Abstract
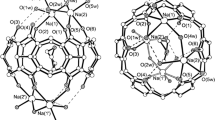
In this study, cucurbit[8]uril (Q[8]) was used as a carrier for Sulfanilamide (G1) and sulfamethoxazole (G2), and the crystals of their inclusion complexes were cultured in a 3M HCl aqueous solution upon the addition of ZnCl2 as an inducer. The crystal structure was characterized using single-crystal X-ray diffraction. The results showed that two new supramolecular self-assemblies were constructed and the main driving forces in the system were hydrogen bond and ion–dipole interactions. In addition, the effects of Q[8] on the solubility and cumulative release rate in vitro of G1 and G2 were investigated by UV Vis spectroscopy. The results showed that the intervention of Q[8] had no effect on the solubility of G1 and G2; the cumulative release rates of G1 and G2 in artificial gastrointestinal juice were reduced and had a certain sustained-release effect.
Graphical abstract

In this study, we reported that Cucurbit[8]uril (Q[8]) was used as the carrier of P-aminobenzenesulfonamide and sulfamethoxazole. The experimental results show that the main driving forces of the system are hydrogen bond and ion-dipole interaction and two new supramolecular self-assemblers were constructed.








Similar content being viewed by others
Supplementary Information (SI)
The X-ray crystallographic data for structures reported in this study have been deposited in the Cambridge Crystallographic Data Center under accession numbers CCDC: 2071851 (1) and 2039124 (2). These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/data_request/cif.
References
Ogoshi T, Yamagishi T and Nakamoto Y 2016 Pillar-shaped macrocyclic hosts pillar[n]arenes: new key players for supramolecular chemistry Chem. Rev. 116 7937
Yang W, Greenaway A and Lin X 2010 Exceptional thermal stability in a supramolecular organic framework: porosity and gas storage J. Am. Chem. Soc. 132 14457
Tian J, Wang H and Zhang D W 2017 Supramolecular organic frameworks (SOFs): homogeneous regular 2D and 3D pores in water Natl. Sci. Rev. 4 426
Lagona J, Mukhopadhyay P and Chakrabarti S 2005 The cucurbit[n]uril family Angew. Chem. Int. Ed. 44 4844
Lee J W, Samal S and Selvapalam N 2003 Cucurbituril homologues and derivatives: new opportunities in supramolecular chemistry Acc. Chem Res. 36 621
Gerasko O A, Samsonenko D G and Fedin V P 2002 Supramolecular chemistry of cucurbiturils Russian Chem Rev. 71 741
Elemans J A A W, Rowan A E and Nolte R J M 2000 Self-assembled architectures from glycoluril Ind. Eng. Chem. Res. 39 3419
Cintas P 1994 Cucurbituril: supramolecular perspectives for an old ligand J. Incl. Phenom. Macro. 17 205
Yang R Y, Deng X Y and Huang Y 2020 Recognition of lanthanide metal cations by t-DSMI@Alkyl-substituted cucurbit[6]uril probes Chem. Select 5 8649
Lin R L, Li R and Shi H 2020 Symmetrical-tetramethyl-cucurbit[6]uril-driven movement of cucurbit[7]uril gives rise to heterowheel [4]pseudorotaxanes J. Org. Chem. 85 3568
Jansen K, Buschmann H J and Wego A 2001 Cucurbit[5]uril, decamethylcucurbit[5]uril and cucurbit[6]uril. Synthesis, solubility and amine complex formation J. Incl. Phenom. Macro. 39 357
Freeman W A, Mock W L and Shih N Y 1981 Cucurbituril J. Am. Chem. Soc. 103 7367
Xu H P, Lin D and Zhang B 2020 Supramolecular self-assembly of a hybrid “hyalurosome” for targeted photothermal therapy in non-small cell lung cancer Drug Deliv. 27 378
Day A I, Blanch R J and Arnold A P 2002 A cucurbituril-based gyroscane: a new supramolecular form Angew. Chem. Int. Ed. 41 275
Kim J, Jung I S and Kim S Y 2000 New cucurbituril homologues: syntheses, isolation, characterization, and X-ray crystal structures of cucurbit[n]uril (n = 5, 7, and 8) J. Am. Chem. Soc. 122 540
Lutz F, Parodi N L and Schmidt T C 2021 Heteroternary cucurbit[8]uril complexes as supramolecular scaffolds for self-assembled bifunctional photoredoxcatalysts Chem. Commun. 57 2887
Behrend R, Meyer F and Rusche F 1905 Ueber condensationsproducte aus glycoluril und formaldehyd Just Liebigs Ann. Chem. 339 1
Kim K, Selvapalam N and Oh D H 2004 Cucurbiturils a new family of host molecules J. Incl. Phenom. Macro. 50 31
Day A, Arnold A P and Blanch R J 2001 Controlling factors in the synthesis of cucurbituril and its homologues J. Org. Chem. 66 8094
Liu S, Zavalij P Y and Isaacs L 2005 Cucurbit[10]uril J. Am. Chem. Soc. 127 16798
Cheng X J, Liang L L and Chen K 2013 Twisted cucurbit[14]uril Angew. Chem. Int. Edit. 52 7252
Cao X H, Gao A P and Hou J T 2021 Fluorescent supramolecular self-assembly gels and their application as sensors: a review Coord. Chem. Rev. 434 213792
Zheng J, Zhao W W and Meng Y 2021 Supramolecular self-assembly of cyclopentyl-substituted cucurbit[n ]uril with Fe 3+, Fe 2+, and HClO 4 based on outer surface interaction cryst Res. Technol. 56 2000183
Lewis J and Wilkins R G 1951 Coordination chemistry Nature 167 434
Hahn F E and Mareike C 2008 Heterocyclic carbenes: synthesis and coordination chemistry Angew. Chem. Int. Edit. 47 3122
Ni X L, Xue S F and Tao Z 2015 Advances in the lanthanide metallosupramolecular chemistry of the cucurbit[n]urils Coord. Chem. Rev. 287 89
Xie J, Zeng Z S and Tao Z 2020 Study on the interaction and properties of cucurbit[8]uril with oroxin B Chem. Res. Chin. U. 36 804
Dwi H, Fuad A K M and Khairul A 2021 Assessing encapsulation of curcumin in cocoliposome: in vitro study Open Chem. 19 358
Hettiarachchi G, Nguyen D and Wu J 2010 Toxicology and drug delivery by cucurbit[n]uril type molecular containers PLOS ONE 5 e10514
Cao L P, Hettiarachchi G and Briken V 2013 Cucurbit[7]uril containers for targeted delivery of oxaliplatin to cancer cells Angew. Chem. Int. Edit. 52 12033
Mccammon J A 1998 Theory of biomolecular recognition Curr. Opin. Struct. Bio. 8 245
Nguyen C N, Young T K and Gilson M K 2012 Grid inhomogeneous solvation theory: hydration structure and thermodynamics of the miniature receptor cucurbit[7]uril J. Chem. Phys. 137 044101
Ganapati S and Isaacs L 2018 Acyclic cucurbit[n]uril-type receptors: preparation, molecular recognition properties and biological applications Isr. J. Chem. 58 250
Andrea S, Takashi O and Antonio M 2003 Anticancer and antiviral sulfonamides Curr. Med. Chem. 10 925
Khan F A, Mushtaq S and Naz S 2018 Sulfonamides as potential bioactive scaffolds Curr. Org. Chem. 22 818
Zhang L, Johnson N W and Liu Y 2021 Biodegradation mechanisms of sulfonamides by phanerochaete chrysosporium – luffa fiber system revealed at the transcriptome level Chemosphere 266 129124
Buschmann H J, Cleve E and Jansen K 2001 Complex formation between cucurbit[n]urils and alkali, alkaline earth and ammonium ions in aqueous solution J. Incl. Phenom. Macro. 40 117
Zhao Y J, Bali M S and Cullinane C 2009 Synthesis, cytotoxicity and cucurbituril binding of triamine linked dinuclear platinum complexes Dalton Trans. 26 5190
Huang J J, Xu Z L and Lian X W 2018 The host-guest interaction between Q[8] and baicalin, its effect on the properties of baicalin Chem. J. Chin. U. 39 2425
Zeng Z S, Xie J, Luo G Y, Tao Z and Zhang Q 2020 Host–guest interaction of cucurbit[8]uril with oroxin A and its effect on the properties of oroxin A Beilstein J. Org. Chem. 16 2332
Xu Z L, Lian X W and Li M J 2017 Effects of inclusion of chrysin in cucurbit[8]uril on its stability, solubility and antioxidant potential Chem. Res. Chin. U. 33 736
Mehta S K, Jindal N and Kaur G 2011 Quantitative investigation, stability and in vitro release studies of anti-TB drugs in triton niosomes Colloid Surf. B 87 173
Acknowledgements
The authors acknowledge the support from the Science and Technology Support Plan of Guizhou Province [Guizhou Science and Technology Cooperation Support(2020) 4Y218].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, L., Zheng, J., Zeng, Z. et al. Supramolecular self-assembly based on Cucurbit[8]urils with sulfanilamide and sulfamethoxazole. J Chem Sci 134, 26 (2022). https://doi.org/10.1007/s12039-021-02017-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-021-02017-x