Abstract
Purpose
We aimed to determine the efficacy and the toxicity of low dose weekly gemcitabine with radiation therapy in medically unfit muscle-invasive bladder cancer patients.
Methods
Twenty-six patients were included into the retrospective analysis. Weekly gemcitabine was administered 75 mg/m2 with a median dose of 63 Gy radiation therapy. Clinical target volume was defined as the urinary bladder only in conformal treatment planning.
Results
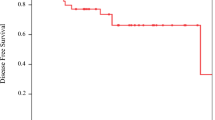
Median follow-up was 51 months (range 14–118 months). Complete response rate was 62.5 %. The 5-year local progression-free survival, disease-specific survival and overall survival rates were 40.6, 59.5 and 58.5 %, respectively. Concurrent chemotherapy was continued in 80.7 % of patients without any interruption. Gemcitabine was stopped due to grade 3 thrombocytopenia (n = 1), cardiac angina (n = 1), chronic obstructive pulmonary disease exacerbation (n = 1) or patients’ reluctance (n = 2).
Conclusions
Low dose weekly gemcitabine with concurrent radiotherapy is a tolerable regimen and have comparable outcomes with platinum-based combined treatments in muscle-invasive bladder cancer. Prospective randomized trials can help in understanding the safety and efficacy of this treatment specially in medically unfit patients.

Similar content being viewed by others
References
Stein JP, Lieskovsky G, Cote R, Groshen S, Feng AC, Boyd S, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1.054 patients. J Clin Oncol. 2001;19:666–75.
Kaufman DS, Winter KA, Shipley WU, Heney NM, Chetner MP, Souhami L, et al. The initial results in muscle-invading bladder cancer of RTOG 95–06: phase I/II trial of transurethral surgery plus radiation therapy by selective bladder preservation or cystectomy depending on the initial response. Oncologist. 2000;5:471–6.
Shipley WU, Winter KA, Kaufman DS, Lee WR, Heney NM, Tester WR, et al. Phase III trial of neoadjuvant chemotherapy in patients with invasive bladder cancer treated with selective bladder preservation by combined radiation therapy and chemotherapy: ınitial results of Radiation Therapy Oncology Group 89–03. J Clin Oncol. 1998;16:3576–83.
Tester W, Caplan R, Heaney J, Venner P, Whittington R, Byhardt R, et al. Neoadjuvant combined modality program with selective organ preservation for invasive bladder cancer: Results of Radiation Oncology Group phase II trial 88–02. J Clin Oncol. 1996;14:119–26.
Tester W, Porter A, Asbell S, Coughlin C, Haeney J, Krall J, et al. Combined modality program with possible organ preservation for invasive bladder carcinoma: results of RTOG protocol 85–12. Int J Radiat Oncol Biol Phys. 1993;25:783–90.
Shipley WU, Kaufman DS, Zehr E, Heney NM, Lane SC, Thakral HK, et al. Selective bladder preservation by combined modality protocol treatment. Long-term outcomes of 190 patients with invasive bladder cancer. Urology. 2002;60:62–8.
Shelley MD, Barber J, Wilt T, Mason MD. Surgery versus radiotherapy for muscle invasive bladder cancer. Cochrane Database Syst Rev. 2002;1:CD002079.
Quilty PM, Duncan W. Primary radical radiotherapy for T3 transitional cell cancer of the bladder: an analysis of survival and control. Int J Radiat Oncol Biol Phys. 1986;12:853–60.
Goffinet DR, Schneider MJ, Glatstein EJ, Ludwig H, Ray GR, Dunnick NR, et al. Bladder cancer: results of radiation therapy in 384 patients. Radiology. 1975;117:149–53.
Advanced Bladder Cancer Meta-analysis Collaboration. Neoadjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis. Lancet. 2003;361:1927–34.
Wilson DG, Bentzen SM, Harari PM. Biologic basis for combining drugs with radiation. Sem Radiat Oncol. 2006;16:2–9.
von der Maase H, Hansen SW, Roberts JT, Dogliotti L, Oliver T, Moore MJ, et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. 2000;17:3068–77.
Caffo O, Fellin G, Graffer U, Valduga F, Bolner A, Luciani L, et al. Phase I study of gemcitabine and radiotherapy plus cisplatin after transurethral resection as conservative treatment for infiltrating bladder cancer. Int J Radiat Oncol Biol Phys. 2003;57:1310–6.
Sangar VK, McBain CA, Lyons J, Ramani VA, Logue JP, Wylie JP, et al. Phase I study of conformal radiotherapy with concurrent gemcitabine in locally advanced bladder cancer. Int J Radiat Oncol Biol Phys. 2005;61:420–5.
Oh KS, Soto DE, Smith DC, Montie JE, Lee CT, Sandler HM. Combined modality therapy with gemcitabine and radiation therapy as a bladder preservation strategy: long term results of a phase I trial. Int J Radiat Oncol Biol Phys. 2009;74:511–7.
Borut K, Lijana ZK. Phase I study of radiochemotherapy with gemcitabine in invasive bladder cancer. Radiother Oncol. 2012;102:412–5.
Abacioglu U, Yumuk PF, Caglar H, Sengoz M, Turhal NS. Concurrent chemoradiotherapy with low dose weekly gemcitabine in stage III non small cell lung cancer. BMC Cancer. 2005;5:71.
Atasoy BM, Dane F, Ucuncu Kefeli A, Caglar H, Cingi A, Turhal NS, et al. Concomitant chemoradiotherapy with low dose weekly gemcitabine for nonmetastatic unresectable pancreatic cancer. Turk J Gastroenterol. 2011;22:60–4.
Cancer Therapy Evaluation Program (1999) Common Toxicity Criteria, Version 2.0.http://www.eortc.be/services/doc/ctc/ctcv20_4-30-992.pdf. Accessed 20 Jan 2011.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Atasoy, B.M., Dane, F., Alsan Cetin, I. et al. Concurrent chemoradiotherapy with low dose weekly gemcitabine in medically inoperable muscle-invasive bladder cancer patients. Clin Transl Oncol 16, 91–95 (2014). https://doi.org/10.1007/s12094-013-1047-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-013-1047-8