Abstract
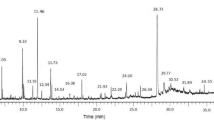
Acacia catechu, commonly known as catechu, cachou and black cutch is an important medicinal plant and an economically important forest tree. The methanolic extract of this plant was found to have antimicrobial activities against six species of pathogenic and non-pathogenic microorganisms: Bacillus subtilis, Staphylococcus aureus, Salmonella typhi, Escherichia coli, Pseudomonas aeruginosa and Candida albicans. The maximum zone of inhibition (20 mm) was found to be exhibited against S. aureus. For this organism the minimum bactericidal concentration (MBC) of the crude extract was 1,000 μg/ml. The extract was found to be equally effective against gram positive and gram negative bacteria. The antimicrobial activity of the extract was found to be decreased during purification. The chemical constituents of organic plant extracts were separated by thin layer chromatography (TLC) and the plant extracts were purified by column chromatography and were further identified by Gas chromatography–mass selection (GC–MS) analysis. The composition of A. catechu extract had shown major components of terpene i.e. camphor (76.40%) and phytol (27.56%) along with other terpenes in minor amounts which are related with their high antibacterial and antifungal properties.


Similar content being viewed by others
References
Parekh J, Karathia N, Chanda S (2006) Screening of some traditionally used medicinal plants for potential antibacterial activity. Indian J Pharm Sci 68(6):832–834
Chambers HF, Deleo FR (2009) Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat Rev Microbiol 7:629–641
Tenover F (2006) Mechanisms of antimicrobial resistance in bacteria. Am J Med 9:3–10
Zhou L, Lin Q, Li B, Li N, Zhang S (2009) Expression and purification of the antimicrobial peptide CM4 in Escherichia coli. Biotechnol Lett 31:437–441
Livermore DM (2002) Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: our worst nightmare. Clin Infect Dis 34:634–640
Arunkumar S, Muthuselvam M (2009) Analysis of phytochemical constituents and antimicrobial activities of Aloe vera L. against clinical pathogens. World J Agric Sci 5(5):572–576
Samie A, Obi CL, Bessong PO, Namrita L (2005) Activity profiles of fourteen selected medicinal plants from Rural Venda communities in South Africa against fifteen clinical bacterial species. Afr J Biotechnol 4(12):1443–1451
Shen D, Wu Q, Wang M, Yang Y, Lavoie EJ, Simon JE (2006) Determination of the predominant catechins in Acacia catechu by liquid chromatography/electrospray ionization-mass spectrometry. J Agric Food Chem 54(9):3219–3224
Berezkin VG (2007) A new approach to the determination of relative retention in thin-layer liquid chromatography. J Anal Chem 62(4):366–368
Vardar-Unlu G, Silici S, Unlu M (2008) Composition and in vitro antimicrobial activity of Populus buds and poplar-type propolis. World J Microbiol Biotechnol 24:1011–1017
Andrews JM (2001) Determination of minimum inhibitory concentrations. J Antimicrob Chemother 48(suppl 1):5–16
Nostro A, Germano MP, D’Angelo V, Marino A, Cannatelli MA (2000) Extraction methods and bioautography for evaluation of medicinal plant antimicrobial activity. Lett Appl Microbiol 30(1):379–384
Clinical and Laboratory Standards Institute (CLSI) (2005) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically—sixth edition: approved standard M7-A6. CLSI, Wayne
Clinical and Laboratory Standards Institute (CLSI) (2008) Reference method for broth dilution antifungal susceptibility testing of yeasts: approved standard, 3rd edn. M27-A3. CLSI, Wayne
Gajera HP, Patel SV, Golakiya BA (2005) Antioxidant properties of some therapeutically active medicinal plants—an overview. JMAPS 27:91–100
Al-Bayati FA, AL-Mola HF (2008) Antibacterial and antifungal activities of different parts of Tribulus terrestris L. growing in Iraq. J Zhejiang Univ Sci B 9(2):154–159
Eloff JN (1998) Which extractant should be used for the screening and isolation of antimicrobial compounds from plants? J Ethanopharmacol 60:1–8
Morrissey JP, Osbourn AE (1999) Fungal resistance to plant antibiotics as a mechanism of pathogenesis. Microbiol Mol Biol Rev 63:708–724
Acknowledgments
We are grateful to Prof. YK Agarwal for the corrections and valuable comments on the manuscript. We are also thankful to Central Salt and Marine Research Institute (CSMRI), Bhavnagar for carrying out GC–MS analysis.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Negi, B.S., Dave, B.P. In Vitro Antimicrobial Activity of Acacia catechu and Its Phytochemical Analysis. Indian J Microbiol 50, 369–374 (2010). https://doi.org/10.1007/s12088-011-0061-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-011-0061-1