Abstract
The type and magnitude of interactions between diisobutyl ketone (DIBK) and a homologous series of 2-alkanols (2-propanol up to 2-pentanol) are studied in this paper using density and viscosity data. Studies have been carried out in a wide range of temperature (293.15 K to 323.15 K), atmospheric pressure, and in the entire range of composition. The calculated excess molar volumes of the above-mentioned systems are positive, and the viscosity deviations are negative. For the correlation of the density values of binary systems, the statistical association fluid theory (SAFT) and perturbed chain statistical association fluid theory (PC-SAFT) have been used, and the output of two models are compared by average absolute deviation. The results show that the PC-SAFT model is more efficient. Densities and viscosities reported for binary systems along with applied models are all novels, and so far no reports have been investigated in the scientific papers.
Graphic abstract
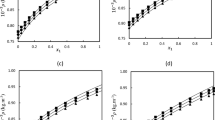
PC-SAFT and SAFT models were applied to model the densities of binary mixtures containing Diisobutyl ketone and 2-alkanol at various temperatures. Results show that models provide satisfactory outcomes and PC-SAFT model produces better results.





Similar content being viewed by others
References
Papa J 2005 Ketones In Ullmann’s Encyclopedia of Industrial Chemistry (Weinheim: Wiley-VCH)
Almasi M and Daneshi R 2018 Investigation of Molecular Interactions in Binary Mixtures of n-Butyl Acetate and (C6 – C10) 1-Alkanol: PC-SAFT Model J. Chem. Eng. Data 63 3881
Moftakhar M and Almasi M 2017 Studies on physicochemical behavior of binary mixtures containing propanal and Alkan-2-ol J. Chem. Thermodyn. 113 315
Ozmen D 2006 (Liquid + liquid) equilibria of (water + propionic acid + methyl isoamyl ketone or diisobutyl ketone or ethyl isoamyl ketone) at \(\text{ T } = 298.2\text{ K }\) Fluid Phase Equilib. 250 70
Stroscs F H, Gablea M and Rounds G C 1947 The Vapor Pressures and Some Other Properties of Di-&butyl Ketone and Di-ibutylcarbinol J. Am. Chem. Soc. 69 1629
Sellers P 1970 Enthalpies of Formation of Some Aliphatic Branched Ketones J. Chem. Thermodyn. 2 211
Pang F, Seng C, Teng T and Ibrahim M H 2007 Densities and viscosities of aqueous solutions of 1-propanol and 2-propanol at temperatures from 293.15 K to 333.15 K J. Mol. Liq. 136 71
Spasojevic V D, Djordjevic B D, Serbanovic S P, Radovic I R and Kijevcanin M L 2014 Densities, Refractive Indices, Viscosities, and Spectroscopic Study of 1-Amino-2-propanol + 1-Butanol and + 2-Butanol Solutions at (288.15 to 333.15) K J. Chem. Eng. Data 59 1817
Riggio R, Martinez H E and Solimo H N 1986 Densities, Viscosities, and Refractive Indexes for the Methyl Isobutyl Ketone + Pentanols Systems, Measurements and Correlations J. Chem. Eng. Data 31 235
Redlich O J and Kister A T 1948 Algebraic representation of thermodynamic properties and the classification of solutions Ind. Eng. Chem. 40 345
Lomte S B, Bawa M J, Lande M K and Arbad B R 2009 Densities and Viscosities of Binary Liquid Mixtures of 2-Butanone with Branched Alcohols at (293.15 to 313.15) K J. Chem. Eng. Data 54 127
Huang S H and Radosz M 1993 Equation of state for small, large, Polydisperse and associating molecules: extension to fluid mixtures Ind. Eng. Chem. Res. 32 762
Huang S H and Radosz M 1990 equation of state for small, large, Polydisperse and associating molecules Ind. Eng. Chem. Res. 29 2284
Gross J and Sadowski G 2001 Perturbed-Chain SAFT. An equation of state based on a perturbation theory for chain molecules Ind. Eng. Chem. Res. 40 1244
Gross J and Sadowski G 2002 Application of the perturbed-chain SAFT equation of state to associating systems Ind. Eng. Chem. Res. 41 5510
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sarkoohaki, B., Almasi, M. & Karimkhani, M. Theoretical and experimental study of physicochemical behavior of binary mixtures: SAFT and PC-SAFT models. J Chem Sci 131, 53 (2019). https://doi.org/10.1007/s12039-019-1630-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-019-1630-9