Abstract
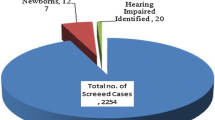
To know the prevalence of hearing loss in preterm infants & to evaluate the sensitivity & specificity of otoacoustic emission (OAE) in detecting hearing loss in preterm infants ≤ 34 weeks. A total of 70 preterm babies from 28 to 34 weeks of gestational age were enrolled in the study. Detailed prenatal, perinatal, postnatal & family history and physical examination of the babies were carried out. First OAE was done at discharge or two weeks after birth, whichever was earlier and the second OAE assessment was done at 36–40 weeks of corrected age. Diagnostic brainstem evoked response audiometry (BERA) was done in all infants at 36–40 weeks of corrected age, at the time of second OAE. Neonates with hearing impairment were advised for early hearing aid amplification and were referred to the rehabilitation center for further management. The sensorineural hearing loss (SNHL) in either one of the ears was identified in 13 (18.57%) preterm babies. Bilateral profound SNHL was found in 5 (7.14%) babies. Auditory neuropathy profile was found in 7 (53.8%) out of 13 babies who had hearing loss. The sensitivity, specificity, positive and negative predictive values of second TEOAE compared with diagnostic BERA was 46.15%, 85.96%, 42.85%, and 87.5% respectively. Neonatal jaundice (p = 0.009) and history of exchange transfusion (p = 0.019) were found to be significant risk factors of hearing loss in our study. Other risk factors like mode of delivery, birth asphyxia, low APGAR score, meningitis, ototoxic drugs, and seizures were not associated with hearing loss. The prevalence of hearing loss in preterm infants < 34 weeks is very high. OAE alone is not an ideal screening test for high-risk neonates ≤ 34 weeks because of its low sensitivity. OAE combined with diagnostic BERA should be done in all high-risk infants preterm neonates ≤ 34 weeks to identify cases of auditory neuropathy spectrum disorders.

Similar content being viewed by others
References
Erenberg A, Lemons J, Sia C, Trunkel D, Ziring P (1999) Newborn and infant hearing loss: detection and intervention. American Academy of Pediatrics. Task force on newborn and infant hearing, 1998–1999. Pediatrics 103:527–530
Berg AL, Spitzer JB, Towers HM, Bartosiewicz C, Diamond BE (2005) Newborn hearing screening in the NICU: profile of failed auditory brainstem response/passed otoacoustic emission. Pediatrics 116:933–938
Al-Kandari JM, Alshuaib WB (2007) Newborn hearing screening in Kuwait. Electromyogr Clin Neurophysiol 47:305–313
Yoshinaga-Itano C, Sedey AL, Coulter DK, Mehl AL (1998) Language of early- and later-identified children with hearing loss. Pediatrics 102:1161–1171
Tasci Y, Muderris II, Erkaya S, Altinbas S, Yucel H, Haberal A (2009) Newborn hearing screening programme outcomes in a research hospital from Turkey. Child Care Health Dev 36:317–322
Xu Z, Li J (2005) Performance of two hearing screening protocols in the NICU. B-ENT 1:11–15
Xu ZM, Cheng WX, Yang XL (2011) Performance of two hearing screening protocols in NICU in Shanghai. Int J Pediatr Otorhinolaryngol 75:1225–1229
Jiang ZD, Wilkinson AR (2008) Normal brainstem responses in moderately preterm infants. Acta Paediatr 97:1366–1369
Colella-Santos MF, Hein TA, de Souza GL, do Amaral MI, Casali RL (2014) Newborn hearing screening and early diagnostic in the NICU. Biomed Res Int. https://doi.org/10.1155/2014/845308
Martines F, Salvago P, Bentivegna D, Bartolone A, Dispenza F, Martines E (2012) Audiologic profile of infants at risk: experience of a Western Sicily tertiary care centre. Int J Pediatr Otorhinolaryngol 76:1285–1291
Pourarian S, Khademi B, Pishva N, Jamali A (2012) Prevalence of hearing loss in newborns admitted to neonatal intensive care unit. Iran J Otorhinolaryngol 24:129–134
Haghshenas M, Zadeh P, Javadian Y, Fard H, Delavari K, Panjaki H, Gorji H (2014) Auditory screening in infants for early detection of permanent hearing loss in northern Iran. Ann Med Health Sci Res 4:340–344
Wolff R, Hommerich J, Riemsma R, Antes G, Lange S, Kleijnen J (2010) Hearing screening in newborns: systematic review of accuracy, effectiveness, and effects of interventions after screening. Arch Dis Child 95:130–135
Kirkim G, Serbetcioglu B, Erdag TK, Ceryan K (2008) The frequency of auditory neuropathy detected by universal newborn hearing screening program. Int J Pediatr Otorhinolaryngol 72:1461–1469
Ngo RY, Tan HK, Balakrishnan A, Lazaroo D, Lim SB, Yan J (2004) Auditory neuropathy detected by universal newborn hearing screening. Cochlear Implants Int 5(Suppl 1):206–208
Swanepoel D, Ebrahim S, Joseph A, Friedland PL (2007) Newborn hearing screening in a South African private health care hospital. Int J Pediatr Otorhinolaryngol 7:881–887
Yousefi J, Ajalloueyan M, Amirsalari S, Hassanali FM (2013) The specificity and sensitivity of transient otoacustic emission in neonatal hearing screening compared with diagnostic test of auditory brain stem response in Tehran hospitals. Iran J Pediatr 23:199–204
Lin HC, Shu MT, Lee KS, Ho GM, Fu TY, Bruna S, Lin G (2005) Comparison of hearing screening programs between one step with transient evoked otoacoustic emissions (TEOAE) and two steps with TEOAE and automated auditory brainstem response. Laryngoscope 115:1957–1962
Benito-Orejas JI, Ramírez B, Morais D, Almaraz A, Fernández-Calvo JL (2008) Comparison of two-step transient evoked otoacoustic emissions (TEOAE) and automated auditory brainstem response (AABR) for universal newborn hearing screening programs. Int J Pediatr Otorhinolaryngol 72:1193–1201
Levit Y, Himmelfarb M, Dollberg S (2015) Sensitivity of the automated auditory brainstem response in neonatal hearing screening. Pediatrics 136(3):e641–e647
Kuki S, Chadha S, Dhingra S, Gulati A (2013) The role of current audiological tests in the early diagnosis of hearing impairment in infant. Indian J Otolaryngol Head Neck Surg 65:244–250
Tobe RG, Mori R, Huang L, Xu L, Han D, Shibuya K (2013) Cost-effectiveness analysis of a national neonatal hearing screening program in China: conditions for the scale-up. PLoS ONE 8:e51990
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial or non-financial conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institution and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed written consent was obtained from parents of the infants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gulati, A., Sakthivel, P., Singh, I. et al. The Hearing Status of Preterm Infant’s ≤ 34 Weeks as Revealed by Otoacoustic Emissions (OAE) Screening and Diagnostic Brainstem Evoked Response Audiometry (BERA): A Tertiary Center Experience. Indian J Otolaryngol Head Neck Surg 74 (Suppl 1), 178–183 (2022). https://doi.org/10.1007/s12070-020-01945-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-020-01945-3