Abstract
Background
Droplet digital PCR (ddPCR) is increasingly used in diagnosing clinical pathogens, but its effectiveness in cirrhosis patients with suspected ascites infection remains uncertain.
Methods
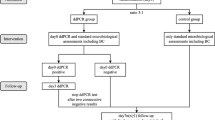
The diagnostic performance of ddPCR was assessed in 305 ascites samples, utilizing culture and clinical composite standards. The quantitative value and potential clinical impact of ddPCR were further analyzed in patients with spontaneous bacterial peritonitis.
Results
With culture standards, ddPCR demonstrated a sensitivity of 86.5% and specificity of 83.2% for bacterial or fungal detection. After adjustment of clinical composite criteria, specificity increased to 96.4%. Better diagnostic performance for all types of targeted pathogens, particularly fungi, was observed with ddPCR compared to culture, and more polymicrobial infections were detected (30.4% versus 5.7%, p < 0.001). Pathogen loads detected by ddPCR correlated with white blood cell count in ascites and blood, as well as polymorphonuclear cell (PMN) count in ascites, reflecting infection status rapidly. A positive clinical impact of 55.8% (43/77) was observed for ddPCR, which was more significant among patients with PMN count ≤ 250/mm3 in terms of medication adjustment and new diagnosis. ddPCR results for fungal detection were confirmed by clinical symptoms and other microbiological tests, which could guide antifungal therapy and reduce the risk of short-term mortality.
Conclusions
ddPCR, with appropriate panel design, has advantages in pathogen detection and clinical management of ascites infection, especially for patients with fungal and polymicrobial infections. Patients with atypical spontaneous bacterial peritonitis benefited more from ddPCR.




Similar content being viewed by others
Data availability
The data presented in this study are available in article or supplementary material.
Abbreviations
- CRP:
-
C-reactive protein
- ddPCR:
-
Droplet digital PCR
- mNGS:
-
Metagenomic next-generation sequencing
- MN:
-
Mononuclear cell
- NIA:
-
Noninfectious ascites
- PMN:
-
Polymorphonuclear cell
- PCT:
-
Procalcitonin
- SBP:
-
Spontaneous bacterial peritonitis
- WBC:
-
White blood cell
References
Aithal GP, Palaniyappan N, China L, Härmälä S, Macken L, Ryan JM, et al. Guidelines on the management of ascites in cirrhosis. Gut. 2021;70:9–29
Rimola A, García-Tsao G, Navasa M, Piddock LJ, Planas R, Bernard B, et al. Diagnosis, treatment and prophylaxis of spontaneous bacterial peritonitis: a consensus document. International Ascites Club J Hepatol. 2000;32:142–153
Coburn B, Morris AM, Tomlinson G, Detsky AS. Does this adult patient with suspected bacteremia require blood cultures? JAMA. 2012;308:502–511
Gu W, Deng X, Lee M, Sucu YD, Arevalo S, Stryke D, et al. Rapid pathogen detection by metagenomic next-generation sequencing of infected body fluids. Nat Med. 2021;27:115–124
Miller S, Naccache SN, Samayoa E, Messacar K, Arevalo S, Federman S, et al. Laboratory validation of a clinical metagenomic sequencing assay for pathogen detection in cerebrospinal fluid. Genome Res. 2019;29:831–842
Timbrook TT, Morton JB, McConeghy KW, Caffrey AR, Mylonakis E, LaPlante KL. The effect of molecular rapid diagnostic testing on clinical outcomes in bloodstream infections: a systematic review and meta-analysis. Clin Infect Dis. 2017;64:15–23
Pliakos EE, Andreatos N, Shehadeh F, Ziakas PD, Mylonakis E. The cost-effectiveness of rapid diagnostic testing for the diagnosis of bloodstream infections with or without antimicrobial stewardship. Clin Microbiol Rev. 2018;31:10–128
Wu HX, Wei FL, Zhang W, Han J, Guo S, Wang Z, et al. Clinical evaluation of metagenomic next-generation sequencing method for the diagnosis of suspected ascitic infection in patients with liver cirrhosis in a clinical laboratory. Microbiol Spectr. 2023;11: e0294622
Miao Q, Ma Y, Wang Q, Pan J, Zhang Y, Jin W, et al. Microbiological diagnostic performance of metagenomic next-generation sequencing when applied to clinical practice. Clin Infect Dis. 2018;67:S231-s240
Toma I, Siegel MO, Keiser J, Yakovleva A, Kim A, Davenport L, et al. Single-molecule long-read 16S sequencing to characterize the lung microbiome from mechanically ventilated patients with suspected pneumonia. J Clin Microbiol. 2014;52:3913–3921
Merino I, de la Fuente A, Domínguez-Gil M, Eiros JM, Tedim AP, Bermejo-Martín JF. Digital PCR applications for the diagnosis and management of infection in critical care medicine. Crit Care. 2022;26:63
Li H, Bai R, Zhao Z, Tao L, Ma M, Ji Z, et al. Application of droplet digital PCR to detect the pathogens of infectious diseases. Biosci Rep. 2018;38:BSR20181170
Wang J, Ye Q, Zheng W, Yu X, Luo F, Fang R, et al. Low-ratio somatic NLRC4 mutation causes late-onset autoinflammatory disease. Ann Rheum Dis. 2022;81:1173–1178
Zhou L, Wang W, Wang F, Yang S, Hu J, Lu B, et al. Plasma-derived exosomal miR-15a-5p as a promising diagnostic biomarker for early detection of endometrial carcinoma. Mol Cancer. 2021;20:57
Tsuchida N, Kunishita Y, Uchiyama Y, Kirino Y, Enaka M, Yamaguchi Y, et al. Pathogenic UBA1 variants associated with VEXAS syndrome in Japanese patients with relapsing polychondritis. Ann Rheum Dis. 2021;80:1057–1061
Sorbini M, Togliatto GM, Simonato E, Boffini M, Cappuccio M, Gambella A, et al. HLA-DRB1 mismatch-based identification of donor-derived cell free DNA (dd-cfDNA) as a marker of rejection in heart transplant recipients: a single-institution pilot study. J Heart Lung Transplant. 2021;40:794–804
Hu B, Tao Y, Shao Z, Zheng Y, Zhang R, Yang X, et al. A comparison of blood pathogen detection among droplet digital PCR, metagenomic next-generation sequencing, and blood culture in critically Ill patients with suspected bloodstream infections. Front Microbiol. 2021;12: 641202
Dickson RP, Schultz MJ, van der Poll T, Schouten LR, Falkowski NR, Luth JE, et al. Lung microbiota predict clinical outcomes in critically Ill patients. Am J Respir Crit Care Med. 2020;201:555–563
Bermejo-Martin JF, González-Rivera M, Almansa R, Micheloud D, Tedim AP, Domínguez-Gil M, et al. Viral RNA load in plasma is associated with critical illness and a dysregulated host response in COVID-19. Crit Care. 2020;24:691
Zhou F, Sun S, Sun X, Chen Y, Yang X. Rapid and sensitive identification of pleural and peritoneal infections by droplet digital PCR. Folia Microbiol (Praha). 2021;66:213–219
European Association for the Study of the Liver. Electronic address eee, European association for the study of the L. EASL clinical practice guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406–460
Biggins SW, Angeli P, Garcia-Tsao G, Ginès P, Ling SC, Nadim MK, et al. Diagnosis, evaluation, and management of ascites, spontaneous bacterial peritonitis and hepatorenal syndrome: 2021 practice guidance by the American association for the study of liver diseases. Hepatology. 2021;74:1014–1048
Xu X, Duan Z, Ding H, Li W, Jia J, Wei L, et al. Chinese guidelines on the management of ascites and its related complications in cirrhosis. Hepatol Int. 2019;13:1–21
Zhu Y, Gan M, Ge M, Dong X, Yan G, Zhou Q, et al. Diagnostic performance and clinical impact of metagenomic next-generation sequencing for pediatric infectious diseases. J Clin Microbiol. 2023;61: e0011523
Moehring RW, Ashley ESD, Davis AE, Dyer AP, Parish A, Ren X, et al. Development of an electronic definition for de-escalation of antibiotics in hospitalized patients. Clin Infect Dis. 2021;73:e4507–e4514
Dever JB, Sheikh MY. Review article: spontaneous bacterial peritonitis–bacteriology, diagnosis, treatment, risk factors and prevention. Aliment Pharmacol Ther. 2015;41:1116–1131
Wu J, Tang B, Qiu Y, Tan R, Liu J, Xia J, et al. Clinical validation of a multiplex droplet digital PCR for diagnosing suspected bloodstream infections in ICU practice: a promising diagnostic tool. Crit Care. 2022;26:243
Lin K, Zhao Y, Xu B, Yu S, Fu Z, Zhang Y, et al. Clinical diagnostic performance of droplet digital PCR for suspected bloodstream infections. Microbiol Spectr. 2023;11: e0137822
Wouters Y, Dalloyaux D, Christenhusz A, Roelofs HMJ, Wertheim HF, Bleeker-Rovers CP, et al. Droplet digital polymerase chain reaction for rapid broad-spectrum detection of bloodstream infections. Microb Biotechnol. 2020;13:657–668
Francés R, Muñoz C, Zapater P, Uceda F, Gascón I, Pascual S, et al. Bacterial DNA activates cell mediated immune response and nitric oxide overproduction in peritoneal macrophages from patients with cirrhosis and ascites. Gut. 2004;53:860–864
Such J, Francés R, Muñoz C, Zapater P, Casellas JA, Cifuentes A, et al. Detection and identification of bacterial DNA in patients with cirrhosis and culture-negative, nonneutrocytic ascites. Hepatology. 2002;36:135–141
Bernardi M, Angeli P, Claria J, Moreau R, Gines P, Jalan R, et al. Albumin in decompensated cirrhosis: new concepts and perspectives. Gut. 2020;69:1127–1138
Zapater P, Francés R, González-Navajas JM, de la Hoz MA, Moreu R, Pascual S, et al. Serum and ascitic fluid bacterial DNA: a new independent prognostic factor in noninfected patients with cirrhosis. Hepatology. 2008;48:1924–1931
Trebicka J, Bork P, Krag A, Arumugam M. Utilizing the gut microbiome in decompensated cirrhosis and acute-on-chronic liver failure. Nat Rev Gastroenterol Hepatol. 2021;18:167–180
Mohan P, Venkataraman J. Prevalence and risk factors for unsuspected spontaneous ascitic fluid infection in cirrhotics undergoing therapeutic paracentesis in an outpatient clinic. Indian J Gastroenterol. 2011;30:221–224
Wu HX, Hou W, Zhang W, Wang Z, Guo S, Chen DX, et al. Clinical evaluation of bacterial DNA using an improved droplet digital PCR for spontaneous bacterial peritonitis diagnosis. Front Cell Infect Microbiol. 2022;12: 876495
Kumar M, Mugunthan M. Evaluation of three DNA extraction methods from fungal cultures. Med J Armed Forces India. 2018;74:333–336
Perez KK, Olsen RJ, Musick WL, Cernoch PL, Davis JR, Land GA, et al. Integrating rapid pathogen identification and antimicrobial stewardship significantly decreases hospital costs. Arch Pathol Lab Med. 2013;137:1247–1254
Lehmann LE, Herpichboehm B, Kost GJ, Kollef MH, Stuber F. Cost and mortality prediction using polymerase chain reaction pathogen detection in sepsis: evidence from three observational trials. Crit Care. 2010;14:R186
Funding
This work was supported by the Beijing Municipal Science & Technology Commission, Administrative Commission of Zhongguancun Science Park (grant number Z211100002921004), and Clinical Research Special Fund of Wu Jieping Medical Foundation of China (grant number 320.6750.2022–08-1), the Capital Health Research and Development of Special Fund Program (grant number 2022–2-2183).
Author information
Authors and Affiliations
Contributions
All authors approved the article. JH (First Author): Conceptualization, Methodology, Formal Analysis, Validation, Visualization Writing-Original Draft. FW (Co-first Author): Conceptualization, Methodology, Resources, Validation. SG: Methodology, Data Curation. HW, LG: Methodology, Resources. YH, YS: Resources. WH (Co-corresponding Author): Supervision, Writing-Review & Editing. ZH (Corresponding Author): Funding Acquisition, Resources, Supervision, Writing-Review & Editing, Project Administration.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Han, J., Wei, Fl., Wu, Hx. et al. Clinical evaluation of droplet digital pcr for suspected ascites infection in patients with liver cirrhosis. Hepatol Int (2024). https://doi.org/10.1007/s12072-024-10669-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12072-024-10669-3