Abstract
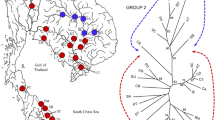
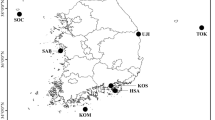
A total of 19 polymorphic microsatellite loci were used to analyse levels of genetic variation for 10 populations of Perna viridis L. collected from all over peninsular Malaysia. The populations involved in this study included Pulau Aman in Penang, Tanjung Rhu in Kedah, Bagan Tiang in Perak, Pulau Ketam in Selangor, Muar, Parit Jawa, Pantai Lido and Kampung Pasir Puteh in Johore, and Kuala Pontian and Nenasi in Pahang state. The number of alleles per locus ranged from two to seven, with an average of 3.1. Heterozygote deficiencies were observed across all the 10 populations. Characterization of the populations revealed that local populations of P. viridis in peninsular Malaysia were genetically similar enough to be used as a biomonitoring agent for heavy metal contamination in the Straits of Malacca. Cluster analysis grouped the P. viridis populations according to their geographical distributions with the exception of Parit Jawa. The analysis also revealed that P. viridis from the northern parts of peninsular Malaysia were found to be the most distant populations among the populations of mussels investigated and P. viridis from the eastern part of peninsular Malaysia were closer to the central and southern populations than to the northern populations.
Similar content being viewed by others
References
Arnaud-Haond S., Vonau V., Bonhomme F., Boudry P., Prou J., Seaman T. et al. 2003 Spat collection of the pearl oyster (Pinctada margaritifera cumingii) in French Polynesia: an evaluation of the potential impact on genetic variability of wild and farmed populations after 20 years of commercial exploitation. Aquaculture 219, 181–192.
Callen D. F., Thompson A. D., Shen Y., Phillips H. A., Richards R. I., Mulley J. C. and Sutherland G. R. 1993 Incidence and origin of ‘null’ alleles in the (AC)n microsatellite markers. Am. J. Hum. Genet. 52, 922–927.
Chua B. H., Yong S. Y., Teh C. H., Yap C. K., Tan S. G. and Ismail A. 2003 Assessment of the population genetic structure of the green-lipped mussel, Perna viridis L. using random amplified polymorphic DNA (RAPD) and random amplified microsatellites (RAMs) markers. In From peas to chips: the globalization of genetics (ed. M. K. Thong), pp. 47–48. The Genetic Society of Malaysia, Bangi.
Gosling E., Astanei I. and Was A. 2008 Genetic variability in Irish populations of the invasive zebra mussel, Dreissena polymorpha: discordant estimates of population differentiation from allozymes and microsatellites. Freshwater Biol. 53, 1303–1315.
Gyllensten U. and Ryman N. 1985 Pollution biomonitoring programs and the genetic structure of indicator species. AMBIO 14, 29–31.
Hare M. P., Karl S. A. and Avise J. C. 1996 Anonymous nuclear DNA markers in the American oyster and their implications for the heterozygote deficiency phenomenon in marine bivalves. Mol. Biol. Evol. 13, 334–345.
Holland B. S. 2001 Invasion without a bottleneck: microsatellite variation in natural and invasive populations of the brown mussel Perna perna (L). Marine Biotechnol. 3, 407–415.
Ismail A., Yap C. K., Zakaria P. M., Tanabe S., Takada H. and Ismail A. R. 2000 Green-lipped mussel Perna viridis (L.) as a biomonitoring agent for heavy metals in the west coast of Peninsular Malaysia. In Towards sustainable management of the straits of malacca (ed. M. Shariff et al.), pp. 553–559. Universiti Putra Malaysia Press, Serdang.
Johnson R. L., Liang Q. L. Milan C. D. and Farris J. L. 1998 Genetic diversity and cellulolytic activity among several species of unionid bivalves in Arkansas. J. Shellfish Res. 17, 1375–1382.
Kalinowski S. T. and Taper M. L. 2006 Maximum likelihood estimation of the frequency of null alleles at microsatellite loci. Conserv. Genet. 7, 991–995.
Kamara D., Gyenai K.B., Geng T., Hammade H. and Smith E. J. 2007 Microsatellite marker-based genetic analysis of relatedness between commercial and heritage turkeys (Meleagris gallopavo). Poultry Sci. 86, 46–49.
Kimura M. and Crow J. F. 1964 The number of alleles that can be maintained in a finite population. Genetics 49, 725–738.
Lowe A., Harris S. and Ashton P. 2004 Ecological genetics: design, analysis and application. Blackwell, Malden.
Monirith I., Ueno D., Takahashi S., Nakata H., Sudaryanto A., Subramanian A. and Karuppiah S. 2003 Asia-Pacific mussel watch: monitoring contamination of persistent organochlorine compounds in coastal waters of Asian countries. Mar. Poll. Bull. 46, 281–300.
Nei M. 1978 Estimation of heterozygosity and genetic distance from a small number of individuals. Genetics 89, 583–590.
Nicholson S. and Lam P. K. S. 2005 Pollution monitoring in Southeast Asia using biomarkers in the Mytilid mussel Perna viridis (Mytilidae: Bivalvia). Environ. Int. 31, 121–132.
Ochsenreither S., Kuhls K., Schaars M., Presber W. and Schonian G. 2006 Multilocus microsatellite typing as a new tool for discrimination of Leishmania infantum MON-1 strains. J. Clin. Microbiol. 44, 495–503.
O’Connell M. and Wright J. M. 1997 Microsatellite DNA in fishes. Rev. Fish Biol. Fish. 7, 331–363.
Ong L. C. C., Tan S. G., Yusof K. and Yap C. K. 2005 Isolation of DNA microsatellite markers in the green-lipped mussel, Perna viridis. Pertanika J. Trop. Agric. Sci. 28, 79–85.
Ong C. C., Teh C. H., Tan S. G., Yusoff K. and Yap C. K. 2008 Eleven novel polymorphic microsatellite DNA markers from the green-lipped mussel Perna viridis. Russ. J. Genet. 44, 498–500.
Rainbow P. S. 1995 Biomonitoring of heavy metal availability in the marine environment. Mar. Poll. Bull. 31, 183–192.
Rohlf F. J. 1989 NTSYS-pc. Numerical taxonomy and multivariate analysis system, version 1.60.
Swofford D. L. and Selander R. B. 1989 BIOSYS-1: a computer program for the analysis of allelic variation in population genetics and biochemical systematic (Release 1.7). Illinois Natural History Survey, Champaign.
Winnepennincks B., Backeljau T. and Wachter R. D. 1993 Extraction of high molecular weight DNA from mollusks. Trends Genet. 9, 407.
Wright S. 1978 Variability within and among natural populations, vol. 4, University of Chicago Press, Chicago.
Yap C. K., Tan S. G., Ismail A. and Omar H. 2002 Genetic variation of the green-lipped mussel Perna viridis (Linnaeus) from the west coast of Peninsular Malaysia. Zool. Studies 241, 376–387.
Yap C. K., Tan S. G., Ismail A. and Omar H. 2004 Allozyme polymorphisms and heavy metal levels in the green-lipped mussel Perna viridis (Linnaeus) collected from contaminated and uncontaminated sites in Malaysia. Environ. Int. 30, 39–46.
Yeh F. C. and Boyle T. H. B. 1997 Population genetic analysis of codominant and dominant markers and quantitative traits. Belgian J. Bot. 129, 157.
Zouros E. and Foltz D. W. 1984 Possible explanation of heterozygote deficiency in bivalve molluscs. Malacologia 25, 583–591.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ong, C.C., Yusoff, K., Yap, C.K. et al. Genetic characterization of Perna viridis L. in peninsular Malaysia using microsatellite markers. J Genet 88, 153–163 (2009). https://doi.org/10.1007/s12041-009-0023-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-009-0023-0