Abstract
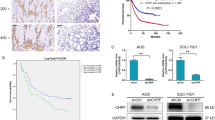
Aberrant composition of glycans in the tumor microenvironment (TME) contributes to tumor progression and metastasis. Chondroitin polymerizing factor (CHPF) is a glycosyltransferase that catalyzes the biosynthesis of chondroitin sulfate (CS). It is also correlated to transforming growth factor-β1 (TGF-β1) expression, a crucial mediator in the interaction of cancer cells with TME. In this study, we investigated the association of CHPF expression with the clinicopathological features of breast cancer (BRCA), as well the oncogenic effect and the underling mechanisms of CHPF upon BRCA cells. We found that CHPF expression is significantly increased in human BRCA tissues, and it is positively associated with TGF-β expression (r = 0.7125). The high-expression of CHPF predicts a poor prognosis and is positively correlated with tumor mass, lymph node metastasis, clinical staging and HER-2 negative-expression. The mechanistic study revealed that it promotes BRCA cell proliferation, migration and invasion through TGF-β1-induced SMAD3 and JNK activation in vitro, JNK (SP600125) or SMAD3 (SIS3) inhibitor can remove the promotion of CHPF upon cell proliferation, migration and invasion in MDA-MB-231 cells, which is derived from triple-negative breast cancer (TNBC). Collectively, our finding suggested CHPF may function as an oncogene and is highly expressed in human BRCA tissues. Pharmacological blockade of the upstream of JNK or SMAD3 signaling may provide a novel therapeutic target for refractory TNBC patients with CHPF abnormal high-expression.





Similar content being viewed by others
Abbreviations
- ANT:
-
Adjacent non-tumor tissue
- AURKA:
-
Aurora-a kinase
- BRCA:
-
Breast cancer
- CAFs:
-
Cancer-associated fibroblasts
- CCA:
-
Cholangiocarcinoma
- CDK:
-
Cyclin-dependent kinase
- CCK8:
-
Cell counting kit 8
- CHPF:
-
Chondroitin polymerizing factor
- CS:
-
Chondroitin sulfate
- DCN:
-
Decorin
- DKK1:
-
Dickkopf-related protein 1
- DMFS:
-
Distant metastasis free survival
- ECM:
-
Extracellular matrix
- ELISA:
-
Enzyme linked immunosorbent assay
- EGFR:
-
Epidermal growth factor receptor
- EMT:
-
Epithelial-mesenchymal transition
- ER:
-
Estrogen receptor
- ERK:
-
Extracellular signal-regulated kinase
- FBS:
-
Fetal bovine serum
- GAGs:
-
Glycosaminoglycans
- GSK-3β:
-
Glycogen synthase kinase-3β
- HCC:
-
Hepatocellular carcinoma
- HER2:
-
Human epidermal growth factor receptor 2
- IGHG1:
-
Immunoglobulin γ-1 heavy chain constant region
- IHC:
-
Immunohistochemistry
- IKK:
-
Inhibitor of kappa kinase
- JNK:
-
C-Jun N-terminal kinase
- LTBP1:
-
Latent transforming growth factor (TGF) beta-binding protein 1
- MAPK:
-
Mitogen activated protein kinase
- OS:
-
Overall survival
- PCP:
-
Planar cell polarity
- PMSF:
-
Phenylmethyl sulfonylfluoride
- PR:
-
Progesterone receptor
- PSG9:
-
Pregnancy-specific glycoprotein 9
- PTPRε:
-
Protein tyrosine phosphatase receptor ε
- RAC1:
-
Ras-related C3 botulinum toxin substrate 1
- RhoA:
-
Ras homolog gene family, member A
- ROCK1:
-
Rho-associated coiled-coil kinase 1
- SMAD3:
-
Small mothers against decapentaplegic 3
- SDC4:
-
Syndecan-4
- Sp1:
-
Specificity protein 1
- TAFs:
-
Tumor-associated fibroblasts
- TAK1:
-
TGF-β activated kinase 1
- TCGA:
-
The cancer genome atlas
- TNF-α:
-
Tumor necrosis factor α
- TGF-β1:
-
Transforming growth factor β 1
- TGFBR:
-
Transforming growth factor beta receptor
- TME:
-
Tumor microenvironment
- TNF-α:
-
Tumor necrosis factor α
- TNBC:
-
Triple-negative breast cancer
- WNT:
-
Wingless and int-1
- WWOX:
-
WW domain-containing oxidoreductase
References
Aashaq S, Batool A, Andrabi KI (2019) TAK1 mediates convergence of cellular signals for death and survival. Apoptosis 24:3–20. https://doi.org/10.1007/s10495-018-1490-7
Chen D, Zhang X, Li Z et al (2021) Metabolic regulatory crosstalk between tumor microenvironment and tumor-associated macrophages. Theranostics 11:1016–1030. https://doi.org/10.7150/thno.51777
Colak S, Ten Dijke P (2017) Targeting TGF-β signaling in cancer. Trends Cancer 3:56–71. https://doi.org/10.1016/j.trecan.2016.11.008
Duan X, Yang J, Jiang B et al (2021) Identification of chondroitin polymerizing factor (CHPF) as tumor promotor in cholangiocarcinoma through regulating cell proliferation, cell apoptosis and cell migration. Cell Cycle 20:591–602. https://doi.org/10.1080/15384101.2021.1890951
Fan YH, Xiao B, Lv SG et al (2017) Lentivirus-mediated knockdown of chondroitin polymerizing factor inhibits glioma cell growth in vitro. Oncol Rep 38:1149–1155. https://doi.org/10.3892/or.2017.5731
Fujiki K, Inamura H, Sugaya T et al (2019) Blockade of ALK4/5 signaling suppresses cadmium- and erastin-induced cell death in renal proximal tubular epithelial cells via distinct signaling mechanisms. Cell Death Differ 26:2371–2385. https://doi.org/10.1038/s41418-019-0307-8
Hao Y, Baker D, Ten Dijke P (2019) TGF-β-mediated epithelial-mesenchymal transition and cancer metastasis. Int J Mol Sci. https://doi.org/10.3390/ijms20112767
Hinshaw DC, Shevde LA (2019) The tumor microenvironment innately modulates cancer progression. Cancer Res 79:4557–4566. https://doi.org/10.1158/0008-5472.Can-18-3962
Hou XM, Baloch Z, Zheng ZH et al (2019a) Knockdown of CHPF suppresses cell progression of non-small-cell lung cancer. Cancer Manag Res 11:3275–3283. https://doi.org/10.2147/cmar.S192036
Hou XM, Zhang T, Da Z et al (2019b) CHPF promotes lung adenocarcinoma proliferation and anti-apoptosis via the MAPK pathway. Pathol Res Pract 215:988–994. https://doi.org/10.1016/j.prp.2019.02.005
Hu B, Xu C, Cao P et al (2018) TGF-β stimulates expression of chondroitin polymerizing factor in nucleus pulposus cells through the Smad3, RhoA/ROCK1, and MAPK signaling pathways. J Cell Biochem 119:566–579. https://doi.org/10.1002/jcb.26215
Huang M, Fu M, Wang J et al (2021) TGF-β1-activated cancer-associated fibroblasts promote breast cancer invasion, metastasis and epithelial-mesenchymal transition by autophagy or overexpression of FAP-α. Biochem Pharmacol 188:114527. https://doi.org/10.1016/j.bcp.2021.114527
Ishimaru D, Sugiura N, Akiyama H et al (2014) Alterations in the chondroitin sulfate chain in human osteoarthritic cartilage of the knee. Osteoarthritis Cartilage 22:250–258. https://doi.org/10.1016/j.joca.2013.11.010
Izumikawa T, Kitagawa H, Mizuguchi S et al (2004) Nematode chondroitin polymerizing factor showing cell-/organ-specific expression is indispensable for chondroitin synthesis and embryonic cell division. J Biol Chem 279:53755–53761. https://doi.org/10.1074/jbc.M409615200
Jalalirad M, Haddad TC, Salisbury JL et al (2021) Aurora-A kinase oncogenic signaling mediates TGF-β-induced triple-negative breast cancer plasticity and chemoresistance. Oncogene 40:2509–2523. https://doi.org/10.1038/s41388-021-01711-x
Ji K, Chen J, Hu J et al (2015) The protective effect of astragaloside IV against benzo[a]pyrene induced endothelial progenitor cell dysfunction. Life Sci 132:13–19. https://doi.org/10.1016/j.lfs.2015.04.002
Khawaled S, Nigita G, Distefano R et al (2020) Pleiotropic tumor suppressor functions of WWOX antagonize metastasis. Signal Transduct Target Ther 5:43. https://doi.org/10.1038/s41392-020-0136-8
Li M, Sun J, Zhang W et al (2021a) Drug delivery systems based on CD44-targeted glycosaminoglycans for cancer therapy. Carbohydr Polym 251:117103. https://doi.org/10.1016/j.carbpol.2020.117103
Li Y, Gong H, Feng L et al (2021b) Chondroitin polymerizing factor promotes breast carcinoma cell proliferation, invasion and migration and affects expression of epithelial-mesenchymal transition-related markers. FEBS Open Bio 11:423–434. https://doi.org/10.1002/2211-5463.13062
Li Y, Wang P, Ye D et al (2021c) IGHG1 induces EMT in gastric cancer cells by regulating TGF-β/SMAD3 signaling pathway. J Cancer 12:3458–3467. https://doi.org/10.7150/jca.56056
Liao SJ, Luo J, Li D et al (2019) TGF-β1 and TNF-α synergistically induce epithelial to mesenchymal transition of breast cancer cells by enhancing TAK1 activation. J Cell Commun Signal 13:369–380. https://doi.org/10.1007/s12079-019-00508-8
Liao Z, Chen L, Zhang X et al (2020) PTPRε acts as a metastatic promoter in hepatocellular carcinoma by facilitating recruitment of SMAD3 to TGF-β receptor 1. Hepatology 72:997–1012. https://doi.org/10.1002/hep.31104
Liao WC, Yen HR, Chen CH et al (2021) CHPF promotes malignancy of breast cancer cells by modifying syndecan-4 and the tumor microenvironment. Am J Cancer Res 11:812–826
Lin X, Han T, Xia Q et al (2021) CHPF promotes gastric cancer tumorigenesis through the activation of E2F1. Cell Death Dis 12:876. https://doi.org/10.1038/s41419-021-04148-y
Liu YY, Zhang S, Yu TJ et al (2020) Pregnancy-specific glycoprotein 9 acts as both a transcriptional target and a regulator of the canonical TGF-β/Smad signaling to drive breast cancer progression. Clin Transl Med 10:e245. https://doi.org/10.1002/ctm2.245
Liu CH, Wu BR, Ho YJ et al (2021) CHPF regulates the aggressive phenotypes of hepatocellular carcinoma cells via the modulation of the decorin and TGF-β pathways. Cancers (basel). https://doi.org/10.3390/cancers13061261
Lu X, Zhong Q, Liu J et al (2020) Efficient expression of chondroitinase ABC I for specific disaccharides detection of chondroitin sulfate. Int J Biol Macromol 143:41–48. https://doi.org/10.1016/j.ijbiomac.2019.11.215
Mashouri L, Yousefi H, Aref AR et al (2019) Exosomes: composition, biogenesis, and mechanisms in cancer metastasis and drug resistance. Mol Cancer 18:75. https://doi.org/10.1186/s12943-019-0991-5
Ooshima A, Park J, Kim SJ (2019) Phosphorylation status at Smad3 linker region modulates transforming growth factor-β-induced epithelial-mesenchymal transition and cancer progression. Cancer Sci 110:481–488. https://doi.org/10.1111/cas.13922
Park SB, Hwang KT, Chung CK et al (2020) Causal bayesian gene networks associated with bone, brain and lung metastasis of breast cancer. Clin Exp Metastasis 37:657–674. https://doi.org/10.1007/s10585-020-10060-0
Pudełko A, Wisowski G, Olczyk K et al (2019) The dual role of the glycosaminoglycan chondroitin-6-sulfate in the development, progression and metastasis of cancer. Febs j 286:1815–1837. https://doi.org/10.1111/febs.14748
Sharma R, Kuche K, Thakor P et al (2022) Chondroitin sulfate: emerging biomaterial for biopharmaceutical purpose and tissue engineering. Carbohydr Polym 286:119305. https://doi.org/10.1016/j.carbpol.2022.119305
Silagi ES, Shapiro IM, Risbud MV (2018) Glycosaminoglycan synthesis in the nucleus pulposus: dysregulation and the pathogenesis of disc degeneration. Matrix Biol 71–72:368–379. https://doi.org/10.1016/j.matbio.2018.02.025
Sun W, Zhao F, Xu Y et al (2020) Chondroitin polymerizing factor (CHPF) promotes development of malignant melanoma through regulation of CDK1. Cell Death Dis 11:496. https://doi.org/10.1038/s41419-020-2526-9
Tamura Y, Tsutsumi S, Miyazono K et al (2022) PolyI: C attenuates transforming growth factor-β signaling to induce cytostasis of surrounding cells by secreted factors in triple-negative breast cancer. Cancer Sci 113:940–949. https://doi.org/10.1111/cas.15241
Vishnubalaji R, Alajez NM (2021) Epigenetic regulation of triple negative breast cancer (TNBC) by TGF-β signaling. Sci Rep 11:15410. https://doi.org/10.1038/s41598-021-94514-9
Xu Q, Lin W, Tao C et al (2020) Chondroitin polymerizing factor (CHPF) contributes to malignant proliferation and migration of hepatocellular carcinoma cells. Biochem Cell Biol 98:362–369. https://doi.org/10.1139/bcb-2019-0227
Yin Y, Chen X, Shu Y (2009) Gene expression of the invasive phenotype of TNF-alpha-treated MCF-7 cells. Biomed Pharmacother 63:421–428. https://doi.org/10.1016/j.biopha.2009.04.032
Yin L, Duan JJ, Bian XW et al (2020) Triple-negative breast cancer molecular subtyping and treatment progress. Breast Cancer Res 22:61. https://doi.org/10.1186/s13058-020-01296-5
Yu H, Wu CL, Wang X et al (2019) SP600125 enhances C-2-induced cell death by the switch from autophagy to apoptosis in bladder cancer cells. J Exp Clin Cancer Res 38:448. https://doi.org/10.1186/s13046-019-1467-6
Zhang J, Pei Y, Yang W et al (2019) Cytoglobin ameliorates the stemness of hepatocellular carcinoma via coupling oxidative-nitrosative stress signals. Mol Carcinog 58:334–343. https://doi.org/10.1002/mc.22931
Zhang J, Yang J, Zuo T et al (2021) Heparanase-driven sequential released nanoparticles for ferroptosis and tumor microenvironment modulations synergism in breast cancer therapy. Biomaterials 266:120429. https://doi.org/10.1016/j.biomaterials.2020.120429
Zhao Y, Ma J, Fan Y et al (2018) TGF-β transactivates EGFR and facilitates breast cancer migration and invasion through canonical Smad3 and ERK/Sp1 signaling pathways. Mol Oncol 12:305–321. https://doi.org/10.1002/1878-0261.12162
Zhuang X, Zhang H, Li X et al (2017) Differential effects on lung and bone metastasis of breast cancer by Wnt signalling inhibitor DKK1. Nat Cell Biol 19:1274–1285. https://doi.org/10.1038/ncb3613
Acknowledgements
This project was granted by the National Natural Science Foundation of China (81860544, 82060565).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were disclosed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, QF., Ouyang, WW., Zhang, MQ. et al. Chondroitin polymerizing factor predicts a poor prognosis and promotes breast cancer progression via the upstream TGF-β1/SMAD3 and JNK axis activation. J. Cell Commun. Signal. 17, 89–102 (2023). https://doi.org/10.1007/s12079-022-00684-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-022-00684-0