieto I, Pardo J, Luna J, et al. Facilitation of accurate and effective radiation therapy using fentanyl pectin nasal spray (FPNS) to reduce incidental breakthrough pain due to procedure positioning. Scand J Pain. 2016;11:52–8.
Mazzola R, Ricchetti F, Fiorentino A, et al. Fentanyl pectin nasal spray for painful mucositis in head and neck cancers during intensity-modulated radiation therapy with o without chemotherapy. Clin Transl Oncol. 2017;19(5):593–8. https://doi.org/10.1007/s12094-016-1570-5(Epub 2016 Nov 16).
Di Franco R, Falivene S, Ravo V, et al. Impact of procedural pain in radiotherapy treatment. World Cancer Res J. 2017;4:e884.
Pardo J, Mena A, Jiménez E, et al. Effectiveness of fentanyl pectin nasal citrate in controlling breakthrough pain episodes caused by routine radiotherapy procedures. Int J Radiat Oncol Biol Phys. 2017;99:E524–5.
Acknowledgements
Authors would thank to Pablo Vivanco (PhD, Meisys) for helping in the elaboration of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Because fentanyl pectin nasal citrate is an approved drug for the treatment of breakthrough pain, ethical approval is not necessary. However the study has been ethically approved.
Informed consent
All patients signed the informed consent to participate in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pardo, J., Mena, A., Jiménez, E. et al. Effectiveness of fentanyl pectin nasal citrate in controlling episodes of breakthrough cancer pain triggered by routine radiotherapy procedures. Clin Transl Oncol 21, 1568–1572 (2019). https://doi.org/10.1007/s12094-019-02125-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02125-8