Abstract
Purpose
Our main aim is to analyze the survival results in women operated on for advanced ovarian cancer with two different HIPEC regimens (cisplatin plus doxorubicin versus paclitaxel).
Patients and methods
A prospective cohort of patients with stage IIIC or IV epithelial ovarian cancer operated on with cytoreductive surgery and HIPEC, from October-2008 to February-2016, was retrospectively analyzed. The two drugs used, cisplatin/doxorubicin (Group A) and paclitaxel (Group B), were compared.
Results
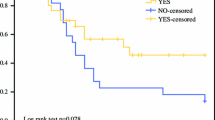
Forty-one patients were treated with cytoreductive surgery and HIPEC; 19 patients (46%) were in Group A and 22 (54%) were in Group B. The extent of peritoneal disease was comparable between groups (Peritoneal Cancer Index of 10 in Group A versus PCI of 12.5 in Group B). There were no differences in morbidity between groups, with a severe morbidity (Dindo–Clavien III or IV) of 36.8% versus 27.3%, respectively. There was no postoperative mortality. Median follow-up was 39 months. Median overall survival was 79 months. Overall survival at 3 years in Group A was 66% versus 82.9% in Group B (p = 0.248). Incomplete cytoreduction (macroscopic residual tumour after surgery) was identified as the only independent factor that influenced overall survival (HR 12.30, 95% CI 1.28–118.33, p = 0.03). The cytostatic used in HIPEC had no influence in overall survival.
Conclusion
The cytostatic used in HIPEC did not have a negative effect in the prognosis of patients with advanced ovarian cancer.


Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62(1):10–29.
Hennessy BT, Coleman RL, Markman M. Ovarian cancer. Lancet. 2009;374(9698):1371–82.
Cascales-Campos P, López-López V, Gil J, Arevalo-Perez J, Nieto A, Barceló F, et al. Hyperthermic intraperitoneal chemotherapy with paclitaxel or cisplatin in patients with stage III-C/IV ovarian cancer. Is there any difference? Surg Oncol. 2016;25(3):164–70.
Chan JK, Cheung MK, Husain A, Teng NN, West D, Whittemore AS, et al. Patterns and progress in ovarian cancer over 14 years. Obstet Gynecol. 2006;108:521–8.
Sugarbaker PH. It´s what the surgeon doesn´t see that kills the patient. J Nippon Med Sch. 2000;67(1):5–8.
Wright AA, Cronin A, Milne DE, Bookman MA, Burger RA, Cohn DE, et al. Use and effectiveness of intraperitoneal chemotherapy for treatment of ovarian cancer. J Clin Oncol. 2015;33(26):2841–7.
Sugarbaker PH. Cytoreductive surgery and perioperative intraperitoneal chemotherapy for the treatment of advanced primary and recurrent ovarian cancer. Curr Opin Obstet Gynecol. 2009;21(1):15–24.
Cascales PA, Gil J, Galindo PJ, Machado F, Frutos IM, Paricio PP. Heterogeneity in patients and methods. A problem for hyperthermic intraoperative intraperitoneal chemotherapy (HIPEC) in ovarian carcinoma. Eur J Obstet Gynecol Reprod Biol. 2011;158(2):361–2.
Portilla AG, Shigeki K, Dario B, Marcello D. The intraoperative staging systems in the management of peritoneal surface malignancy. J Surg Oncol. 2008;98(4):228–31.
Gonzalez-Moreno S, Kusamura S, Baratti D, Deraco M. Postoperative residual disease evaluation in the locoregional treatment of peritoneal surface malignancy. J Surg Oncol. 2008;98(4):237–41.
Elias DM, Ouellet JF. Intraperitoneal chemohyperthermia: rationale, technique, indications, and results. Surg Oncol Clin N Am. 2001;10(4):915–33.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13.
Markman M, Bundy BN, Alberts DS, Fowler JR, Clark-Pearson DL, Carson LF, et al. Phase III trial of standard-dose intravenous cisplatin plus paclitaxel versus moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small-volume stage III ovarian carcinoma: an intergroup study of the Gynecologic Oncology Group, Southwestern Oncology Group, and Eastern Cooperative Oncology Group. J Clin Oncol. 2001;19(4):1001–7.
Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med. 2006;354(1):34–433.
Takahashi I, Emi Y, Hasuda S, Kakeji Y, Maehara Y, Sugimachi K. Clinical application of hyperthermia combined with anticancer drugs for the treatment of solid tumors. Surgery. 2002;131:S78–84.
Muñoz-Casares FC, Rufian S, Rubio MJ, Díaz CJ, Díaz R, Casado A, et al. The role of hyperthermic intraoperative intraperitoneal chemotherapy (HIPEC) in the treatment of peritoneal carcinomatosis in recurrent ovarian cancer. Clin Transl Oncol. 2009;11(11):753–9.
Cascales-Campos PA, Gil J, Gil E, Feliciangeli E, Gonzalez-Gil A, Parrilla JJ, et al. Treatment of microscopic disease with hyperthermic intraoperative intraperitoneal chemotherapy after complete cytoreduction improves disease-free survival in patients with stage IIIC/IV ovarian cancer. Ann Surg Oncol. 2014;21(7):2383–9.
Bae JH, Lee JM, Ryu KS, Lee YS, Park YG, Hur SY, et al. Treatment of ovarian cancer with paclitaxel- or carboplatin-based intraperitoneal hyperthermic chemotherapy during secondary surgery. Gynecol Oncol. 2007;106(1):193–200.
Chang SJ, Hodeib M, Chang J, Bristow RE. Survival impact of complete cytoreduction to no gross residual disease for advanced-stage ovarian cancer: a meta-analysis. Gynecol Oncol. 2013;130(3):493–8.
Chiva LM, Gonzalez-Martin A. A critical appraisal of hyperthermic intraperitoneal chemotherapy (HIPEC) in the treatment of advanced and recurrent ovarian cancer. Gynecol Oncol. 2015;136(1):130–5.
Bakrin N, Bereder JM, Decullier E, Classe JM, Msika S, Lorimier G, et al. Peritoneal carcinomatosis treated with cytoreductive surgery and Hyperthermic Intraperitoneal Chemotherapy (HIPEC) for advanced ovarian carcinoma: a French multicentre retrospective cohort study of 566 patients. Eur J Surg Oncol. 2013;39(12):1435–43.
Gonzalez Bayon L, Steiner MA, Vasquez Jimenez W, Asencio JM, Alvarez de Sierra P, Atahualpa Arenas F, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for the treatment of advanced epithelial ovarian carcinoma: upfront therapy, at first recurrence, or later? Eur J Surg Oncol. 2013;39(10):1109–15.
Deraco M, Virzi S, Iusco DR, Puccio F, Macrí A, Famulari C, et al. Secondary cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for recurrent epithelial ovarian cancer: a multi-institutional study. BJOG. 2012;119(7):800–9.
Helm CW. The role of hyperthermic intraperitoneal chemotherapy (HIPEC) in ovarian cancer. Oncologist. 2009;14(7):683–94.
Manzanedo I, Pereira F, Perez-Viejo E, Serrano A, Hernández-García M, Martínez-Torres B, et al. Hyperthermic intraoperative intraperitoneal chemotherapy (HIPEC) with primary or secondary cytoreductive surgery in the treatment of advanced epithelial ovarian cancer. Minerva Ginecol. 2017;69(2):119–27.
Spiliotis J, Halkia E, Lianos E, Kalantzi N, Grivas A, Efstathiou E, et al. Cytoreductive surgery and HIPEC in recurrent epithelial ovarian cancer: a prospective randomized phase III study. Ann Surg Oncol. 2015;22(5):1570–75.
Van Driel WJ, Koole SN, Sikorska K, Schagen van Leeuwen JH, Schreuder HWR, Hermans RHM, et al. Hyperthermic intraperitoneal chemotherapy in ovarian cancer. N Engl J Med. 2018;378(3):230–40.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The current study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
All the patients included in the study have signed an informed consent for the intervention, in which they have indicated their implicit consent for the collection of data for future oncological analyzes.
Rights and permissions
About this article
Cite this article
Manzanedo, I., Pereira, F., Serrano, Á. et al. The use of cisplatin plus doxorubicin or paclitaxel in hyperthermic intraperitoneal chemotherapy (HIPEC) for stage IIIC or IV epithelial ovarian cancer: a comparative study. Clin Transl Oncol 21, 1357–1363 (2019). https://doi.org/10.1007/s12094-019-02065-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02065-3