Abstract
Purpose
Current evidence suggests the need to improve the management of breakthrough cancer pain (BTcP). For this reason, we aimed to assess the opinion of a panel of experts composed exclusively of physicians from pain units, who play a major role in BTcP diagnosis and treatment, regarding the key aspects of BTcP management.
Methods
An ad hoc questionnaire was developed to collect real-world data on the management of BTcP. The questionnaire had 5 parts: (a) organizational aspects of pain units (n = 12), (b) definition and diagnosis (n = 3), (c) screening (n = 3), (d) treatment (n = 8), and (e) follow-up (n = 7).
Results
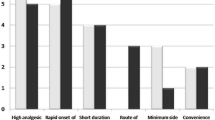
A total of 89 pain-unit physicians from 13 different Spanish regions were polled. Most of them agreed on the traditional definition of BTcP (78.9%) and the key features of BTcP (92.1%). However, only 30.3% of participants used the Davies’ algorithm for BTcP diagnosis. Respondents preferred to prescribe rapid-onset opioids [mean 77.0% (SD 26.7%)], and most recommended transmucosal fentanyl formulations as the first option for BTcP. There was also considerable agreement (77.5%) on the need for early follow-up (48–72 h) after treatment initiation. Finally, 65.2% of participants believed that more than 10% of their patients underused rapid-onset opioids.
Conclusions
There was broad agreement among pain experts on many important areas of BTcP management, except for the diagnostic method. Pain-unit physicians suggest that rapid-onset opioids may be underused by BTcP patients in Spain, an important issue that need to be evaluated in future studies.

Similar content being viewed by others
References
Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain. 1990;41(3):273–81.
Davies AN, Dickman A, Reid C, Stevens AM, Zeppetella G, Science Committee of the Association for Palliative Medicine of Great B, et al. The management of cancer-related breakthrough pain: recommendations of a task group of the Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. Eur J Pain. 2009;13(4):331–8. https://doi.org/10.1016/j.ejpain.2008.06.014.
Davies A, Buchanan A, Zeppetella G, Porta-Sales J, Likar R, Weismayr W, et al. Breakthrough cancer pain: an observational study of 1000 European oncology patients. J Pain Symptom Manag. 2013;46(5):619–28. https://doi.org/10.1016/j.jpainsymman.2012.12.009.
Breuer B, Fleishman SB, Cruciani RA, Portenoy RK. Medical oncologists' attitudes and practice in cancer pain management: a national survey. J Clin Oncol. 2011;29(36):4769–75. https://doi.org/10.1200/JCO.2011.35.0561.
van den Beuken-van Everdingen MH, de Rijke JM, Kessels AG, Schouten HC, van Kleef M, Patijn J. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol. 2007;18(9):1437–49. https://doi.org/10.1093/annonc/mdm056.
Deandrea S, Corli O, Consonni D, Villani W, Greco MT, Apolone G. Prevalence of breakthrough cancer pain: a systematic review and a pooled analysis of published literature. J Pain Symptom Manag. 2014;47(1):57–76. https://doi.org/10.1016/j.jpainsymman.2013.02.015.
American Pain F. Breakthrough cancer pain: mending the break in the continuum of care. J Pain Palliat Care Pharmacother. 2011;25(3):252–64. https://doi.org/10.3109/15360288.2011.599920.
Portenoy RK, Payne D, Jacobsen P. Breakthrough pain: characteristics and impact in patients with cancer pain. Pain. 1999;81(1–2):129–34.
Hagen NA, Stiles C, Nekolaichuk C, Biondo P, Carlson LE, Fisher K, et al. The Alberta Breakthrough Pain Assessment Tool for cancer patients: a validation study using a delphi process and patient think-aloud interviews. J Pain Symptom Manag. 2008;35(2):136–52. https://doi.org/10.1016/j.jpainsymman.2007.03.016.
Davies A, Zeppetella G, Andersen S, Damkier A, Vejlgaard T, Nauck F, et al. Multi-centre European study of breakthrough cancer pain: pain characteristics and patient perceptions of current and potential management strategies. Eur J Pain. 2011;15(7):756–63. https://doi.org/10.1016/j.ejpain.2010.12.004.
Zeppetella G, Ribeiro MD. Opioids for the management of breakthrough (episodic) pain in cancer patients. Cochrane Database Syst Rev. 2006;1:CD0043. https://doi.org/10.1002/14651858.CD004311.pub2.
Jara C, Del Barco S, Gravalos C, Hoyos S, Hernandez B, Munoz M, et al. SEOM clinical guideline for treatment of cancer pain (2017). Clin Transl Oncol. 2018;20(1):97–107. https://doi.org/10.1007/s12094-017-1791-2.
Caraceni A, Hanks G, Kaasa S, Bennett MI, Brunelli C, Cherny N, et al. Use of opioid analgesics in the treatment of cancer pain: evidence-based recommendations from the EAPC. Lancet Oncol. 2012;13(2):e58–68. https://doi.org/10.1016/S1470-2045(12)70040-2.
Ripamonti CI, Santini D, Maranzano E, Berti M, Roila F, Group EGW. Management of cancer pain: ESMO Clinical Practice Guidelines. Ann Oncol. 2012;23(Suppl 7):vii139–vii154. https://doi.org/10.1093/annonc/mds233.
Videla S, Catala E, Ribera MV, Montes A, Samper D, Fuentes J, et al. Characteristics and outcomes of chronic pain patients referred to hospital pain clinics: a prospective observational study. Minerva Anestesiol. 2017;83(1):12–22. https://doi.org/10.23736/S0375-9393.16.10999-X.
Mercadante S, Adile C, Torta R, Varetto A, Fulfaro F, Giarratano A, et al. Meaningful cut-off pain intensity for breakthrough pain changes in advanced cancer patients. Curr Med Res Opin. 2013;29(1):93–7. https://doi.org/10.1185/03007995.2012.755120.
EMA. European Medicines Agency (EMA). https://www.ema.europa.eu/ema/index.jsp?curl=pages/includes/medicines/medicines_landing_page.jsp&mid. Accessed 14 Feb 2018.
Elsner F, Zeppetella G, Porta-Sales J, Tagarro I. Newer generation fentanyl transmucosal products for breakthrough pain in opioid-tolerant cancer patients. Clin Drug Investig. 2011;31(9):605–18. https://doi.org/10.2165/11592910-000000000-00000.
Mercadante S, Marchetti P, Cuomo A, Mammucari M, Caraceni A, Group IMS. Breakthrough pain and its treatment: critical review and recommendations of IOPS (Italian Oncologic Pain Survey) expert group. Support Care Cancer. 2016;24(2):961–8. https://doi.org/10.1007/s00520-015-2951-y.
Wiffen PJ, Derry S, Moore RA. Tramadol with or without paracetamol (acetaminophen) for cancer pain. Cochrane Database Syst Rev. 2017;5:CD012508. https://doi.org/10.1002/14651858.CD012508.pub2.
Porta-Sales J, Perez C, Escobar Y, Martinez V. Diagnosis and management of breakthrough cancer pain: Have all the questions been resolved? A Delphi-based consensus assessment (DOIRON). Clin Transl Oncol. 2016;18(9):945–54. https://doi.org/10.1007/s12094-015-1468-7.
Boceta J, De la Torre A, Samper D, Farto M, Sanchez-de la Rosa R. Consensus and controversies in the definition, assessment, treatment and monitoring of BTcP: results of a Delphi study. Clin Transl Oncol. 2016;18(11):1088–97. https://doi.org/10.1007/s12094-016-1490-4.
Mercadante S, Lazzari M, Reale C, Cuomo A, Fusco F, Marchetti P, et al. Italian Oncological Pain Survey (IOPS): a multicentre Italian study of breakthrough pain performed in different settings. Clin J Pain. 2015;31(3):214–21. https://doi.org/10.1097/AJP.0000000000000161.
Webber K, Davies AN, Cowie MR. Accuracy of a diagnostic algorithm to diagnose breakthrough cancer pain as compared with clinical assessment. J Pain Symptom Manag. 2015;50(4):495–500. https://doi.org/10.1016/j.jpainsymman.2015.05.006.
Hanks GW, Conno F, Cherny N, Hanna M, Kalso E, McQuay HJ, et al. Morphine and alternative opioids in cancer pain: the EAPC recommendations. Br J Cancer. 2001;84(5):587–93. https://doi.org/10.1054/bjoc.2001.1680.
Jandhyala R, Fullarton JR, Bennett MI. Efficacy of rapid-onset oral fentanyl formulations vs. oral morphine for cancer-related breakthrough pain: a meta-analysis of comparative trials. J Pain Symptom Manag. 2013;46(4):573–80. https://doi.org/10.1016/j.jpainsymman.2012.09.009.
Kleeberg UR, Filbet M, Zeppetella G. Fentanyl buccal tablet for breakthrough cancer pain: why titrate? Pain Pract. 2011;11(2):185–90. https://doi.org/10.1111/j.1533-2500.2010.00414.x.
Kongsgaard UE, Eeg M, Greisen H. The use of Instanyl(R) in the treatment of breakthrough pain in cancer patients: a 3-month observational, prospective, cohort study. Support Care Cancer. 2014;22(6):1655–62. https://doi.org/10.1007/s00520-014-2128-0.
Berterame S, Erthal J, Thomas J, Fellner S, Vosse B, Clare P, et al. Use of and barriers to access to opioid analgesics: a worldwide, regional, and national study. Lancet. 2016;387(10028):1644–56. https://doi.org/10.1016/S0140-6736(16)00161-6.
Kaye AD, Jones MR, Kaye AM, Ripoll JG, Galan V, Beakley BD, et al. Prescription opioid abuse in chronic pain: an updated review of opioid abuse predictors and strategies to curb opioid abuse: part 1. Pain Phys. 2017;20(2S):S93–S109.
Kwon JH. Overcoming barriers in cancer pain management. J Clin Oncol. 2014;32(16):1727–33. https://doi.org/10.1200/JCO.2013.52.4827.
Acknowledgements
We would like to thank the following researchers for their contribution to this study: Hermann Ribera, María José Pampin Conde, Jorge Luis Sobrino Ramallo, Joan Coma, María de la Luz Cánovas Martínez, Daniel Samper Bernal, Sergi Suarez Diaz, Elvira Pelet Pascual, José Santamaría, Enrique Latorre Marqués, Jenaro Mañero Rey, Margarita López Rouco, Ignacio Velázquez, Jose Luis Cid Calzada, Francisco Javier Robaina Padrón, Inmaculada Herrador Montiel, Rafael de Alba Moreno, Vicente de Sanctis, Manuel Ruiz Castro, Josefina Castillo Rodríguez, Luis Fuentetaja Martin Portugués, Juanma Mercado, Mar Domínguez, Ignacio Javier Hernández Ferreras, Concepción Pérez Hernández, Cristina del Pozo Martin, Manuel Sánchez del Águila, Nacho Calvo, Ángel Martínez Navas, Francisco Heredia, Manuel López Rodríguez, Fernando Neira Reina, Rosa Zueras Batista, Agustín Gerri Cebollada, José Miguel Esparza, Ángeles Canós, Juan Manuel Vaca, Lourdes Marugan, Emilio Miguel Bronte Borraz, Juan Carlos Carrión Pareja, Antonio Pajuelo Gallego, Dolores Bedmar Cruz, Antonio Carrascosa Fernandez, Francisco Sánchez Montero, David Abejón González, Rafael Salazar, Juan Caballero Callejas, Jesus Maldonado Contreras, Fernando Remartínez, Juan Carlos de la Pinta, Antonio Alcántara Montero, Martín Arcas Molina, María López Gómez, Pedro José Moñino Ruiz, Estrella Uriarte Brizuela, Jose Antonio Sánchez Tirado, Rosa María Albores Albores, Pere Ortells Nebot, Javier de Andrés Ares, Mª Dolores Rodrigo Royo, Antonio Montero Matamala, Jordi Guitart, Victoria Ribera, Jose Gil Fuentes Bellido, Vicente Domingo, Inmaculada Muro Castillo, Aurora de la Iglesia, Francisco Leal Quiñones, Cristina Martin, José Javier Carceller Ruiz, Consuelo Nieto Iglesias, Carlos Tornero Tornero, Juan Pérez Cajaraville, Mercedes Mozas Calabaza, Alfonso Diz Villar, Marta del Valle Hoyos and Manuel Gutiérrez Ramírez. The authors thank Lucía Perez at MSC (Valencia, Spain) for editorial support in writing of this manuscript. All authors revised the manuscript versions and approved the final draft.
Funding
The Relevium project was sponsored by the Grupo de trabajo de dolor oncológico of the SED (Sociedad Española de Dolor). Takeda pharmaceutics sponsored the creation and maintenance of the study´s on-line database, but was not involved in data collection, analysis and conclusions of the study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The following authors declare potential conflicts of interest for individual activities for the industry: Villegas Estévez F. has received payments for consultancies and lecture fees from Kyowa kirin, Gebro pharma, Grünenthal, Ferrer and Mylan. López Alarcón M.D. has received payments for consultancies and lecture fees from Kyowa kirin, Takeda, Gebro pharma, Lab Esteve, Teva and Grünenthal. Mayoral V. has received payments for consultancies and lecture fees from Menarini, Pfizer, Grünenthal, Mundipharma, Takeda y Kyowa Kirin. Madariaga M. has received lecture fees from Kyowa kirin, Grünenthal, Mundipharma, Ferrer and Medtronic. Carregal A. has received payments for consultancies and lecture fees from Takeda, Gebro pharma, Lab Esteve, Teva, Grünenthal and Kyowa kirin. Trinidad Martín-Arroyo JM. has received payments for consultancies and lecture fees from Takeda, Gebro pharma, Lab Esteve, Teva, Grünenthal, and Kyowa kirin.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Estévez, F.V., Alarcón, M.D.L., Mayoral, V. et al. Current management of breakthrough cancer pain according to physicians from pain units in Spain. Clin Transl Oncol 21, 1168–1176 (2019). https://doi.org/10.1007/s12094-019-02044-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02044-8