Abstract
Objective
To explore the imaging features of adrenal primitive neuroectodermal tumors (PNETs).
Materials and methods
This retrospective study included seven patients with surgically and pathologically confirmed adrenal PNETs. Among them, six underwent computed tomography (CT) scans, and one underwent magnetic resonance imaging. The imaging findings, including size, shape, margin, hemorrhage, calcification, cystic degeneration, regional lymph nodes involvement, tumor thrombus formation and enhancement pattern, were retrospectively analyzed.
Results
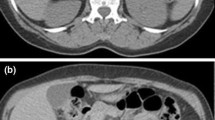
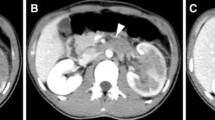
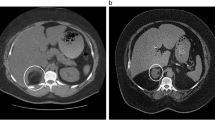
Among the seven adrenal PNET patients, six were male, and one was female. The median age was 26 years (range 2–56 years). The disease generally presented with either insidious symptoms (n = 4) or non-specific symptoms, including right flank pain (n = 1) and left upper abdominal discomfort (n = 2). On the pre-enhanced CT images, the tumor usually appeared as a well-defined, rounded or oval, heterogeneous mass without calcification. Certain tissue characteristics, such as cystic degeneration (n = 5), capsule (n = 4) and hemorrhage (n = 2), were observed. Regional lymph node involvement was observed in three cases, and renal vein thrombus was observed in one case. All cases showed mild heterogeneous enhancement of the tumor on the enhanced CT images.
Conclusion
An adrenal PNET commonly presents as a relatively large, well-defined, heterogeneous mass with cystic degeneration, necrosis and a characteristic mild contrast-enhancement pattern on multiphase enhanced images. PNET should be considered when the diagnosis of common tumors is not favored by signs on images.
Clinical Trial Registration Statement
This study was approved by the medical ethics committee of Xiangya Hospital, Central South University. The approval number is 201512538.






Similar content being viewed by others
References
Biegel JA, Rorke LB, Packer RJ, Sutton LN, Schut L, Bonner K, et al. Isochromosome 17q in primitive neuroectodermal tumors of the central nervous system. Genes Chromosomes Cancer. 1989;1:139–47.
Dehner LP. Peripheral and central primitive neuroectodermal tumors. A nosologic concept seeking a consensus. Arch Pathol Lab Med. 1986;110:997–1005.
Kushner BH, Hajdu SI, Gulati SC, Erlandson RA, Exelby PR, Lieberman PH. Extracranial primitive neuroectodermal tumors. The Memorial Sloan-Kettering Cancer Center experience. Cancer. 1991;67:1825–9.
Hari S, Jain TP, Thulkar S, Bakhshi S. Imaging features of peripheral primitive neuroectodermal tumours. Br J Radiol. 2008;81:975–83.
Jürgens H, Bier V, Harms D, Beck J, Brandeis W, Etspüler G, et al. Malignant peripheral neuroectodermal tumors. A retrospective analysis of 42 patients. Cancer. 1988;61:349–57.
Duan XH, Ban XH, Liu B, Zhong XM, Guo RM, Zhang F, et al. Intraspinal primitive neuroectodermal tumor: imaging findings in six cases. Eur J Radiol. 2011;80:426–31.
Mandal PK, Mukherjee S, Roy S, Bhattacharyya NK. PNET of kidney: report of four cases. Indian J Med Paediatr Oncol. 2012;33:130–3.
Karpate A, Menon S, Basak R, Yuvaraja TB, Tongaonkar HB, Desai SB. Ewing sarcoma/primitive neuroectodermal tumor of the kidney: clinicopathologic analysis of 34 cases. Ann Diagn Pathol. 2012;16:267–74.
Dutta D, Shivaprasad KS, Das RN, Ghosh S, Chowdhury S. Primitive neuroectodermal tumor of adrenal: clinical presentation and outcomes. J Cancer Res Ther. 2013;9:709–11.
Yoon JH, Kim H, Lee JW, Kang HJ, Park HJ, Park KD, et al. Ahn, Ewing sarcoma/peripheral primitive neuroectodermal tumor in the adrenal gland of an adolescent: a case report and review of the literature. J Pediatr Hematol Oncol. 2014;36:e456–9.
Sasaki T, Onishi T, Yabana T, Hoshina A. Ewing’s sarcoma/primitive neuroectodermal tumor arising from the adrenal gland: a case report and literature review. Tumori. 2013;99:e104–6.
Abi-Raad R, Manetti GJ, Colberg JW, Hornick JL, Shah JG, Prasad ML. Ewing sarcoma/primitive neuroectodermal tumor arising in the adrenal gland. Pathol Int. 2013;63:283–6.
Zhang L, Yao M, Hisaoka M, Sasano H, Gao H, Zhang Y, et al. Primary Ewing sarcoma/primitive neuroectodermal tumor in the adrenal gland. APMIS. 2016;. doi:10.1111/apm.12544.
Teixeira U, Goldoni M, Unterleider M, Diedrich J, Balbinot D, Rodrigues P, Fontes F Waechter, et al. Primitive neuroectodermal tumor of the pancreas: a case report and review of the literature. Case Rep Surg. 2015;2015:276869.
Dunnick NR. Hanson lecture. Adrenal imaging: current status. AJR Am J Roentgenol. 1990;154:927–36.
Taffel M, Haji-Momenian S, Nikolaidis P, Miller FH. Adrenal imaging: a comprehensive review. Radiol Clin N Am. 2012;50:219–43.
Qian X, Kai X, Shaodong L, Gaohong C, Hong M, Jingjing L. Radiological and clinicopathological features of pPNET. Eur J Radiol. 2013;82:e888–93.
Li X, Zhang W, Song T, Sun C, Shen Y. Primitive neuroectodermal tumor arising in the abdominopelvic region: CT features and pathology characteristics. Abdom Imaging. 2011;36:590–5.
Dick EA, Mchugh K, Kimber C, Michalski A. Imaging of non-central nervous system primitive neuroectodermal tumours: diagnostic features and correlation with outcome. Clin Radiol. 2001;56:206–15.
Gong J, Zhang Y, Zuo M, Yang Z, Zang D, Bao S, et al. Imaging findings of abdominal peripheral primitive neuroectodermal tumor: report of four cases with pathological correlation. Clin Imaging. 2009;33:196–9.
Ba L, Tan H, Xiao H, Guan Y, Gao J, Gao X. Radiologic and clinicopathologic findings of peripheral primitive neuroectodermal tumors. Acta Radiol. 2015;56:820–8.
Seo JM, Park BK, Park SY, Kim CK. Characterization of lipid-poor adrenal adenoma: chemical-shift MRI and washout CT. Am J Roentgenol. 2014;202:1043–50.
Zhang YM, Lei PF, Chen MN, Lv XF, Ling YH, Cai PQ, et al. CT findings of adrenal schwannoma. Clin Radiol. 2016;71:464–70.
Mannelli M, Colagrande S, Valeri A, Parenti G. Incidental and metastatic adrenal masses. Semin Oncol. 2010;37:649–61.
Acknowledgements
This study was supported by the Freedom Exploration Program of Central South University (No. 2011QNZT153) and Natural Science Foundation of Hunan Province (No. 14JJ6001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
This study was conducted in accordance with the Declaration of Helsinki, all its amendments, and national regulations. This retrospective study was approved by the hospital’s Institutional Review Board. Written or verbal informed consent was obtained from all patients.
Conflict of interest
None of the authors has declared any conflict of interest.
Additional information
Y. Zhang, P. Cai and M. Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, Y., Cai, P., Chen, M. et al. Imaging findings of adrenal primitive neuroectodermal tumors: a series of seven cases. Clin Transl Oncol 19, 641–649 (2017). https://doi.org/10.1007/s12094-016-1580-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-016-1580-3