Abstract
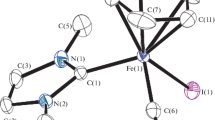
In methanol, FeCl3 reacted readily with L1H (L1H = bis(1-isoquinolylcarbonyl)amide) and afforded a complex having the formula [Fe(L2)Cl2] (1) {L2 − = N-((1-isoquinolyl)(methoxy)methyl)isoquinoline-1-carboxamide ion}. This reaction involves reduction of one of the two carbonyl groups present in L1H to (methoxy)methyl group. A plausible mechanism for the conversion of L1H to L2 − has been proposed. Determination of molecular structure of 1 confirmed this conversion. Fe(III) ion is surrounded by three nitrogen atoms of the ligand and two chloride ions, imparting a rare distorted trigonal bipyramidal N3Cl2 coordination environment.

In methanol, FeCl3 reacted with bis(1-isoquinolylcarbonyl)amide affording N-((1-isoquinolyl)(methoxy)methyl)isoquinoline-1-carboxamide bound iron(III) complex. This reaction involves reduction of one of the two carbonyl groups to (methoxy)methyl group.






Similar content being viewed by others
References
Bolm C, Legros J, Le Paih J and Zani L 2004 Chem. Rev. 104 6217
Plietker B 2008 In Iron Catalysis in Organic Chemistry: Reactions and Applications (Weinheim: Wiley- VCH)
Stamatatos T C, Efthymiou C G, Stoumpos C and Perlepes S P 2009 Eur. J. Inorg. Chem. 3361
Thirupathi P and Kim S S 2010 J. Org. Chem. 75 5240
Csihony S, Mehdi S S, Homonnay Z, Vértes A, Farkas Ö and Horváth I T 2002 J. Chem. Soc., Dalton Trans. 680
Kidwai M, Mishra N K, Bansal V, Kumar A and Mozumdar S 2009 Tetrahedron Lett. 50 1355
Christoffers J 1997 Chem. Commun. 943
Li H, Xu X, Yang J, Xie X, Huang H and Li Y 2011 Tetrahedron Lett. 52 530
Anxionnat B, Guérinot A, Reymond S and Cossy J 2009 Tetrahedron Lett. 50 3470
Niu T, Huang L, Wu T and Zhang Y 2011 Org. Biomol. Chem. 9 273
Li R, Wang S R and Lu W 2007 Org. Lett. 9 2219
Jenner B E L and Lindsey Jr. R V 1960 J. Am. Chem. Soc. 83 1911
Yiu S, Wu Z, Mak C and Lau T 2004 J. Am. Chem. Soc. 126 14921
Noda D, Sunada Y, Hatakeyama T, Nakamura M and Nagashima H 2009 J. Am. Chem. Soc. 131 6078
Stoyanovsky D A, Clancy R and Cederbaum A I 1999 J. Am. Chem. Soc. 121 5093
Trost B M and Ornstein P L 1983 J. Org. Chem. 48 1133
Toda F, Tanaka K and Iwata S 1989 J. Org. Chem. 54 3007
Wang J, Huang W, Zhang Z, Xiang X, Liu R and Zhou X 2009 J. Org. Chem. 74 3299
Li R, Wang S R and Lu W 2007 Org. Lett. 9 2219
Wang S, Wang Z and Zheng X 2009 Chem. Commun. 7372
Chatterjee S K and Nuhn P 1998 Chem. Commun. 1729
Liu C, Yang F, Jin Y, Ma X, Cheng D, Li N and Tian S 2010 Org. Lett. 12 3832
Chu C, Huang W, Lu C, Wu P, Liu J and Yao C 2006 Tetrahedron Lett. 47 7375
Iwanami K, Aoyagi M and Oriyama T 2005 Tetrahedron Lett. 46 7487
Karamé I, Alamé M, Kanj A, Baydoun G N, Hazimeh H, Masri M and Christ L 2011 C. R. Chim. 14 525
Elinson M N, Ilovaisky A I, Merkulova V M, Barba F and Batanero B 2008 Tetrahedron 64 5915
Sahu R, Fulwa V K, Jena H S and Manivannan V 2012 Polyhedron 33 9
Sahu R, Padhi S K, Jena H S and Manivannan V 2010 Inorg. Chim. Acta 363 1448
Sheldrick G M 1996 SADABS, University of Göttingen, Gttingen, Germany
SMART and SAINT, Siemens Analytical X-ray Instruments Inc., Madison, WI, 1996
Sheldrick G M 2008 Acta. Cryst. A 64 112
Ostendorf M, Romagnoli R, Cabeza Pereiro I C, Roos E C, Moolenaar M J, Speckamp W N and Hiemstra H 1997 Tetrahedron: Asymmetry 8 1773
Wocadlo S, Massa W and Folgado J 1993 Inorg. Chim. Acta. 207 199
Bedford R B, Bruce D W, Frost R M, Goodby J W and Hird M 2004 Chem. Commun. 2822
Yamaguchi Y, Ando H, Nagaya M, Hinago H, Ito T and Asami M 2011 Chem. Lett. 40 983
Addison A W, Rao T N, Reedijk J, van Rijn J and Verschor G C 1984 J. Chem. Soc. Dalton Trans. 1349
Small B L, Brookhart M and Bennett A M A 1998 J. Am. Chem. Soc. 120 4049
Britovsek G J P, Bruce M, Gibson V C, Kimberley B S, Maddox P J, Mastroianni S, McTavish S J, Redshaw C, Solan G A, Strömberg S, White A J P and Williams D J 1999 J. Am. Chem. Soc. 121 8728
Mandon D, Machkour A, Goetz S and Welter R 2002 Inorg. Chem. 41 5364
Acknowledgements
We are grateful to the Department of Science and Technology (DST), New Delhi, for financial grant and establishing the single crystal X-ray diffractometer facility under FIST scheme. The authors are thankful to Dr. Babulal Das for the X-ray data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Animesh Chakravorty on the occasion of his 80th birth anniversary.
Supplementary Information (SI)
CCDC number 987001 contains the supplementary crystallographic data for complex 1. This data can be obtained free of charge via http://www.ccdc.ac.uk/conts/retrieving.html or from the Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax: (+44) 1223-336-033; or e-mail: deposit@ccdc.cam.ac.uk. IR spectrum of 1 is provided which is available at www.ias.ac.in/chemsci.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
SAHU, R., CHALIHA, P. & MANIVANNAN, V. Iron(III) Chloride mediated reduction of Bis(1-isoquinolylcarbonyl)amide to an Amide† . J Chem Sci 128, 37–42 (2016). https://doi.org/10.1007/s12039-015-1001-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-015-1001-0