Abstract
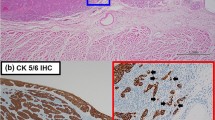
There is a need for novel prognostic parameters in assessing laryngeal squamous cell carcinoma (LSCC). Tumor budding is an instrumental parameter, which has hardly been studied before in this organ. This study aimed to assess tumor budding in LSCCs with routine hematoxylin and eosin (H&E) stain as well as cytokeratin (CK) immunohistochemistry (IHC). Objectives were to compare the effectiveness of both these methods to assess tumor budding, to investigate the association of tumor budding and clinicopathologic features, and to determine the prognostic significance of tumor budding in LSCCs. Fifty laryngectomy specimens were included. Tumor budding was counted (20x) on slides stained with IHC–CK, and highest count per slide was noted. The cases were classified as positive (> 1 buds) or negative (no buds present). The budding index was categorized as low (< 5 buds) or high (> 5 buds). Tumor budding on H&E was absent, low and high grade in 28%, 30% and 42% cases respectively, when compared to CK-IHC in 17%, 24% and 59% of cases, respectively. Presence of lymphoplasmacytic infiltration significantly correlated with tumor budding and higher grade. Transglottic location of tumor and pT stage was associated with high budding. Presence of lymphoplasmacytic infiltrate significantly correlated with worse prognosis. Tumor budding, an easily assessable, inexpensive histopathologic parameter has seldom been studied in LSCCs. Presence of lymphoplasmacytic infiltrate in routine preoperative biopsy reporting could be useful in prognostication. CK-IHC is helpful to detect especially cases with low-grade tumor budding.


Similar content being viewed by others
Availability of Data and Materials
Available on request.
Code Availability
Not applicable.
References
Bobdey S, Jain A, Balasubramanium G (2015) Epidemiological review of laryngeal cancer: an Indian perspective. Indian J Med Paediatr Oncol 36:154–160. https://doi.org/10.4103/0971-5851.166721
Wang C, Huang H, Huang Z, Wang A, Chen X, Huang L et al (2011) Tumor budding correlates with poor prognosis and epithelial mesenchymal transition in tongue squamous cell carcinoma. J Oral Pathol Med 40(7):545–551. https://doi.org/10.1111/j.1600-0714.2011.01041.x
Xie N, Wang C, Liu X, Li R, Hou J, Chen X et al (2015) Tumor budding correlates with occult cervical lymph node metastasis and poor prognosis in clinical early-stage tongue squamous cell carcinoma. J Oral Pathol Med 44:266–272. https://doi.org/10.1111/jop.12242
Kaneoya A, Hasegawa S, Tanaka Y, Omura K (2009) Quantitative analysis of invasive front in tongue cancer using ultrasonography. J Oral Maxillofac Surg 67:40–46. https://doi.org/10.1016/j.joms.2007.08.006
Brandwein-Gensler M, Smith RV, Wang B et al (2010) Validation of the histologic risk model in a new cohort of patients with head and neck squamous cell carcinoma. Am J Surg Pathol 34:676–688. https://doi.org/10.1097/PAS.0b013e3181d95c37
Hayes BD, Maguire A, Conlon N, Gibbons D, Wang LM, Sheahan K (2010) Reproducibility of the rapid bud count method for assessment of tumor budding in stage II colorectal cancer. Am J Surg Pathol 34:746–748. https://doi.org/10.1097/PAS.0b013e3181da76b6
Prall F, Ostwald C, Linnebacher M (2009) Tubular invasion and the morphogenesis of tumor budding in colorectal carcinoma. Hum Pathol 40:1510–1512. https://doi.org/10.1016/j.humpath.2009.06.015
Wang LM, Kevans D, Mulcahy H et al (2009) Tumor budding is a strong and reproducible prognostic marker in T3N0 colorectal cancer. Am J Surg Pathol 33:134–141. https://doi.org/10.1097/PAS.0b013e318184cd55
Brown M, Sillah K, Griffiths EA et al (2010) Tumour budding and a low host inflammatory response are associated with a poor prognosis in oesophageal and gastro-oesophageal junction cancers. Histopathology 56:893–899. https://doi.org/10.1111/j.1365-2559.2010.03559.x
Yamaguchi Y, Ishii G, Kojima M et al (2010) Histopathologic features of the tumor budding in adenocarcinoma of the lung: tumor budding as an index to predict the potential aggressiveness. J Thorac Oncol 5:1361–1368. https://doi.org/10.1111/j.1365-2559.2010.03559.x
Ohike N, Coban I, Kim GE et al (2010) Tumor budding as a strong prognostic indicator in invasive ampullary adenocarcinomas. Am J Surg Pathol 34:1417–1424. https://doi.org/10.1097/PAS.0b013e3181f0b05a
Ueno H, Murphy J, Jass J et al (2002) Tumour ‘budding’ as an index to estimate the potential of aggressiveness in rectal cancer. Histopathology 40:127–132. https://doi.org/10.1046/j.1365-2559.2002.01324.x
Zlobec I (2010) Tumor budding predicts response to anti-EGFR therapies in metastatic colorectal cancer patients. World J Gastroenterol 16:4823–4831. https://doi.org/10.3748/wjg.v16.i38.4823
Thies S, Guldener L, Slotta-Huspenina J et al (2016) Impact of peritumoral and intratumoral budding in esophageal adenocarcinomas. Hum Pathol 52:1–8. https://doi.org/10.1016/j.humpath.2016.01.016
Giger O, Comtesse S, Lugli A et al (2012) Intra-tumoral budding in preoperative biopsy specimens predicts lymph node and distant metastasis in patients with colorectal cancer. Mod Pathol 25:1048–1053. https://doi.org/10.1038/modpathol.2012.56
Miyata H, Yoshioka A, Yamasaki M et al (2009) Tumor budding in tumor invasive front predicts prognosis and survival of patients with esophageal squamous cell carcinomas receiving neoadjuvant chemotherapy. Cancer 115:3324–3334. https://doi.org/10.1002/cncr.24390
Jesinghaus M, Boxberg M, Konukiewitz B et al (2017) A novel grading system based on tumor budding and cell nest size is a strong predictor of patient outcome in esophageal squamous cell carcinoma. Am J Surg Pathol 41:1112–1120. https://doi.org/10.1097/PAS.0000000000000865
Wartenberg M, Zlobec I, Perren A et al (2015) Accumulation of FOXP3+T-cells in the tumor microenvironment is associated with an epithelial-mesenchymal-transition-type tumor budding phenotype and is an independent prognostic factor in surgically resected pancreatic ductal adenocarcinoma. Oncotarget 6:4190–4201. https://doi.org/10.18632/oncotarget.2775
Ekmekci S, Kucuk U, Kokkoz S, Cakir E, Gumussoy M (2019) Tumor budding in laryngeal carcinoma. Indian J Pathol Microbiol 62:7–10. https://doi.org/10.4103/IJPM.IJPM_428_17
Sarioglu S, Acara C, Akman FC, Dag N, Ecevit C, Ikiz AO, Cetinayak OH, Ada E, for Dokuz Eylül Head and Neck Tumour Group (DEHNTG) (2010) Tumor budding as a prognostic marker in laryngeal carcinoma. Pathol Res Pract 206:88–92. https://doi.org/10.1016/j.prp.2009.09.006
Berg KB, Schaeffer DF (2018) Tumor budding as a standardized parameter in gastrointestinal carcinomas: more than just the colon. Mod Pathol 31(6):862–872. https://doi.org/10.1038/s41379-018-0028-4
Funding
This study was funded by the Indian Council of Medical Research (STS 2018–00552).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SG, and SS. The first draft of the manuscript was written by SG, and SS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Conceptualization: SS; Methodology: SG and SS, Formal analysis and investigation: SG and SS; Writing—original draft preparation: SG and SS; Writing—review and editing: ACP, PKS, SHB; Funding acquisition: SG guided by SS; Supervision: SS.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Consent to Participate
Not applicable, performed on de-identified tissue blocks.
Consent for Publication
Not applicable.
Ethics Approval
Approval was obtained from the Institutional Ethics Committee of Kasturba Medical College, Mangalore (IEC KMC MLR 04-18/60).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gupta, S., Sreeram, S., Pinto, A.C. et al. Tumor Budding Assessment with Cytokeratin and Its Significance in Laryngeal Squamous Cell Carcinomas. Indian J Otolaryngol Head Neck Surg 74, 494–500 (2022). https://doi.org/10.1007/s12070-021-02981-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-021-02981-3