Abstract
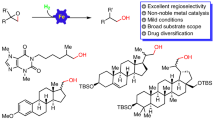
Herein, we describe a practical approach for direct vinylogous aldol condensation of Biginelli product for carbon–carbon double bond formation at methyl group of 6-methyl dihydropirimidinone by using hydrated ferric chloride. With aromatic aldehydes, the method yields stereoselectively trans products. The olefination reaction is applicable with aldehyde without chelating (with Fe III) functional group. DHPMs with carboxylate ester and arylidene at C-6 methyl are not known. The method opens up a new avenue for the synthesis of a wide variety of DHPMs. The olefin products were subjected to dihydroxylation reaction.
Graphical Abstract
SYNOPSIS A convenient and facile C6-olefination at methyl centre of dihydropyrimidinones (DHPMs) has been observed in acetonitrile in the presence of hydrated \(\hbox {FeCl}_{3}\). The method is simple, inexpensive, and stereoselectively trans.





Similar content being viewed by others
References
Terracciano S, Russo A, Chini M G, Vaccaro M C, Potenza M, Vassallo A, Riccio R, Bifulco G and Bruno I 2018 Discovery of new molecular entities able to strongly interfere with Hsp90 C-terminal domain Sci. Rep. 8 Art. No. 1709
Kamal A, Shaheer Malik M, Bajee S, Azeeza S, Faazil S, Ramakrishna S, Naidu V G M and Vishnuwardhan M V P S 2011 Synthesis and biological evaluation of conformationally flexible as well as restricted dimers of monastrol and related dihydropyrimidones Eur. J. Med. Chem. 46 3274
Kim J, Ok T, Park C, So W, Jo M, Kim Y, Seo M, Lee D, Jo S, Ko Y, Choi I, Park Y, Yoon J, Ju M K, Ahn J, Kim J, Han S-J, Kim T-H, Cechetto J, Nam J, Liuzzi M, Sommer P and No Z 2012 A novel 3,4-dihydropyrimidin-2(1H)-one: HIV-1 replication inhibitors with improved metabolic stability Bioorg. Med. Chem. Lett. 22 2522
Chhillar A K, Arya P, Mukherjee C, Kumar P, Yadav Y, Sharma A K, Yadav V, Gupta J, Dabur R, Jha H N, Watterson A C, Parmar V S, Prasad A K and Sharma G L 2006 Microwave-assisted synthesis of antimicrobial dihydropyridines and tetrahydropyrimidin-2-ones: Novel compounds against aspergillosis Biorg. Med. Chem. 14 973
Chiang A N, Valderramos J-C, Balachandran R, Chovatiya R J, Mead B P, Schneider C, Bell S L, Klein M G, Huryn D M, Chen X S, Day B W, Fidock D A, Wipf P and Brodsky J L 2009 Select pyrimidinones inhibit the propagation of the malarial parasite, Plasmodium falciparum Biorg. Med. Chem. 17 1527
Mokale S N, Shinde S S, Elgire R D, Sangshetti J N and Shinde D B 2010 Synthesis and anti-inflammatory activity of some 3-(4,6-disubtituted-2-thioxo-1,2,3,4-tetrahydropyrimidin-5-yl) propanoic acid derivatives Bioorg. Med. Chem. Lett. 20 4424
Atwal K S, Swanson B N, Unger S E, Floyd D M, Moreland S, Hedberg A and O’Reilly B C 1991 Dihydropyrimidine calcium channel blockers. 3. 3-Carbamoyl-4-aryl-1,2,3,4-tetrahydro-6-methyl-5-pyrimidinecarboxylic acid esters as orally effective antihypertensive agents J. Med. Chem. 34 806
Acharya B N, Rao G B D, Kumar D, Kumar P and Kaushik M P 2015 Design, synthesis, and evaluation of dihydropyrimidinone (DHPM) based muscarinic receptor antagonist Med. Chem. Res. 24 1763
Ahmad S, Iftikhar F, Ullah F, Sadiq A and Rashid U 2016 Rational design and synthesis of dihydropyrimidine based dual binding site acetylcholinesterase inhibitors Bioorg. Chem. 69 91
Lacotte P, Buisson D-A and Ambroise Y 2013 Synthesis, evaluation and absolute configuration assignment of novel dihydropyrimidin-2-ones as picomolar sodium iodide symporter inhibitors Eur. J. Med. Chem. 62 722
Dhumaskar K L, Meena S N, Ghadi S C and Tilve S G 2014 Graphite catalyzed solvent free synthesis of dihydropyrimidin-2(1H)-ones/thiones and their antidiabetic activity Bioorg. Med. Chem. Lett. 24 2897
Lewis R W, Mabry J, Polisar J G, Eagen K P, Ganem B and Hess G P 2010 Dihydropyrimidinone positive modulation of \(\updelta \)-subunit-containing \(\upgamma \)-aminobutyric acid type A receptors, including an epilepsy-linked mutant variant Biochemistry 49 4841
Liu J, Wu F, Chen L, Hu J, Zhao L, Chen C and Peng L 2011 Evaluation of dihydropyrimidin-(2H)-one analogues and rhodanine derivatives as tyrosinase inhibitors Bioorg. Med. Chem. Lett. 21 2376
Celik F, Arslan M, Kaya M O, Yavuz E, Gencer N and Arslan O 2014 Synthesis and carbonic anhydrase inhibitory properties of tetrazole- and oxadiazole substituted 1,4-dihydropyrimidinone compounds Artif. Cells Blood Substit. Biotechnol. 42 58
Mohammadi B and Behbahani F K 2018 Recent developments in the synthesis and applications of dihydropyrimidin-2(1H)-ones and thiones Mol. Divers. 2018 1
(a) Nagarajaiah H, Mukhopadhyay A and Moorthy J N 2016 Biginelli reaction: an overview Tetrahedron Lett. 57 5135; (b) Kappe C O 2000 Recent Advances in the Biginelli Dihydropyrimidine Synthesis. New Tricks from an Old Dog Acc. Chem. Res. 33 879
Piqani B and Zhang W 2011 Synthesis of diverse dihydropyrimidine-related scaffolds by fluorous benzaldehyde-based Biginelli reaction and post-condensation modifications Beilstein J. Org. Chem. 7 1294
Quan Z-J, Wei Q-B, Ma D-D, Da Y-X, Wang X-C and Shen M-S 2009 General and green synthesis of C-6 sulfonylmethyl 4-aryl-3,4-dihydropyrimidinones in water Synth. Commun. 39 2230
Patra G C, Bhunia S C, Roy M K and Pal S C 2013 Synthesis of 4-Aryl-4,6-dihydropyrimido[4,5-d]pyridazine-2,5(1H,3H)-diones from Biginelli Compounds Helv. Chim. Acta 96 130
Kappe C O 1990 Synthesis and reactions of biginelli compounds, 3. Unexpected formation of nitriles from reactions of 6-(dibromomethyl)-1,2,3,4-tetrahydro-2-oxo-5-pyrimidinecarboxylates with sodium azide Liebigs Ann. Chem. 1990 505
Tale N P, Shelke A V, Bhong B Y and Karade N N 2013 Regioselective chlorination at C-6 methyl position of 3,4-dihydropyrimidin-2(1H)-ones using (dichloroiodo)benzene Monatsh. Chem. 144 981
Puchala A, Belaj F, Kappe C O and Bergman J 2001 On the reaction of 3,4-dihydropyrimidones with nitric acid. Preparation and X-ray structure analysis of a stable nitrolic acid J. Heterocycl. Chem. 38 1345
Singh K, Singh S and Mahajan A 2005 Metalation of Biginelli compounds. A general unprecedented route to C-6 functionalized 4-aryl-3,4-dihydropyrimidinones J. Org. Chem. 70 6114
Khanina E L and Dubur G 1982 Oxidation of some derivatives of tetrahydropyrimidine-5-carboxylic acid with selenium dioxide Chem. Heterocycl. Compd. 18 412
Kolosov M A, Orlov V D, Vashchenko V V, Shishkina S V and Shishkin O V 2007 5-cinnamoyl- and 5-(ethoxycarbonyl)-6-styryl derivatives of 4-aryl-3,4-dihydropyrimidin-2(1h)-ones Collect. Czech. Chem. Commun. 72 1219
Shutalev A D and Aksionov A N 2005 Simple synthesis of 4-aryl-6-styryl-1,2,3,4-tetrahydropyrimidin-2-ones by the alkaline hydrolysis of Biginelli compounds Mendeleev Commun. 15 73
Zhang L, Zhang Z, Liu Q, Liu T and Zhang G 2014 Iron-catalyzed vinylogous aldol condensation of Biginelli products and its application toward pyrido[4,3-d]pyrimidinones J. Org. Chem. 79 2281
Falsone F S and Kappe C O 2001 The Biginelli dihydropyrimidone synthesis using polyphosphate ester as a mild and efficient cyclocondensation/dehydration reagent ARKIVOC 2001 122
Acknowledgements
We thank the SERB New Delhi, for financial support under EMR scheme (EMR/2014/000542).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mondal, M.A., Khan, A.A. & Mitra, K. Iron(III)-catalyzed selective direct olefination of dihydropyrimidinone with aromatic aldehyde. J Chem Sci 130, 151 (2018). https://doi.org/10.1007/s12039-018-1543-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-018-1543-z