Abstract
CREB-regulated transcription coactivator2 (CRTC2 or TORC2) is a transcriptional coactivator of CREB(cAMP response element binding protein), which affects human energy metabolism through cyclic adenosine phosphate pathway, Mammalian target of rapamycin (mTOR) pathway, Sterol regulatory element binding protein 1(SREBP1), Sterol regulatory element binding protein 2 (SREBP2) and other substances Current studies on CRTC2 mainly focus on glucose and lipid metabolism, relevant studies show that CRTC2 can participate in the occurrence and development of related diseases by affecting metabolic homeostasis. It has been found that Crtc2 acts as a signaling regulator for cAMP and Ca2 + signaling pathways in many cell types, and phosphorylation at ser171 and ser275 can regulate downstream biological functions by controlling CRTC2 shuttling between cytoplasm and nucleus.
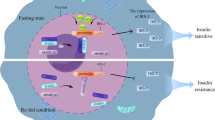
Graphical abstract


Source: Figure to: https://www.uniprot.org/

Source: Figure to: https://www.uniprot.org/

Source: Figure to: https://www.uniprot.org/

Similar content being viewed by others
References
Adeghate E, Singh J (2014) Structural changes in the myocardium during diabetes-induced cardiomyopathy. Heart Fail Rev 19(1):15–23
Altarejos JY, Montminy M (2011) CREB and the CRTC co-activators: sensors for hormonal and metabolic signals. Nat Rev Mol Cell Biol 12(3):141–151
Austin MA, Breslow JL, Hennekens CH, Buring JE, Willett WC, Krauss RM (1988) Low-density lipoprotein subclass patterns and risk of myocardial infarction. JAMA 260(13):1917–1921
Bruno NE, Nwachukwu JC, Hughes DC, Srinivasan S, Hawkins R, Sturgill D, Hager GL, Hurst S, Sheu S-S, Bodine SC, Conkright MD, Nettles KW (2021) Activation of Crtc2/Creb1 in skeletal muscle enhances weight loss during intermittent fasting. Faseb J. https://doi.org/10.1096/fj.202100171R
Chen Y, Zhang Z, Zhu D, Zhao W, Li F (2019) Long non-coding RNA MEG3 serves as a ceRNA for microRNA-145 to induce apoptosis of AC16 cardiomyocytes under high glucose condition. Biosci Rep. https://doi.org/10.1042/BSR20190444
Chen Y, Wang J, Wang Y, Wang P, Zhou Z, Wu R, Xu Q, You H, Liu Y, Wang L, Zhou L, Wu Y, Hu L, Liu H, Liu Y (2022) A propolis-derived small molecule ameliorates metabolic syndrome in obese mice by targeting the CREB/CRTC2 transcriptional complex. Nat Commun. https://doi.org/10.1038/s41467-021-27533-9
Ch’ng TH, Uzgil B, Lin P, Avliyakulov NK, O’Dell TJ, Martin KC (2012) Activity-dependent transport of the transcriptional coactivator CRTC1 from synapse to nucleus. Cell 150(1):207–221
Conkright MD, Canettieri G, Screaton R, Guzman E, Miraglia L, Hogenesch JB, Montminy M (2003) TORCs: transducers of regulated CREB activity. Mol Cell 12(2):413–423
Dentin R, Liu Y, Koo S-H, Hedrick S, Vargas T, Heredia J, Yates J III, Montminy M (2007) Insulin modulates gluconeogenesis by inhibition of the coactivator TORC2. Nature 449(7160):366
Dentin R, Hedrick S, Xie J, Yates J 3rd, Montminy M (2008a) Hepatic glucose sensing via the CREB coactivator CRTC2. Science 319(5868):1402–1405
Eberhard CE, Fu A, Reeks C, Screaton RA (2013) CRTC2 is required for beta-cell function and proliferation. Endocrinology 154(7):2308–2317
Forbes JM, Cooper ME (2013) Mechanisms of diabetic complications. Physiol Rev 93(1):137–188
Geer JC, McGill HC Jr, Strong JP (1961) The fine structure of human atherosclerotic lesions. Am J Pathol 38:263–287
Gimbrone MA Jr (1987) Vascular endothelium: nature’s blood-compatible container. Ann N Y Acad Sci 516:5–11
Gimbrone MA Jr, Garcia-Cardena G (2013) Vascular endothelium, hemodynamics, and the pathobiology of atherosclerosis. Cardiovasc Pathol 22(1):9–15
Gonzalez GA, Montminy MR (1989) Cyclic AMP stimulates somatostatin gene transcription by phosphorylation of CREB at serine 133. Cell 59(4):675–680
Han J, Li E, Chen L, Zhang Y, Wei F, Liu J, Deng H, Wang Y (2015) The CREB coactivator CRTC2 controls hepatic lipid metabolism by regulating SREBP1. Nature 524(7564):243
Han H-S, Choi BH, Kim JS, Kang G, Koo S-H (2017) Hepatic Crtc2 controls whole body energy metabolism via a miR-34a-Fgf21 axis. Nat Commun. https://doi.org/10.1038/s41467-017-01878-6
Han H-S, Kwon Y, Koo S-H (2020) Role of CRTC2 in metabolic homeostasis: key regulator of whole-body energy metabolism? Diabetes Metab J 44(4):498–508
He L (2021) Alterations of gut microbiota by overnutrition impact gluconeogenic gene expression and insulin signaling. Int J Mol Sci 22(4):2121
Herzig S, Shaw RJ (2018) AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol 19(2):121–135
Herzig S, Long F, Jhala US, Hedrick S, Quinn R, Bauer A, Rudolph D, Schutz G, Yoon C, Puigserver P, Spiegelman B, Montminy M (2001) CREB regulates hepatic gluconeogenesis through the coactivator PGC-1. Nature 413(6852):179–183
Horton JD, Goldstein JL, Brown MS (2002) SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver. J Clin Investig 109(9):1125–1131
Hudson C, Kimura TE, Duggirala A, Sala-Newby GB, Newby AC, Bond M (2018) Dual role of CREB in the regulation of VSMC proliferation: mode of activation determines Pro- or anti-mitogenic function. Sci Rep. https://doi.org/10.1038/s41598-018-23199-4
Iourgenko V, Zhang W, Mickanin C, Daly I, Jiang C, Hexham JM, Orth AP, Miraglia L, Meltzer J, Garza D, Chirn G-W, McWhinnie E, Cohen D, Skelton J, Terry R, Yu Y, Bodian D, Buxton FP, Zhu J, Song C, Labow MA (2003) Identification of a family of cAMP response element-binding protein coactivators by genome-scale functional analysis in mammalian cells. Proc Natl Acad Sci USA 100(21):12147–12152
Ji Y-X, Wang Y, Li P-L, Cai L, Wang X-M, Bai L, Liu Z, Tian H, Tian S, Zhang P, Zhang X-J, Cheng X, Yuan Y, She Z-G, Hu Y, Li H (2021) A kinome screen reveals that Nemo-like kinase is a key suppressor of hepatic gluconeogenesis. Cell Metab 33(6):1171
Jia G, DeMarco VG, Sowers JR (2016) Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol 12(3):144–153
Jia G, Whaley-Connell A, Sowers JR (2018) Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 61(1):21–28
Kanki H, Sasaki T, Matsumura S, Kawano T, Todo K, Okazaki S, Nishiyama K, Takemori H, Mochizuki H (2020a) CREB coactivator CRTC2 Plays a crucial role in endothelial function. J Neurosci 40(49):9533–9546
Kloeting N, Blueher M (2014) Adipocyte dysfunction, inflammation and metabolic syndrome. Rev Endocr Metab Disord 15(4):277–287
Koo S-H, Flechner L, Qi L, Zhang X, Screaton RA, Jeffries S, Hedrick S, Xu W, Boussouar F, Brindle P, Takemori H, Montminy M (2005) The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature 437(7062):1109–1111
Kumar A, Rahnert JA, Zheng B, Hudson MB, Woodworth-Hobbs ME, Price SR (2016) Glucocorticoids Alter CRTC-CREB signaling in muscle cells: impact on PGC-1α expression and atrophy markers. PLoS ONE 11(7):e0159181
Lee J-M, Seo W-Y, Song K-H, Chanda D, Kim YD, Kim D-K, Lee M-W, Ryu D, Kim Y-H, Noh J-R, Lee C-H, Chiang JYL, Koo S-H, Choi H-S (2010) AMPK-dependent repression of hepatic gluconeogenesis via disruption of CREB center dot CRTC2 complex by orphan nuclear receptor small heterodimer partner. J Biol Chem 285(42):32182–32191
Lee J-H, Wen X, Cho H, Koo S-H (2018) CREB/CRTC2 controls GLP-1-dependent regulation of glucose homeostasis. FASEB J 32(3):1566–1578
Li Y, Song Y, Zhao M, Guo Y, Yu C, Chen W, Shao S, Xu C, Zhou X, Zhao L, Zhang Z, Bo T, Xia Y, Proud CG, Wang X, Wang L, Zhao J, Gao L (2017a) A novel role for CRTC2 in hepatic cholesterol synthesis through SREBP-2. Hepatology 66(2):481–497
Li Y, Wang L, Zhou L, Song Y, Ma S, Yu C, Zhao J, Xu C, Gao L (2017b) Thyroid stimulating hormone increases hepatic gluconeogenesis via CRTC2. Mol Cell Endocrinol 446:70–80
Lim H-Y, Wang W, Wessells RJ, Ocorr K, Bodmer R (2011) Phospholipid homeostasis regulates lipid metabolism and cardiac function through SREBP signaling in Drosophila. Genes Dev 25(2):189–200
Liu Y, Dentin R, Chen D, Hedrick S, Ravnskjaer K, Schenk S, Milne J, Meyers DJ, Cole P, Yates J III, Olefsky J, Guarente L, Montminy M (2008) A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange. Nature 456(7219):269-U274
Lu Y, Ma J, Li P, Liu B, Wen X, Yang J (2022) Ilexgenin A restrains CRTC2 in the cytoplasm to prevent SREBP1 maturation via AMP kinase activation in the liver. Br J Pharmacol 179(5):958–978
Luo Q, Viste K, Urday-Zaa JC, Kumar GS, Tsai W-W, Talai A, Mayo KE, Montminy M, Radhakrishnan I (2012) Mechanism of CREB recognition and coactivation by the CREB-regulated transcriptional coactivator CRTC2. Proc Natl Acad Sci USA 109(51):20865–20870
Luo J, Yang H, Song B-L (2020) Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol 21(4):225–245
Madison BB (2016) Srebp2: a master regulator of sterol and fatty acid synthesis. J Lipid Res 57(3):333–335
Mair W, Morantte I, Rodrigues AP, Manning G, Montminy M, Shaw RJ, Dillin A (2011) Lifespan extension induced by AMPK and calcineurin is mediated by CRTC-1 and CREB. Nature 470(7334):404–408
Marti CN, Gheorghiade M, Kalogeropoulos AP, Georgiopoulou VV, Quyyumi AA, Butler J (2012) Endothelial dysfunction, arterial stiffness, and heart failure. J Am Coll Cardiol 60(16):1455–1469
Medina DL, Fraldi A, Bouche V, Annunziata F, Mansueto G, Spampanato C, Puri C, Pignata A, Martina JA, Sardiello M, Palmieri M, Polishchuk R, Puertollano R, Ballabio A (2011) Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev Cell 21(3):421–430
Meisner HM, Lamers WH, van Lelyveld PH, Hanson RW (1982) Cyclic AMP and the regulation of gene expression of rat cytosolic phosphoenolpyruvate carboxykinase (GTP). Progr Clin Biol Res 102:241–250
Miano JM, Fisher EA, Majesky MW (2021) Fate and state of vascular smooth muscle cells in atherosclerosis. Circulation 143(21):2110–2116
Montminy M, Koo SH, Zhang X (2004) The CREB family: key regulators of hepatic metabolism. Ann Endocrinol 65(1):73–75
Montori-Grau M, Aguilar-Recarte D, Zarei M, Pizarro-Delgado J, Palomer X, Vazquez-Carrera M (2022) Endoplasmic reticulum stress downregulates PGC-1alpha in skeletal muscle through ATF4 and an mTOR-mediated reduction of CRTC2. Cell Commun Signal 20(1):53
Mueller TD, Finan B, Bloom SR, D’Alessio D, Drucker DJ, Flatt PR, Fritsche A, Gribble F, Grill HJ, Habener JF, Holst JJ, Langhans W, Meier JJ, Nauck MA, Perez-Tilve D, Pocai A, Reimann F, Sandoval DA, Schwartz TW, Seeley RJ, Stemmer K, Tang-Christensen M, Woods SC, DiMarchi RD, Tschoep MH (2019) Glucagon-like peptide 1 (GLP-1). Mol Metab 30:72–130
Nakatsu Y, Sakoda H, Kushiyama A, Ono H, Fujishiro M, Horike N, Yoneda M, Ohno H, Tsuchiya Y, Kamata H, Tahara H, Isobe T, Nishimura F, Katagiri H, Oka Y, Fukushima T, Takahashi S-I, Kurihara H, Uchida T, Asanoa T (2010) Pin1 associates with and induces translocation of CRTC2 to the cytosol, thereby suppressing cAMP-responsive element transcriptional activity. J Biol Chem 285(43):33018–33027
Nezich CL, Wang C, Fogel AI, Youle RJ (2015) MiT/TFE transcription factors are activated during mitophagy downstream of Parkin and Atg5. J Cell Biol 210(3):435–450
Peterson TR, Sengupta SS, Harris TE, Carmack AE, Kang SA, Balderas E, Guertin DA, Madden KL, Carpenter AE, Finck BN, Sabatini DM (2011) mTOR complex 1 regulates lipin 1 localization to control the SREBP pathway. Cell 146(3):408–420
Pilkis SJ, Claus TH (1991) Hepatic gluconeogenesis/glycolysis: regulation and structure/function relationships of substrate cycle enzymes. Annu Rev Nutr 11(1):465–515
Pilkis SJ, Claus TH, El-Maghrabi MR (1988) The role of cyclic AMP in rapid and long-term regulation of gluconeogenesis and glycolysis. Adv Second Messenger Phosphoprotein Res 22:175–191
Puertollano R, Ferguson SM, Brugarolas J, Ballabio A (2018) The complex relationship between TFEB transcription factor phosphorylation and subcellular localization. The EMBO J. https://doi.org/10.15252/embj.201798804
Qiao A, Zhou J, Xu S, Ma W, Boriboun C, Kim T, Yan B, Deng J, Yang L, Zhang E, Song Y, Ma YC, Richard S, Zhang C, Qiu H, Habegger KM, Zhang J, Qin G (2021) Sam68 promotes hepatic gluconeogenesis via CRTC2. Nat Commun. https://doi.org/10.1038/s41467-021-23624-9
Rahnert JA, Zheng B, Hudson MB, Woodworth-Hobbs ME, Price SR (2016) Glucocorticoids Alter CRTC-CREB signaling in muscle cells: impact on PGC-1alpha expression and atrophy markers. PLoS ONE 11(7):e0159181
Rines AK, Sharabi K, Tavares CDJ, Puigserver P (2016) Targeting hepatic glucose metabolism in the treatment of type 2 diabetes. Nat Rev Drug Discov 15(11):786–804
Rossetti C, Cherix A, Guiraud LF, Cardinaux J-R (2022) New insights into the pivotal role of CREB-regulated transcription coactivator 1 in depression and comorbid obesity. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2022.810641
Rui L (2014) Energy Metabolism in the Liver. Compr Physiol 4(1):177–197
Sakamoto K, Norona FE, Alzate-Correa D, Scarberry D, Hoyt KR, Obrietan K (2013) Clock and Light Regulation of the CREB Coactivator CRTC1 in the Suprachiasmatic Circadian Clock. J Neurosci 33(21):9021–9027
Screaton RA, Conkright MD, Katoh Y, Best JL, Canettieri G, Jeffries S, Guzman E, Niessen S, Yates JR 3rd, Takemori H, Okamoto M, Montminy M (2004) The CREB coactivator TORC2 functions as a calcium- and cAMP-sensitive coincidence detector. Cell 119(1):61–74
Settembre C, Di Malta C, Polito VA, Garcia-Arencibia M, Vetrini F, Erdin S, Erdin SU, Huynh T, Medina D, Colella P, Sardiello M, Rubinsztein DC, Ballabio A (2011) TFEB links autophagy to lysosomal biogenesis. Science 332(6036):1429–1433
Settembre C, De Cegli R, Mansueto G, Saha PK, Vetrini F, Visvikis O, Tuong H, Carissimo A, Palmer D, Klisch TJ, Wollenberg AC, Di Bernardo D, Chan L, Irazoqui JE, Ballabio A (2013) TFEB controls cellular lipid metabolism through a starvation-induced autoregulatory loop. Nat Cell Biol 15(6):647
Sokos GG, Nikolaidis LA, Mankad S, Elahi D, Shannon RP (2006) Glucagon-like peptide-1 infusion improves left ventricular ejection fraction and functional status in patients with chronic heart failure. J Cardiac Fail 12(9):694–699
Song Y, Altarejos J, Goodarzi MO, Inoue H, Guo X, Berdeaux R, Kim JH, Goode J, Igata M, Paz JC, Hogan MF (2010) CRTC3 links catecholamine signalling to energy balance. Nature 468(7326):933–939
Song Y, Zhai L, Valencia Swain J, Chen Y, Wang P, Chen L, Liu Y, Xiang S (2018) Structural insights into the CRTC2-CREB complex assembly on CRE. J Mol Biol 430(13):1926–1939
Steinberg HO, Baron AD (2002) Vascular function, insulin resistance and fatty acids. Diabetologia 45(5):623–634
Steingrimsson E, Copeland NG, Jenkins NA (2004) Melanocytes and the microphthalmia transcription factor network. Annu Rev Genet 38:365–411
Than TA, Lou H, Ji C, Win S, Kaplowitz N (2011) Role of cAMP-responsive element-binding protein (CREB)-regulated transcription coactivator 3 (CRTC3) in the initiation of mitochondrial biogenesis and stress response in liver cells. J Biol Chem 286(25):22047–22054
Trivedi PC, Bartlett JJ, Perez LJ, Brunt KR, Legare JF, Hassan A, Kienesberger PC, Pulinilkunnil T (2016) Glucolipotoxicity diminishes cardiomyocyte TFEB and inhibits lysosomal autophagy during obesity and diabetes. BBA-Mol Cell Biol L 1861(12):1893–1910
van Bilsen M, van Nieuwenhoven FA, van der Vusse GJ (2009) Metabolic remodelling of the failing heart: beneficial or detrimental? Cardiovasc Res 81(3):420–428
Vergnes L, Chin RG, de Aguiar T, Vallim LG, Fong TF, Osborne SG, Young KR (2016) SREBP-2-deficient and hypomorphic mice reveal roles for SREBP-2 in embryonic development and SREBP-1c expression. J Lipid Res 57(3):410–421. https://doi.org/10.1194/jlr.M064022
Wang B, Goode J, Best J, Meltzer J, Schilman PE, Chen J, Garza D, Thomas JB, Montminy M (2008) The insulin-regulated CREB coactivator TORC promotes stress resistance in Drosophila. Cell Metab 7(5):434–444
Wang Y, Li G, Goode J, Paz JC, Ouyang K, Screaton R, Fischer WH, Chen J, Tabas I, Montminy M (2012) Inositol-1,4,5-trisphosphate receptor regulates hepatic gluconeogenesis in fasting and diabetes. Nature 485(7396):128-U166
Wang B, Yang Q, Sun Y-Y, Xing Y-F, Wang Y-B, Lu X-T, Bai W-W, Liu X-Q, Zhao Y-X (2014) Resveratrol-enhanced autophagic flux ameliorates myocardial oxidative stress injury in diabetic mice. J Cell Mol Med 18(8):1599–1611
Wang C, Song D, Fu J, Wen X (2020) SIK1 regulates CRTC2-mediated gluconeogenesis signaling pathway in human and mouse liver cells. Front Endocrinol. https://doi.org/10.3389/fendo.2020.00580
Winkels H, Ehinger E, Vassallo M, Buscher K, Dinh HQ, Kobiyama K, Hamers AAJ, Cochain C, Vafadarnejad E, Saliba A-E, Zernecke A, Pramod AB, Ghosh AK, Michel NA, Hoppe N, Hilgendorf I, Zirlik A, Hedrick CC, Ley K, Wolf D (2018) Atlas of the immune cell repertoire in mouse atherosclerosis defined by single-cell RNA-sequencing and mass cytometry. Circ Res 122(12):1675–1688
Wittenbecher C, Eichelmann F, Toledo E, Guasch-Ferre M, Ruiz-Canela M, Li J, Aros F, Lee C-H, Liang L, Salas-Salvado J, Clish CB, Schulze MB, Martinez-Gonzalez MA, Hu FB (2021) Lipid profiles and heart failure risk results from two prospective studies. Circ Res 128(3):309–320
Wu Z, Huang X, Feng Y, Handschin C, Feng Y, Gullicksen PS, Bare O, Labow M, Spiegelman B, Stevenson SC (2006) Transducer of regulated CREB-binding proteins (TORCs) induce PGC-1 alpha transcription and mitochondrial biogenesis in muscle cells. Proc Natl Acad Sci USA 103(39):14379–14384
Wu L, Wang K, Wang W, Wen Z, Wang P, Liu L, Wang DW (2018) Glucagon-like peptide-1 ameliorates cardiac lipotoxicity in diabetic cardiomyopathy via the PPAR alpha pathway. Aging Cell 17(4):e12763
Yang S, Yuan H-Q, Hao Y-M, Ren Z, Qu S-L, Liu L-S, Wei D-H, Tang Z-H, Zhang J-F, Jiang Z-S (2020) Macrophage polarization in atherosclerosis. Clin Chim Acta 501:142–146
Yoon Y-S, Lee M-W, Ryu D, Kim JH, Ma H, Seo W-Y, Kim Y-N, Kim SS, Lee CH, Hunter T, Choi CS, Montminy MR, Koo S-H (2010) Suppressor of MEK null (SMEK)/protein phosphatase 4 catalytic subunit (PP4C) is a key regulator of hepatic gluconeogenesis. Proc Natl Acad Sci USA 107(41):17704–17709
Yoon Y-S, Liu W, Van de Velde S, Matsumura S, Wiater E, Huang L, Montminy M (2021) Activation of the adipocyte CREB/CRTC pathway in obesity. Commun Biol. https://doi.org/10.1038/s42003-021-02735-5
Zhang X, Luo Y, Wang C, Ding X, Yang X, Wu D, Silva F, Yang Z, Zhou Q, Wang L, Wang X, Zhou J, Boyd N, Spafford M, Burge M, Yang XO, Liu M (2018) Adipose mTORC1 suppresses prostaglandin signaling and beige adipogenesis via the CRTC2-COX-2 pathway. Cell Rep 24(12):3180–3193
Zhu X, Li H, Wu Y, Zhou J, Yang G, Wang W, Kang D, Ye S (2019) CREB-upregulated lncRNA MEG3 promotes hepatic gluconeogenesis by regulating miR-302a-3p-CRTC2 axis. J Cell Biochem 120(3):4192–4202
Acknowledgements
This work was supported by National Natural Science Foundation of China (81670429, 91839103 to ZSJ), International Joint Laboratory for Arteriosclerotic Disease Research of Hunan Province (2018WK4031), Special Funding for Construction of Innovative Provinces in Hunan Province (2020SK2105, to ZSJ) and “Double First-Class” project for innovative Group of Basic Medicine, University of South China (to ZSJ).
Funding
National Natural Science Foundation of China, 81670429, zhisheng jiang, 91839103, zhisheng jiang, International Joint Laboratory for MicroNano Manufacturing and Measurement Technologies, 2018WK4031, zhisheng jiang, Special Funding for Construction of Innovative Provinces in Hunan Province, 2020SK2105, zhisheng jiang, “Double First-Class” project for innovative Group of Basic Medicine, University of South China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, HY., Wang, YX., Zhou, K. et al. Biological functions of CRTC2 and its role in metabolism-related diseases. J. Cell Commun. Signal. 17, 495–506 (2023). https://doi.org/10.1007/s12079-023-00730-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-023-00730-5