Abstract
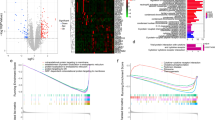
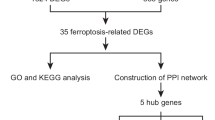
Hepatic ischemia–reperfusion (I/R) injury is a severe clinical syndrome, causing a profound medical and socioeconomic burden worldwide. This study aimed to explore underlying biomarkers and treatment targets in the progression of hepatic I/R injury. We screened gene expression profiles of the hepatic I/R injury from the Gene Expression Omnibus (GEO) database, downloaded expression profiles data (GSE117066). Differentially expressed genes (DEGs) were identified through cluster of the PPI network, and enrichment pathways were conducted based on gene ontology (GO), Kyoto Encyclopedia of Genes and Genomes (KEGG) database. The bioinformatics analysis was used to identify biomarkers that alleviate hepatic I/R injury. Finally, the effects of hub gene were investigated by in vitro and in vivo experiments. A total of 162 DEGs (76 up-regulated and 86 down-regulated genes) were extracted between sham and I/R, and 248 DEGs (118 up-regulated and 130 down-regulated genes) were extracted between I/R and ischemic postconditioning (IPO). The cluster of the PPI network and maximal clique centrality (MCC) method of the common DEGs were performed to identify the phosphoserine aminotransferase 1 (PSAT1) as the potential gene for hepatic I/R injury. Then, the H-E, TUNEL and PCNA staining were indicated that the hepatic injury score was highest in I/R 6 h. The expression level of apoptosis-related proteins was consistent with the pathological results. Both gain- and loss-of-function assays demonstrated that hepatic I/R injury was alleviated by PSAT1. PSAT1 may play crucial roles in hepatic I/R injury and thus serves as a hub biomarker for hepatic I/R injury prognosis and individual-based treatment.
Graphical abstract








Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Amelio I, Cutruzzolá F, Antonov A, Agostini M, Melino G (2014) Serine and glycine metabolism in cancer. Trends Biochem Sci 39(4):191–198
DeNicola GM, Chen PH, Mullarky E, Sudderth JA, Hu Z, Wu D, Tang H, Xie Y, Asara JM, Huffman KE et al (2015) NRF2 regulates serine biosynthesis in non-small cell lung cancer. Nat Genet 47(12):1475–1481
Elias-Miró M, Jiménez-Castro MB, Rodés J, Peralta C (2013) Current knowledge on oxidative stress in hepatic ischemia/reperfusion. Free Radical Res 47(8):555–568
Hu Y, Tao X, Han X, Xu L, Yin L, Sun H, Qi Y, Xu Y, Peng J (2018) MicroRNA-351-5p aggravates intestinal ischaemia/reperfusion injury through the targeting of MAPK13 and Sirtuin-6. Br J Pharmacol 175(17):3594–3609
Jaeschke H (2000) Reactive oxygen and mechanisms of inflammatory liver injury. J Gastroenterol Hepatol 15(7):718–724
Konishi T, Lentsch AB (2017) Hepatic ischemia/reperfusion: mechanisms of tissue injury, repair, and regeneration. Gene Expr 17(4):277–287
Pacold ME, Brimacombe KR, Chan SH, Rohde JM, Lewis CA, Swier LJ, Possemato R, Chen WW, Sullivan LB, Fiske BP et al (2016) A PHGDH inhibitor reveals coordination of serine synthesis and one-carbon unit fate. Nat Chem Biol 12(6):452–458
Penna C, Tullio F, Moro F, Folino A, Merlino A, Pagliaro P (2010) Effects of a protocol of ischemic postconditioning and/or captopril in hearts of normotensive and hypertensive rats. Basic Res Cardiol 105(2):181–192
Possemato R, Marks KM, Shaul YD, Pacold ME, Kim D, Birsoy K, Sethumadhavan S, Woo HK, Jang HG, Jha AK et al (2011) Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 476(7360):346–350
Suyavaran A, Thirunavukkarasu C (2017) Preconditioning methods in the management of hepatic ischemia reperfusion-induced injury: Update on molecular and future perspectives. Hepatol Res off J Jpn Soc Hepatol 47(1):31–48
Tao X, Sun X, Xu L, Yin L, Han X, Qi Y, Xu Y, Zhao Y, Wang C, Peng J (2016) Total flavonoids from Rosa Laevigata Michx fruit ameliorates hepatic ischemia/reperfusion injury through inhibition of oxidative stress and inflammation in rats. Nutrients 8(7):418
Uehara T, Bennett B, Sakata ST, Satoh Y, Bilter GK, Westwick JK, Brenner DA (2005) JNK mediates hepatic ischemia reperfusion injury. J Hepatol 42(6):850–859
Yang Y, Wu J, Cai J, He Z, Yuan J, Zhu X, Li Y, Li M, Guan H (2015) PSAT1 regulates cyclin D1 degradation and sustains proliferation of non-small cell lung cancer cells. Int J Cancer 136(4):E39-50
Zhang P, Ming Y, Cheng K, Niu Y, Ye Q (2019) Gene expression profiling in ischemic postconditioning to alleviate mouse liver ischemia/reperfusion injury. Int J Med Sci 16(2):343–354
Acknowledgements
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, Y., Luo, H. The protective effects of phosphoserine aminotransferase 1 (PSAT1) against hepatic ischemia–reperfusion injury. J. Cell Commun. Signal. 17, 851–862 (2023). https://doi.org/10.1007/s12079-023-00727-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-023-00727-0