Abstract
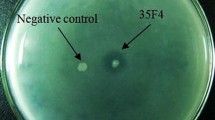
The human oral metagenomic DNA cloned into plasmid pUC19 was used to construct a DNA library in Escherichia coli. Functional screening of 40,000 metagenomic clones led to identification of a clone LIP2 that exhibited halo on tributyrin agar plate. Sequence analysis of LIP2 insert DNA revealed a 939 bp ORF (omlip1) which showed homology to lipase 1 of Acinetobacter junii SH205. The omlip1 ORF was cloned and expressed in E. coli BL21 (DE3) using pET expression system. The recombinant enzyme was purified to homogeneity and the biochemical properties were studied. The purified OMLip1 hydrolyzed p-nitrophenyl esters and triacylglycerol esters of medium and long chain fatty acids, indicating the enzyme is a true lipase. The purified protein exhibited a pH and temperature optima of 7 and 37 °C respectively. The lipase was found to be stable at pH range of 6–7 and at temperatures lower than 40 °C. Importantly, the enzyme activity was unaltered, by the presence or absence of many divalent cations. The metal ion insensitivity of OMLip1offers its potential use in industrial processes.







Similar content being viewed by others
References
Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE (2005) Defining the normal bacterial flora of the oral cavity. J Clin Microbiol 43:5721–5732
Wilson M (2005) Microbial Inhabitants of Humans; Their Ecology and Role in Health and Disease. Cambridge University Press, Cambridge
Sharma R, Chisti Y, Banerjee UC (2001) Production, purification, characterization, and applications of lipases. Biotechnol Adv 19:627–662
Suzuki T, Nakayama T, Kurihara T, Nishino T, Esaki N (2001) Cold-active lipolytic activity of psychrotrophic Acinetobacter sp. strain no. 6. J Biosci Bioeng 92:144–148
Rhee JK, Ahn DG, Kim YG, Oh JW (2005) New thermophilic and thermostable esterase with sequence similarity to the hormone-sensitive lipase family, cloned from a metagenomic library. Appl Environ Microbiol 71:817–825
Mhetras NC, Bastawde KB, Gokhale DV (2009) Purification and characterization of acidic lipase from Aspergillus niger NCIM 1207. Bioresour Technol 100:1486–1490
Holmquist M (2000) Alpha/Beta-hydrolase mechanisms fold enzymes: structures, functions and mechanisms. Curr Protein Pept Sci 1:209–235
Arpigny JL, Jaeger KE (1999) Bacterial lipolytic enzymes: classification and properties. Biochem J 343:177–183
Lee MH, Lee CH, Oh TK, Song JK, Yoon JH (2006) Isolation and characterization of a novel lipase from a metagenomic library of tidal flat sediments: evidence for a new family of bacterial lipases. Appl Environ Microbiol 72:7406–7409
Liu K, Wang J, Bu D, Zhao S, McSweeney C, Yu P, Li D (2009) Isolation and biochemical characterization of two lipases from a metagenomic library of China Holstein cow rumen. Biochem Biophys Res Commun 385:605–611
Bunterngsook B, Kanokratana P, Thongaram T, Tanapongpipat S, Uengwetwanit T, Rachdawong S, Vichitsoonthonkul T, Eurwilaichitr L (2010) Identification and characterization of lipolytic enzymes from a peat-swamp forest soil metagenome. Biosci Biotechnol Biochem 74:1848–1854
Glogauer A, Martini VP, Faoro H, Couto GH, Müller-Santos M, Monteiro RA, Mitchell DA, de Souza EM, Pedrosa FO, Krieger N (2011) Identification and characterization of a new true lipase isolated through metagenomic approach. Microbl Cell Fact 10:54–67
Belda-Ferre P, Alcaraz LD, Cabrera-Rubio R, Romero H, Simón-Soro A, Pignatelli M, Mira A (2012) The oral metagenome in health and disease. ISME J 6:46–56
Warburton P, Roberts AP, Allan E, Seville L, Lancaster H, Mullany P (2009) Characterization of tet(32) genes from the oral metagenome. Antimicrob Agents Chemother 53:273–276
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, New York
Lee SW, Won K, Lim HK, Kim JC, Choi GJ, Cho KY (2004) Screening for novel lipolytic enzymes from uncultured soil microorganisms. Appl Microbiol Biotechnol 65:720–726
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Sommer P, Bormann C, Götz F (1997) Genetic and biochemical characterization of a new extracellular lipase from Streptomyces cinnamomeus. Appl Environ Microbiol 63:3553–3560
Winkler UK, Stuckmann M (1979) Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia marcescens. J Bacteriol 138:663–670
Pinsirodom P, Parkin KL (2001) Lipase assays. In: Current protocols in food analytical chemistry, John Wiley and Sons, Inc., pp C3.1.1–C3.1.13
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Jeune CL, Navarro MAA, Iratxeta CP (2011) Prediction of protein secondary structure from circular dichroism using theoretically derived spectra. Proteins 80:374–381
Snellman EA, Sullivan ER, Colwell RR (2002) Purification and properties of the extracellular lipase, LipA, of Acinetobacter sp. RAG-1. Eur J Biochem 269:5771–5779
Salameh MA, Wiegel J (2010) Effects of detergents on activity, thermostability and aggregation of two alkalithermophilic lipases from Thermosyntropha lipolytica. Open Biochem J 4:22–28
Pollock MR, Richmond MH (1962) Low cyst(e)ine content of bacterial extracellular proteins: its possible physiological significance. Nature 194:446–449
Benjamin S, Pandey A (1998) Candida rugosa lipases: molecular biology and versatility in biotechnology. Yeast 14:1069–1087
Acknowledgments
Authors thank Department of Science and Technology, India for providing Innovation in Science Pursuit for Inspired Research (INSPIRE) fellowship to AP. The Centre for Advanced studies in Functional Genomics, Centre for Excellence in Genomic Sciences, and the Networking Resource Centre in Biological Sciences at Madurai Kamaraj University are highly acknowledged for the support facilities. The School of Chemistry, MKU is gratefully acknowledged for providing the circular dichroism facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Preeti, A., Hemalatha, D., Rajendhran, J. et al. Cloning, Expression and Characterization of a Lipase Encoding Gene from Human Oral Metagenome. Indian J Microbiol 54, 284–292 (2014). https://doi.org/10.1007/s12088-014-0455-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-014-0455-y