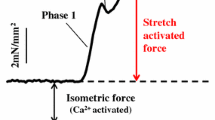
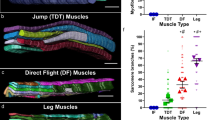
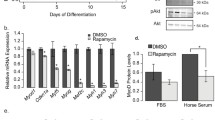
Abstract
Troponin proteins in cooperative interaction with tropomyosin are responsible for controlling the contraction of the striated muscles in response to changes in the intracellular calcium concentration. Contractility of the muscle is determined by the constituent protein isoforms, and the isoforms can switch over from one form to another depending on physiological demands and pathological conditions. In Drosophila, a majority of the myofibrillar proteins in the indirect flight muscles (IFMs) undergo post-transcriptional and post-translational isoform changes during pupal to adult metamorphosis to meet the high energy and mechanical demands of flight. Using a newly generated Gal4 strain (UH3-Gal4) which is expressed exclusively in the IFMs, during later stages of development, we have looked at the developmental and functional importance of each of the troponin subunits (troponin-I, troponin-T and troponin-C) and their isoforms. We show that all the troponin subunits are required for normal myofibril assembly and flight, except for the troponin-C isoform 1 (TnC1). Moreover, rescue experiments conducted with troponin-I embryonic isoform in the IFMs, where flies were rendered flightless, show developmental and functional differences of TnI isoforms and importance of maintaining the right isoform.




Similar content being viewed by others
References
Agianian B., Krzic U., Qiu F., Linke W. A., Leonard K. and Bullard B. 2004 A troponin switch that regulates muscle contraction by stretch instead of calcium. EMBO J. 23, 772–729.
Barbas J. A., Galceran J., Torroja L., Prado A. and Ferrús A. 1993 Abnormal muscle development in the heldup3 mutant of Drosophila melanogaster is caused by a splicing defect affecting selected troponin I isoforms. Mol. Cell. Biol. 13, 1433–1439.
Benoist P., Mas J. A., Marco R. and Cervera M. 1998 Differential muscle-type expression of the Drosophila troponin T gene. A 3-base pair microexon is involved in visceral and adult hypodermic muscle specification. J. Biol. Chem. 273, 7538–7546.
Bullard B. and Pastore A. 2011 Regulating the contraction of insect flight muscle. J. Muscle Res. Cell Motil. 32, 303–313.
De Nicola G., Burkart C., Qiu F., Agianian B., Labeit S., Martin S. et al. 2007 The structure of Lethocerus troponin C: insights into the mechanism of stretch activation in muscles. Structure 15, 813–824.
Dietzl G., Chen D., Schnorrer F., Su K. C., Barinova Y., Fellner M. et al. 2007 A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, 151–156.
Drummond D. R., Hennessey E. S. and Sparrow J. C. 1991 Characterization of missense mutations in the Act88F gene of Drosophila melanogaster. Mol. Gen. Genet. 226, 70–80.
Farah C. S. and Reinach F. C. 1995 The troponin complex and regulation of muscle contraction. FASEB J. 9, 755–767.
Fyrberg C., Ketchum A., Ball E. and Fyrberg E. 1998 Characterization of lethal Drosophila melanogaster alpha-actinin mutants. Biochem. Genet. 36, 299–310.
Gordon A. M., Homsher E. and Regnier M. 2000 Regulation of contraction in striated muscle. Physiol. Rev. 80, 853–924.
Herranz R., Mateos J. and Marco R. 2005a Diversification and independent evolution of troponin C genes in insects. J. Mol. Evol. 60, 31–44.
Herranz R., Mateos J., Mas J. A., Garcia-Zaragoza E., Cervera M. and Marco R. 2005b The co-evolution of insect muscle TpnT and TpnI gene isoforms. Mol. Biol. Evol. 22, 2231–2242.
Johnston J. J., Kelley R. I., Crawford T. O., Morton D. H., Agarwala R., Koch T. et al. 2000 A novel nemaline myopathy in the Amish caused by a mutation in troponin T1. Am. J. Hum. Genet. 67, 814–821.
Krzic U., Rybin V., Leonard K. R., Linke W. A. and Bullard B. 2010 Regulation of oscillatory contraction in insect flight muscle by troponin. J. Mol. Biol. 397, 110–118.
Linari M., Reedy M. K., Reedy M. C., Lombardi V. and Piazzesi G. 2004 Ca-activation and stretch-activation in insect flight muscle. Biophys. J. 87, 1101–1111.
Marden J. H. 2006 Functional and ecological effects of isoform variation in insect flight muscle. In: Nature’s versatile engine: insect flight muscle inside and out (ed) J. O. Vigoreaux, pp. 214–220. Landes Bioscience/Eurekah.com. Texas, USA.
Marin M. C., Rodriguez J. R. and Ferrus A. 2004 Transcription of Drosophila Troponin I gene is regulated by two conserved, functionally identical, synergistic elements. Mol. Biol. Cell. 15, 1185–1196.
Marston S. B. and Redwood C. S. 2003 Modulation of thin filament activation by breakdown or isoform switching of thin filament proteins. Circ. Res. 93, 1170–1178.
Martin S. R., Avella G., Adrover M, de Nicola G. F., Bullard B. and Pastore A. 2011 Binding properties of the calcium-activated F2 isoform of Lethocerus troponin C. Biochemistry 50, 1839–1847.
Mas J. A., Garcia-Zaragoza E. and Cervera M. 2004 Two functionally identical modular enhancers in Drosophila troponin T gene establish the correct protein levels in different muscle types. Mol. Biol. Cell. 15, 1931–1945.
McArdle K., Allen T. S. and Bucher E. A. 1998 Ca2+-dependent muscle dysfunction caused by mutation of the Caenorhabditis elegans troponin T-1 gene. J. Cell Biol. 143, 1201–1213.
Miller R. C., Schaaf R., Maughan D. W. and Tansey T. R. 1993 A non-flight muscle isoform of Drosophila tropomyosin rescues an indirect flight muscle tropomyosin mutant. J. Muscle Res. Cell. Motil. 14, 85–98.
Moore J. R. 2006 Stretch activation: towards a molecular mechanism. In Nature’s versatile engine: insect flight muscle inside and out (ed) J. O. Vigoreaux, pp. 44–60. Landes Bioscience/Eurekah.com.
Morimoto S., Lu Q. W., Harada K., Takahashi-Yanaga F., Minakami R. et al. 2002 Ca2+ desensitizing effect of a deletion mutation Delta K210 in cardiac troponin T that causes familial dilated cardiomyopathy. Proc. Natl. Acad. Sci. USA 99, 913–918.
Myers C. D., Goh P. Y., Allen T. S., Bucher E. A. and Bogaert T. 1996 Developmental genetic analysis of troponin T mutations in striated and non striated muscle cells of Caenorhabditis elegans. J. Cell Biol. 132, 1061–1077.
Nongthomba U. and Ramachandra N. B. 1999 A direct screen identifies new flight muscle mutants on the Drosophila second chromosome. Genetics 153, 261–274.
Nongthomba U., Ansari M., Thimmaiya D., Stark M. and Sparrow J. C. 2007 Aberrant splicing of an alternative exon in the Drosophila troponin-T gene affects flight muscle development. Genetics 177, 295–306.
Nongthomba U., Cummins M., Clark S., Vigoreaux J. O. and Sparrow J. C. 2003 Suppression of muscle hypercontraction by mutations in the myosin heavy chain gene of Drosophila melanogaster. Genetics 164, 209–222.
Nongthomba U., Clark S., Cummins M., Ansari M., Stark M. and Sparrow J. C. 2004 Troponin I is required for myofibrillogenesis and sarcomere formation in Drosophila flight muscle. J. Cell. Sci. 117, 1795–1805.
O’Kane C. J. and Gehring W. J. 1987 Detection in situ of genomic regulatory elements in Drosophila. Proc. Natl. Acad. Sci. USA 84, 9123–9127.
Ohte N., Miyoshi I., Sane D. C. and Little W. C. 2009 Zebrafish with antisense-knockdown of cardiac troponin C as a model of hereditary dilated cardiomyopathy. Circ. J. 73, 1595–1596.
Orfanos Z. and Sparrow J. C. 2013 Myosin isoform switching during assembly of Drosophila flight muscle thick filament lattice. J. Cell Sci. 126, 139–148.
Peckham M., Molloy J. E., Sparrow J. C. and White D. C. 1990 Physiological properties of the dorsal longitudinal flight and the tergal depressor of the trochanter muscle of Drosophila melanogaster. J. Muscle Res. Cell Motil. 11, 203–215.
Perry S. V. 1998 Troponin T: genetics, properties and function. J. Muscle Res. Cell Motil. 19, 575–602.
Potter J. D. and Gergely J. 1975 The calcium and magnesium binding sites on troponin and their role in the regulation ofmyofibrillar adenosine triphosphate. J. Biol. Chem. 250, 4628–4633.
Qiu F., Lakey A., Agianian B., Hutchings A., Butcher G. W., Labeit S. et al. 2003 Troponin C in different insect muscle types: identification of two isoforms in Lethocerus, Drosophila and Anopheles that are specific to asynchronous flight muscle in the adult insect. Biochem. J. 371, 811–821.
Rai M. and Nongthomba U. 2013 Effect of myonuclear number and mitochondrial fusion on Drosophila indirect flight muscle organization and size. Exp. Cell Res. 319, 2566–2577.
Roberts R. and Sigwart U. 2001 New concepts in hypertrophic cardiomyopathies, part I. Circulation 104, 2113–2116.
Sahota V. K., Grau B. F., Mansilla A. and Ferrús A. 2009 Troponin I and Tropomyosin regulate chromosomal stability and cell polarity. J. Cell Sci. 122, 2623–2631.
Sehnert A. J., Huq A., Weinstein B. M., Walker C., Fishman M. and Stainier D. Y. 2002 Cardiac troponin T is essential in sarcomere assembly and cardiac contractility. Nat. Genet. 31, 106–110.
Swank D. M., Knowles A. F., Suggs J. A., Sarsoza F., Lee A., Maughan D. W. et al. 2002 The myosin converter domain modulates muscle performance. Nat. Cell Biol. 4, 312–316.
Towbin J. A. and Bowles N. E. 2002 The failing heart. Nature 415, 227–233.
Vigoreaux J. O. 2006 Molecular basis of muscle structure. In: Muscle development in Drosophila (ed) H. Sink, pp. 143–152. Landes Bioscience/Eurekah.com, Texas.
Wei B. and Jin J. P. 2011 Troponin T isoforms and posttranscriptional modifications: Evolution, regulation and function. Arch. Biochem. Biophys. 505, 144–154.
Wells L., Edwards K. A. and Bernstein S. I. 1996 Myosin heavy chain isoforms regulate muscle function but not myofibril assembly. EMBO J. 15, 4454–4459.
Acknowledgements
We thank our laboratory members for their critical comments. This work was supported by financial assistance from Department of Biotechnology, Department of Science and Technology, Government of India, New Delhi and Indian Institute of Science, Bangalore.
Author information
Authors and Affiliations
Corresponding authors
Additional information
[Singh S. H., Kumar P., Ramachandra N. B. and Nongthomba U. 2014 Roles of the troponin isoforms during indirect flight muscle development in Drosophila. J. Genet. 93, xx–xx]
Rights and permissions
About this article
Cite this article
SINGH, S.H., KUMAR, P., RAMACHANDRA, N.B. et al. Roles of the troponin isoforms during indirect flight muscle development in Drosophila . J Genet 93, 379–388 (2014). https://doi.org/10.1007/s12041-014-0386-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-014-0386-8