Abstract
Background/purpose
A subgroup analysis of a GLOBE study identified subgroups of chronic hepatitis B (CHB) patients with excellent outcomes to telbivudine (LdT) treatment. The aim of this study was to validate this concept using a real-world clinical population.
Methods
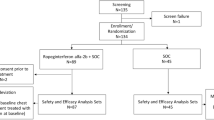
This prospective, retrospective, and multicenter study examined both HBeAg-positive and HBeAg-negative CHB patients treated with LdT for 2 years.
Results
A total of 116 CHB patients were recruited. Of the 64 HBeAg-positive patients, 35 had favorable baseline characteristics [hepatitis B virus (HBV) DNA ≤ 9 log10 copies/mL and alanine aminotransferase ≥ 2× the upper limit of normal (ULN)], but only 40 % (14/35) achieved polymerase chain reaction (PCR) negativity at week 24. Among the 14 patients with favorable baseline characteristics and on-treatment response, the rates of virologic, biochemical, and serologic response and genotypic resistance were 78.6 % (11/14), 64.3 % (9/14), 50 % (7/14), and 7.1 % (1/14), respectively, at week 104 of therapy. Of the 52 HBeAg-negative patients, 34 met the criteria of a baseline serum HBV-DNA level less than 7 log10 copies/mL, and 29 (85.3 %) achieved PCR negativity at week 24. Among the 29 patients with favorable baseline characteristics and on-treatment response, the rates of virologic and biochemical response and genotypic resistance were 96.6 % (28/29), 72.4 % (21/29), and 6.9 % (2/29), respectively. In addition, the PCR negativity at week 24 was the only factor associated with the virologic response and genotypic resistance to LdT treatment.
Conclusion
The efficacy and resistance to LdT treatment in CHB patients with favorable predictors were comparable between a real-world clinical population and the GLOBE study. In addition, PCR negativity at week 24 could predict virologic response and genotypic resistance to LdT treatment.



Similar content being viewed by others
References
Kao JH, Chen DS. Global control of hepatitis B virus infection. Lancet Infect Dis 2002;2(7):395–403
Chen DS. Fighting against viral hepatitis: lessons from Taiwan. Hepatology 2011;54:381–392
Kao JH, Chen PJ, Chen DS. Recent advances in the research of hepatitis B virus-related hepatocellular carcinoma: epidemiologic and molecular biological aspects. Adv Cancer Res 2010;108:21–72
Iloeje UH, Yang HI, Su J, Jen CL, You SL, Chen CJ. Risk evaluation of viral load elevation and associated liver disease/cancer-in HBV (the REVEAL-HBV) study group. Predicting cirrhosis risk based on the level of circulating hepatitis B viral load. Gastroenterology 2006;130(3):678–686
Chen CJ, Yang HI, Su J, Jen CL, You SL, Lu SN, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295(1):65–73
Fattovich G, Pantalena M, Zagni I, Realdi G, Schalm SW, Christensen E, et al. Effect of hepatitis B and C virus infections on the natural history of compensated cirrhosis: a cohort study of 297 patients. Am J Gastroenterol 2002;97:2886–2895
Liaw YF, Sung JJ, Chow WC, Farrell G, Lee CZ, Yuen H, et al. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N Engl J Med 2004;351(15):1521–1531
Chang TT, Liaw YF, Wu SS, Schiff E, Han KH, Lai CL, et al. Long-term entecavir therapy results in the reversal of fibrosis/cirrhosis and continued histological improvement in patients with chronic hepatitis B. Hepatology 2010;52(3):886–893
Wong JS, Wong GL, Tsoi KK, Wong VW, Cheung SY, Chong CN, et al. Meta-analysis: the efficacy of anti-viral therapy in prevention of recurrence after curative treatment of chronic hepatitis B-related hepatocellular carcinoma. Aliment Pharmacol Ther 2011;33(10):1104–1112
Wong GL, Chan HL, Mak CW, Lee SK, Ip ZM, Lam AT, et al. Entecavir treatment reduces hepatic events and deaths in chronic hepatitis B patients with liver cirrhosis. Hepatology 2013;58(5):1537–1547
Wu CY, Lin JT, Ho HJ, Su CW, Lee TY, Wang SY, et al. Association of nucleos(t)ide analogue therapy with reduced risk of hepatocellular carcinoma in patients with chronic hepatitis B—a Nationwide Cohort Study. Gastroenterology 2014;147(1):143–151
Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology 2009;50:661–662
European Association For the Study of the Liver. EASL clinical practice guidelines: management of chronic hepatitis B virus infection. J Hepatol 2012;57:167–185
Liaw YF, Kao JH, Piratvisuth T, Chan HLY, Chien RN, Liu CJ, et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2012 update. Hepatol Int 2012;6:531–561
Peng CY, Hsieh TC, Hsieh TY, Tseng KC, Lin CL, Su TH, et al. HBV-DNA level at 6 months of entecavir treatment predicts HBeAg loss in HBeAg-positive chronic hepatitis B patients. J Formos Med Assoc 2015;114(4):308–313
Yuen MF, Fong DY, Wong DK, Yuen JC, Fung J, Lai CL. Hepatitis B virus DNA levels at week 4 of lamivudine treatment predict the 5-year ideal response. Hepatology 2007;46:1695–1703
Lai CL, Gane E, Liaw YF, Hsu CW, Thongsawat S, Wang Y, et al. Telbivudine versus lamivudinein patients with chronic hepatitis B. N Engl J Med 2007;357:2576–2588
Liaw YF, Gane E, Leung N, Zeuzem S, Wang Y, Lai CL, et al. 2-Year GLOBE trial results: telbivudine is superior to lamivudine in patients with chronic hepatitis B. Gastroenterology 2009;136:486–495
Zeuzem S, Gane E, Liaw YF, Lim SG, DiBisceglie A, Buti M, et al. Baseline characteristics and early on-treatment response predict the outcomes of 2 years of telbivudine treatment of chronic hepatitis B. J Hepatol 2009;51:11–20
Tsai MC, Lee CM, Chiu KW, Hung CH, Tung WC, Chen CH, et al. A comparison of telbivudine and entecavir for chronic hepatitis B in real-world clinical practice. J Antimicrob Chemother 2012;67(3):696–699
Keeffe EB, Zeuzem S, Koff RS, Dieterich DT, Esteban-Mur R, Gane EJ, et al. Report of an international workshop: Roadmap for management of patients receiving oral therapy for chronic hepatitis B. Clin Gastroenterol Hepatol 2007;5:890–897
Gane EJ. The Roadmap concept: using early on-treatment virologic responses to optimize long-term outcomes for patients with chronic hepatitis B. Hepatol Int 2008;2:304–307
Sun J, Xie Q, Tan D, Ning Q, Niu J, Bai X, et al. The 104-week efficacy and safety of telbivudine-based optimization strategy in chronic hepatitis B patients: A randomized, controlled study. Hepatology 2014;59(4):1283–1292
Yu HC, Lin KH, Hsu PI, Tsay FW, Wang HM, Tsai TJ, et al. Real-world application of the roadmap model in chronic hepatitis B patients with telbivudine therapy. Clin Ther 2013;35(9):1386–1399
Zhu XF, Lu LX, Wang Y, Xu KW, Li DJ, Zhu X, et al. Effect and predictive elements for 52 Weeks’ Telbivudine Treatment on Naïve HBeAg positive Chronic Hepatitis B. Hepat Mon 2011;11(12):980–985
Lv GC, Ma WJ, Ying LJ, Jin X, Zheng L, Yang YD. Efficacy of telbivudine in HBeAg-positive chronic hepatitis B patients with high baseline ALT levels. World J Gastroenterol 2010;16(32):4095–4099
Lü W, Yang HH, Fan YM, Li T, Zhang LF, Mui C, et al. Serum HBV DNA level at week 12 is superior to viral response at week 24 in predicting long-term treatment outcome of telbivudine for chronic hepatitis B patients. Chin Med J (Engl) 2013;126(12):2333–2336
Tseng TC, Kao JH. Clinical utility of quantitative HBsAg in natural history and nucleos(t)ide analogue treatment of chronic hepatitis B: new trick of old dog. J Gastroenterol 2013;48(1):13–21
Lee JM, Ahn SH, Kim HS, Park H, Chang HY, Kim do Y, et al. Quantitative hepatitis B surface antigen and hepatitis B e antigen titers in prediction of treatment response to entecavir. Hepatology 2011;53(5):1486–1493
Su TH, Liu CJ, Yang HC, Jeng YM, Cheng HR, Liu CH, et al. Clinical significance and evolution of hepatic HBsAg expression in HBeAg-positive patients receiving interferon therapy. J Gastroenterol 2014;49(2):356–362
Zhao S, Tang L, Fan X, Chen L, Zhou R, Dai X. Comparison of the efficacy of lamivudine and telbivudine in the treatment of chronic hepatitis B: a systematic review. Virol J 2010;7:211
Su QM, Ye XG. Effects of telbivudine and entecavir for HBeAg-positive chronic hepatitis B: a meta-analysis. World J Gastroenterol 2012;18(43):6290–6301
Liang J, Jiang MJ, Deng X, Xiao Zhou X. Efficacy and safety of telbivudine compared to entecavir among HBeAg+ chronic hepatitis B patients: a Meta-Analysis Study. Hepat Mon 2013;13(6):e7862
Wang N, Hu HD, Sun H, Feng Q, Hu P, Liu Q, et al. Comparison of the forty-eight week efficacy between telbivudine and entecavir in HBeAg-positive Asian patients with chronic hepatitis B: A meta-analysis. Turk J Gastroenterol 2013;24(3):230–240
Han GR, Cao MK, Zhao W, Jiang HX, Wang CM, Bai SF, et al. A prospective and open-label study for the efficacy and safety of telbivudine in pregnancy for the prevention of perinatal transmission of hepatitis B virus infection. J Hepatol 2011;55(6):1215–1221
Gane EJ, Deray G, Liaw YF, Lim SG, Lai CL, Rasenack J, et al. Telbivudine improves renal function in patients with chronic hepatitis B. Gastroenterology 2014;146(1):138–146
Acknowledgements
Novartis provided support for the statistical analyses.
Funding
This work was supported by grants from the Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict and interest
Chia-Chi Wang, Chih-Lin Lin, Tsai-Yuan Hsieh, Kuo-Chih Tseng, Cheng-Yuan Peng, Tung-Hung Su, Sheng-Shun Yang, Yu-Chun Hsu, Tsung-Ming Chen and Jia-Horng Kao declare that they have no conflicts of interest.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Taiwan) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all patients for inclusion in the study. This article does not contain any studies with animal subjects.
Rights and permissions
About this article
Cite this article
Wang, CC., Lin, CL., Hsieh, TY. et al. Efficacy and resistance to telbivudine treatment in chronic hepatitis B patients with favorable predictors: a multicenter study in Taiwan. Hepatol Int 10, 294–301 (2016). https://doi.org/10.1007/s12072-015-9662-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-015-9662-9