Abstract
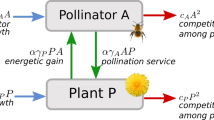
The present study theoretically examines the process by which interspecific mutualism is established with trait matching. The mathematical model includes joint evolution of the mutualistic relationship between two species and regulation of variation of interaction in one-dimensional trait space, assuming abiotic directional selection. The model considers three types of regulation: homeostasis against environmental variation, developmental stability, and acceptability of dissimilar mutualism partners (mutualism kernel). Mainly focusing on the developmental stability, the analysis indicates that the mutualism can evolve when (1) higher levels of developmental stability are more intensively degenerated by deleterious mutations, (2) the basal rate of deleterious mutation is low, (3) trait expression is less influenced by environmental factors, and (4) the specificity of mutualism is high. It also shows that the evolution of developmental stability can promote the evolution of mutualism with trait matching when the deleterious mutation bias disappears at a certain level of developmental instability. Evolution of homeostasis and mutualism kernel can be discussed in the similar way because of formal similarities in the model. In plant–pollinator interactions, it has recently been proposed that evolutionary increments of developmental stability in mutualistic traits might promote plant diversification. The present results partly support this hypothesis with respect to the evolutionary relationship between mutualism and developmental stability.







Similar content being viewed by others
References
Abrams PA (2000) The evolution of predator-prey interactions: theory and evidence. Annu Rev Ecol Syst 31:79–105
Ackermann M, Doebeli M (2004) Evolution of niche width and adaptive diversification. Evolution 58:2599–2612
Altshuler DL, Clark CJ (2003) Darwin’s hummingbirds. Science 300:588–589
Armbruster WS (2014) Floral specialization and angiosperm diversity: phenotypic divergence, fitness trade-offs and realized pollination accuracy. Aob Plants 6
Armbruster WS, Hansen TF, Pelabon C, Perez-Barrales R, Maad J (2009a) The adaptive accuracy of flowers: measurement and microevolutionary patterns. Ann Bot 103:1529–1545
Armbruster WS, Pelabon C, Hansen TF, Bolstad GH (2009b) Macroevolutionary patterns of pollination accuracy: a comparison of three genera. New Phytol 183:600–617
Balogh ACV, Leimar O (2005) Mullerian mimicry: an examination of Fisher’s theory of gradual evolutionary change. Proc Roy Soc B Biol Sci 272:2269–2275
Bascompte J, Jordano P, Melian CJ, Olesen JM (2003) The nested assembly of plant-animal mutualistic networks. Proc Natl Acad Sci USA 100:9383–9387
Cohen AA, Martin LB, Wingfield JC, McWilliams SR, Dunne JA (2012) Physiological regulatory networks: ecological roles and evolutionary constraints. Trends Ecol Evol 27:428–435
Dawkins R, Krebs JR (1979) Arms races between and within species. Proc R Soc Ser B Biol 205:489–511
Debat V, David P (2001) Mapping phenotypes: canalization, plasticity and developmental stability. Trends Ecol Evol 16:555–561
Dieckmann U, Doebeli M (1999) On the origin of species by sympatric speciation. Nature 400:354–357
Doebeli M (1996) An explicit genetic model for ecological character displacement. Ecology 77:510–520
Gangestad SW, Thornhill R (1999) Individual differences in developmental precision and fluctuating asymmetry: a model and its implications. J Evol Biol 12:402–416
Gomulkiewicz R, Nuismer SL, Thompson JN (2003) Coevolution in variable mutualisms. Am Nat 162:S80–S93
Hansen TF, Carter AJR, Pelabon C (2006) On adaptive accuracy and precision in natural populations. Am Nat 168:168–181
Iseki N, Sasaki A, Toju H (2011) Arms race between weevil rostrum length and camellia pericarp thickness: geographical cline and theory. J Theor Biol 285:1–9
Iwasa Y, Pomiankowski A, Nee S (1991) The evolution of costly mate preferences. II. The handicap principle. Evolution 45:1431–1442
Johnson SD, Steiner KE (2000) Generalization versus specialization in plant pollination systems. Trends Ecol Evol 15:140–143
Joron M, Mallet JLB (1998) Diversity in mimicry: paradox or paradigm? Trends Ecol Evol 13:461–466
Larson DL (1996) Seed dispersal by specialist versus generalist foragers: the plant’s perspective. Oikos 76:113–120
MacArthur R (1969) Species packing, and what interspecies competition minimizes. Proc Natl Acad Sci USA 64:1369–1371
MacArthur R, Levins R (1967) Limiting similarity convergence and divergence of coexisting species. Am Nat 101:377–385
Malcolm SB (1990) Mimicry—status of a classical evolutionary paradigm. Trends Ecol Evol 5:57–62
McPeek MA (2017) The ecological dynamics of natural selection: traits and the coevolution of community structure. Am Nat 189:E91–E117
Melzer R, Theißen G (2016) The significance of developmental robustness for species diversity. Ann Bot. doi:10.1093/aob/mcw1018
Nuismer SL, Gomulkiewicz R, Ridenhour BJ (2010) When is correlation coevolution? Am Nat 175:525–537
Pomiankowski A, Iwasa Y, Nee S (1991) The evolution of costly mate preferences. 1. Fisher and biased mutation. Evolution 45:1422–1430
Roughgarden J (1972) Evolution of niche width. Am Nat 106:683
Scheiner SM, Caplan RL, Lyman RF (1991) The genetics of phenotypic plasticity. III. Genetic correlations and fluctuating asymmetries. J Evol Biol 4:51–68
Slatkin M (1980) Ecological character displacement. Ecology 61:163–177
Taper ML, Case TJ (1985) Quantitative genetic models for the coevolution of character displacement. Ecology 66:355–371
Temeles EJ, Kress WJ (2003) Adaptation in a plant-hummingbird association. Science 300:630–633
Thoday JM (1958) Homeostasis in a selection experiment. Heredity 12:401–415
Thostesen AM, Olesen JM (1996) Pollen removal and deposition by specialist and generalist bumblebees in Aconitum septentrionale. Oikos 77:77–84
Van Dongen S, Lens L (2000) The evolutionary potential of developmental instability. J Evol Biol 13:326–335
Woods HA, Wilson JK (2013) An information hypothesis for the evolution of homeostasis. Trends Ecol Evol 28:283–289
Zhang XS, Hill WG (2005) Evolution of the environmental component of the phenotypic variance: stabilizing selection in changing environments and the cost of homogeneity. Evolution 59:1237–1244
Zhang F, Hui C, Pauw A (2013) Adaptive divergence in Darwin’s race: how coevolution can generate trait diversity in a pollination system. Evolution 67:548–560
Acknowledgements
I thank the members of the Centre for Ecological Research for their comments and encouragement. This research was supported financially by the JSPS KAKENHI Grant Numbers 15K07219. This research was partly supported by the International Research Unit of Advanced Future Studies at Kyoto University. I also appreciate helpful suggestions and comments of Dr. A. Rossberg and an anonymous reviewer.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 6925 kb)
Rights and permissions
About this article
Cite this article
Yamauchi, A. Joint evolution of interspecific mutualism and regulation of variation of interaction under directional selection in trait space. Theor Ecol 10, 477–491 (2017). https://doi.org/10.1007/s12080-017-0343-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-017-0343-2