Abstract
We explore herein the interconnection between the collective intermolecular solvent modes (CIM) and ultrafast reaction rate, assuming that frequency-dependent solvent friction controls the rate of such reactions. We attempt to find a possible explanation for the observed near-insensitivity of ultrafast reaction rates (for example, charge transfer reaction) to the medium viscosity. Results are presented here by employing an analytical scheme that estimates the high-frequency solvent frictional response. Representative room temperature reaction media considered here are an ionic liquid (BMIMPF6, \(\eta \sim 310\mathrm{ cP}\)), a dipolar solvent (ethanol, \(\eta \sim 1.09\mathrm{ cP}\)) and a deep eutectic solvent (Acetamide+ LiBr, \(\eta \sim 1950\mathrm{ cP}\)). It is found that the wavenumber and frequency-dependent rotational friction, \({\Gamma }_{R}(\kappa ,z)\), estimated by using the available experimental dielectric relaxation (DR) data for the ionic liquid and the deep eutectic solvent (DES), cannot predict the viscosity independence of \({\Gamma }_{R}(\kappa ,z)\) at high frequency. Missing dispersion in the DR data of the DES appears to be critical and incorporation of this missing amplitude via collective solvent intermolecular modes centered around 100 cm-1 markedly improves the high-frequency behaviour of \({\Gamma }_{R}\left(\kappa ,z\right).\) Subsequently, the calculated \({\Gamma }_{R}(\kappa ,z)\) for these solvents at high frequency exhibits near-insensitivity to medium viscosity and explain the viscosity independence of ultrafast reaction rates.
Graphical abstract
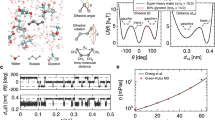
Comparison of the frequency dependent rotational friction among three different solvent systems considered – normal liquid (ethanol), deep eutectic solvent (acetamide/LiBr) and ionic liquid ([BMIM][BF6]). Note the high frequency friction values for these solvents are very similar, although their zero frequency values differ quite considerably because of widely different viscosities.




Similar content being viewed by others
References
Hynes J T 1986 Chemical reaction rates and solvent friction J. Stat. Phys. 42 149
Bagchi B 2012 Molecular Relaxation in Liquids (USA: OUP) p. 312
Straub J E, Borkovec M and Berne B J 1986 Non-Markovian activated rate processes: comparison of current theories with numerical simulation data J. Chem. Phys. 84 1788
Carmeli B and Nitzan A 1983 Non-Markovian theory of activated rate processes. I. Formalism J. Chem. Phys. 79 393
Chavanis P H 2019 The generalized stochastic Smoluchowski equation Entropy 21 1006
Smoluchowski M V 1916 Über Brownsche Molekularbewegung Unter Einwirkung Äußerer Kräfte Und Deren Zusammenhang Mit Der Verallgemeinerten Diffusionsgleichung Ann. Phys. 353 1103
Li X, Liang M, Chakraborty A, Kondo M and Maroncelli M 2011 Solvent-controlled intramolecular electron transfer in ionic liquids J. Phys. Chem. B 115 6592
Lynden-Bell R 2007 Does Marcus theory apply to redox processes in ionic liquids? A simulation study Electrochem. Commun. 9 1857
Shim Y and Kim H J 2007 Free energy and dynamics of electron-transfer reactions in a room temperature ionic liquid J. Phys. Chem. B 111 4510
Bagchi B and Biswas R 1999 Polar and nonpolar solvation dynamics, ion diffusion, and vibrational relaxation: Role of biphasic solvent response in chemical dynamics Adv. Chem. Phys. 109 207
Bagchi B and Biswas R 1998 Ionic mobility and ultrafast solvation: control of a slow phenomenon by fast dynamics Acc. Chem. Res. 31 181
Daschakraborty S and Biswas R 2012 Ultrafast solvation response in room temperature ionic liquids: Possible origin and importance of the collective and the nearest neighbour solvent modes J. Chem. Phys. 137 114501
Daschakraborty S, Pal T and Biswas R 2013 Stokes shift dynamics of ionic liquids: Solute probe dependence, and effects of self-motion, dielectric relaxation frequency window, and collective intermolecular solvent modes J. Chem. Phys. 139 164503
Kashyap H K and Biswas R 2008 Dipolar solvation dynamics in room temperature ionic liquids: An effective medium calculation using dielectric relaxation data J. Phys. Chem. B 112 12431
González B, Calvar N, Gómez E and Domínguez Á 2007 Density, dynamic viscosity, and derived properties of binary mixtures of methanol or ethanol with water, ethyl acetate, and methyl acetate at T=(293.15, 298.15, and 303.15)K J. Chem. Thermodyn. 39 1578
Mukherjee K, Das A, Choudhury S, Barman A and Biswas R 2015 Dielectric relaxations of (acetamide + electrolyte) deep eutectic solvents in the frequency window, 0.2 ≤ ν/GHz ≤ 50: Anion and cation dependence J. Phys. Chem. B 119 8063
Roy S and Bagchi B 1994 Solvation dynamics, energy distribution and trapping of a light solute ion Chem. Phys. 183 207
Roy S and Bagchi B 1993 Solvation dynamics in liquid water. A novel interplay between librational and diffusive modes J. Chem. Phys. 99 9938
Roy S and Bagchi B 1993 Ultrafast underdamped solvation: Agreement between computer simulation and various theories of solvation dynamics J. Chem. Phys. 99 1310
Bagchi B 1989 Dynamics of solvation and charge transfer reactions in dipolar liquids Annu. Rev. Phys. Chem. 40 115
Bagchi B and Chandra A 1991 Collective orientational relaxation in dense dipolar liquids Adv. Chem. Phys. 80 1
Waisman E and Lebowitz J L 1972 Mean spherical model integral equation for charged hard spheres. II. Results J. Chem. Phys. 56 3093
Waisman E and Lebowitz J L 1972 Mean spherical model integral equation for charged hard spheres I. Method of solution J. Chem. Phys. 56 3086
Stoppa A, Hunger J, Buchner R, Hefter G, Thoman A and Helm H 2008 Interactions and dynamics in ionic liquids J. Phys. Chem. B 112 4854
Kindt J T and Schmuttenmaer C A 1996 Far-infrared dielectric properties of polar liquids probed by femtosecond terahertz pulse spectroscopy J. Phys. Chem. 100 10373
Biswas R and Bagchi B 1996 Solvation dynamics in slow, viscous liquids: Application to amides J. Phys. Chem. 100 1238
Nandi N, Roy S and Bagchi B 1995 Ultrafast solvation dynamics in water: Isotope effects and comparison with experimental results J. Chem. Phys. 102 1390
Kashyap H K and Biswas R 2010 Stokes shift dynamics in ionic liquids: Temperature dependence J. Phys. Chem. B 114 16811
Chakraborty A, Inagaki T, Banno M, Mochida T and Tominaga K 2011 Low-frequency spectra of metallocenium ionic liquids studied by terahertz time-domain spectroscopy J. Phys. Chem. A 115 1313
Giraud G, Gordon C M, Dunkin I R and Wynne K 2003 The effects of anion and cation substitution on the ultrafast solvent dynamics of ionic liquids: A time-resolved optical Kerr-effect spectroscopic study J. Chem. Phys. 119 464
Shirota H and Castner E W 2005 Physical properties and intermolecular dynamics of an ionic liquid compared with its isoelectronic neutral binary solution J. Phys. Chem. A 109 9388
Shirota H and Castner E W 2005 Why Are viscosities lower for ionic liquids with −CH2Si(CH3)3 vs −CH2C(CH3)3 substitutions on the imidazolium cations? J. Phys. Chem. B 109 21576
Shirota H, Nishikawa K and Ishida T 2009 Atom substitution effects of [XF6]− in ionic liquids. 1. Experimental study J. Phys. Chem. B 113 9831
Fujisawa T, Nishikawa K and Shirota H 2009 Comparison of interionic/intermolecular vibrational dynamics between ionic liquids and concentrated electrolyte solutions J. Chem. Phys. 131 244519
Turton D A, Hunger J, Stoppa A, Hefter G, Thoman A, Walther M, et al. 2009 Dynamics of imidazolium ionic liquids from a combined dielectric relaxation and optical Kerr effect study: Evidence for mesoscopic aggregation J. Am. Chem. Soc. 131 11140
Shim Y and Kim H J 2008 Dielectric relaxation, ion conductivity, solvent rotation, and solvation dynamics in a room-temperature ionic liquid J. Phys. Chem. B 112 11028
Schröder C and Steinhauser O 2009 On the dielectric conductivity of molecular ionic liquids J. Chem. Phys. 131 114504
Schröder C, Hunger J, Stoppa A, Buchner R and Steinhauser O 2008 On the collective network of ionic liquid/water mixtures. II. Decomposition and interpretation of dielectric spectra J. Chem. Phys. 129 18450
Biswas R, Das A and Shirota H 2014 Low-frequency collective dynamics in deep eutectic solvents of acetamide and electrolytes: A femtosecond Raman-induced Kerr effect spectroscopic study J. Chem. Phys. 141 134506
Das S, Biswas R and Mukherjee B 2015 Orientational jumps in (acetamide+ electrolyte) deep eutectics: Anion dependence J. Phys. Chem. B 119 11157
Pal T and Biswas R 2013 Rank-dependent orientational relaxation in an ionic liquid: An all-atom simulation study Theor. Chem. Acc. 132 1348
Saiz L, Padró J A and Guàrdia E 1997 Structure and dynamics of liquid ethanol J. Phys. Chem. B 101 78
Matyushov D V 2004 Solvent reorganization energy of electron-transfer reactions in polar solvents J. Chem. Phys. 120 7532
Marcus R A 1993 Electron transfer reactions in chemistry. Theory and experiment Rev. Mod. Phys. 65 599
Marcus R A 1956 On the theory of oxidation-reduction reactions involving electron transfer. I J. Chem. Phys. 24 966
Biswas R, Lewis J E and Maroncelli M 1999 Electronic spectral shifts, reorganization energies, and local density augmentation of Coumarin 153 in supercritical solvents Chem. Phys. Lett. 310 485
Horng M L, Gardecki J A, Papazyanand A and Maroncelli M 1995 Subpicosecond measurements of polar solvation dynamics: Coumarin 153 revisited J. Phys. Chem. 99 17311
Zhang X X, Liang M, Hunger J, Buchner R and Maroncelli M 2013 Dielectric relaxation and solvation dynamics in a prototypical ionic liquid + dipolar protic liquid mixture: 1-Butyl-3-methylimidazolium tetrafluoroborate + water J. Phys. Chem. B 117 15356
Guchhait B, Das S, Daschakraborty S and Biswas R 2014 Interaction and dynamics of (alkylamide + electrolyte) deep eutectics: Dependence on alkyl chain-length, temperature, and anion identity J. Chem. Phys. 140 104514
Subba N, Tarif E, Sen P and Biswas R 2020 Subpicosecond solvation response and partial viscosity decoupling of solute diffusion in ionic acetamide deep eutectic solvents: Fluorescence up-conversion and fluorescence correlation spectroscopic measurements J. Phys. Chem. B 124 1995
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
BAKSI, A., BISWAS, R. Why do some reactions possess similar reaction rate in wildly different viscous media? A possible explanation via frequency-dependent friction. J Chem Sci 134, 51 (2022). https://doi.org/10.1007/s12039-022-02045-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-022-02045-1