Abstract
1H MAS NMR spectra of Bi0.05Eu0.05Y0.90PO4·xH2O show chemical shift from −0.56 ppm at 300 K to −3.8 ppm at 215 K and another one at 5–6 ppm, which are related to the confined or interstitial water in the hexagonal structure and water molecules on the surface of the particles, respectively. Negative value of the chemical shift indicates that H of H2O is closer to metal ions (Y3 + or Eu3 + ), which is a source of luminescence quencher. H coupling and decoupling 31P MAS NMR spectra at 300 and 250 K show the same chemical shift (−0.4 ppm) indicating that there is no direct bond between P and H. It is concluded that the confined water is not frozen even at 215 K because of the less number of H-bonding.









Similar content being viewed by others
References
M N Luwang, R S Ningthoujam, S K Srivastava, Jaganath and R K Vatsa, J. Am. Chem. Soc. 132, 2759 (2010)
M N Luwang, R S Ningthoujam, S K Srivastava and R K Vatsa, J. Am. Chem. Soc. 133, 2998 (2011)
K Koga, G T Gao, H Tanaka and X C Zeng, Nature 412, 802 (2001)
G Hummer, J C Rasaiah and J P Noworyta, Nature 414, 188 (2001)
S Ghosh, K V Ramanathan and A K Sood, Europhys. Lett. 65, 678 (2004)
V V Chaban and O V Prezhdo, ACS Nano 5, 5647 (2011)
S Ayyappan, N Suryaprakash, K V Ramanathan and C N R Rao, J. Porous Mater. 6, 5 (1999)
J H Strange and M Rahman, Phys. Rev. Lett. 71, 3589 (1993)
K Bhattacharyya, Acc. Chem. Res. 36, 95 (2003)
A A Vartia and W H Thompson, J. Phys. Chem. B 116, 5414 (2012)
S Roy and B Bagchi, J. Phys. Chem. B 116, 2958 (2012)
J A McGuire and Y R Shen, Science 313, 1945 (2006)
J J Gilijamse, A J Lock and H J Bakker, Proc. Natl. Acad. Sci. 102, 3202 (2005)
M Smits, A Ghosh, M Sterrer, M Müller and M Bonn, Phys. Rev. Lett. 98, 098302 (2007)
D E Moilanen, I R Piletic and M D Fayer, J. Phys. Chem. C 111, 8884 (2007)
V V Volkov, D J Palmer and R Righini, J. Phys. Chem. B 111, 1377 (2007)
R K Campen, T T M Ngo, M Sovago, J-M Ruysschaert and M Bonn, J. Am. Chem. Soc. 132, 8037 (2010)
J A Mondal, S Nihonyanagi, S Yamaguchi and T Tahara, J. Am. Chem. Soc. 134, 7842 (2012)
A C L Mooney, Acta Crystallogr. 3, 337 (1957)
G Phaomei, W R Singh and R S Ningthoujam, J. Lumin. 131, 1164 (2011)
S Banerjee, H Ghosh and A Datta, J. Phys. Chem. C 115, 19023 (2011)
P Ghosh and A Patra, J. Phys. Chem. C 112, 3223 (2008)
Y Matsumoto, U Unal, Y Kimura, S Ohashi and K Izawa, J. Phys. Chem. B 109, 12748 (2005)
G L Turner, K A Smith, R J Kirrpatrick and E Oldfield, J. Magn. Resonance 70, 408 (1986)
M Bose, M Bhattacharya and S Ganguli, Phys. Rev. B 19, 72 (1979)
A C Palke and J F Stebbins, American Mineralogist 96, 1343 (2011)
V Ladizhansky, G Hodes and S Vega, J. Phys. Chem. B 104, 1939 (2000)
R S Ningthoujam, V Sudarsan, R K Vatsa, R M Kadam, Jagannath and A Gupta, J. Alloys Compounds 486, 864 (2009)
S Banerjee, S Maity and A Datta, J. Phys. Chem. C 115, 22804 (2011)
S Banerjee and A Datta, Langmuir 26, 1172 (2010)
Acknowledgement
The author is grateful to Dr S Srivastava, Mr M Naik, Ms M Joshi and Mr D Jadhav, National Facility for High Field NMR, Mumbai, India for their extensive help during the experiments.
Author information
Authors and Affiliations
Corresponding author
Appendix: XRD, FTIR, TGA-DTA and PL characterization techniques
Appendix: XRD, FTIR, TGA-DTA and PL characterization techniques
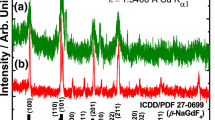
The sample was characterized by the following methods: X-ray diffraction (XRD) patterns were recorded by Inel X-ray diffractometer EQUINOX 1000. Fourier transform infrared (FTIR) spectrum was recorded by FT-IR spectrometer (Bomem MB 102). Thermogravimetric and differential thermal data of sample prepared at 500°C were recorded by TGA-DTA instrument (SETARAM 92-16.18). Weight changes are due to the water content in the sample. Photoluminescence (PL) emission spectra were carried out by Hitachi F-4500 fluorescence spectrometer having a 150 W Xe lamp as the excitation source.
Rights and permissions
About this article
Cite this article
NINGTHOUJAM, R.S. Finding confined water in the hexagonal phase of Bi0.05Eu0.05Y0.90PO4·xH2O and its impact for identifying the location of luminescence quencher. Pramana - J Phys 80, 1055–1064 (2013). https://doi.org/10.1007/s12043-013-0537-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12043-013-0537-0