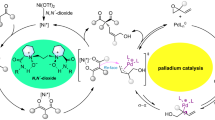
Abstract
Electrophilic allylation of phenolic substrates including salicylaldehydes with (α-hydroxy) allylphosphonates is presented. It is observed that catalytic FeCl3 is sufficient to accomplish the allylation. Interestingly, the reaction led to the formation of allylphosphonates in addition to vinylphosphonates, depending upon the substituent. The vinylphosphonates obtained here are E-isomers. More importantly, the reaction occurred regioselectively with respect to the phenolic substrates. Substituted allylphosphonates are formed when salicylaldehyde or (2-hydroxy-phenyl)arylmethanones are used. Conclusive proof for the formation of allylphosphontates as well as vinylphosphonates has been provided by single crystal X-ray crystallography.

Regioselective allylation of phenolic substrates using phosphono-allyl alcohols was achieved by using even catalytic amounts of FeCl3. Both vinylphosphonates and allylphosphonates are obtained, the ratio depending upon the substrate.





Similar content being viewed by others
References
(a) Larock R C 1999 In Comprehensive Organic Transformations (VCH: New York); (b) Olah G A, Krishnamurti R and Prakash G K S 1991 In Comprehensive Organic Synthesis B M Trost and I Fleming (ed.) (Oxford: Pergamon) vol. 3 pp. 293–339; (c) Roberts R M and Khalaf A A 1984 In Friedel–Crafts Alkylation Chemistry: A Century of Discovery (New York: Dekker); (d) Olah G A 1964 In Friedel–Crafts and Related Reactions (Wiley-Interscience: New York) vol. II, Part 1
(a) Tsuchimoto T, Tobita K, Hiyama T and Fukuzawa S.-i 1996 Synlett 557; (b) Nishibayashi Y, Yamanashi M, Takagi Y and Hidai M 1997 Chem. Commun. 859; (c) Malkov A V, Spoor P, Vinader V and Kočovský P 1999 J. Org. Chem. 64 5308; (d) Bonrath W, Haas A, Hoppmann E, Netscher T, Pauling H, Schager F and Wildermann A 2002 Adv. Synth. Catal. 344 37; (e) Basavaiah D, JaganmohanRao A and Satyanarayana T 2003 Chem. Rev. 103 811; (f) Hasegawa A, Ishihara K and Yamamoto H 2003 Angew. Chem., Int. Ed. 42 5731; (g) Kimura M, Fukasaka M and Tamaru Y 2006 Synthesis 3611; (h) Yadav J S, Reddy B V S, Aravind S, Kumar G G K S N and Reddy A S 2007 Tetrahedron Lett. 48 6117; (i) Jana U, Biswas S and Maiti S 2007 Tetrahedron Lett. 48 4065; (j) Rao W and Chan P W H 2008 Org. Biomol. Chem. 6 2426; (k) Yamamoto Y and Itonaga K 2009 Org. Lett. 11 717; (l) Usui I, Schmidt S, Keller M and Breit B 2008 Org. Lett. 10 1207; (m) Wang J, Zhang L, Jing Y, Huang W and Zhou X 2009 Tetrahedron Lett. 50 4978; (n) Zaitsev A B, Gruber S, Plüss P A, Pregosin P S, Veiros L F and Wörle M 2008 J. Am. Chem. Soc. 130 11604; (o) Guerinot A, Serra-Muns A, Gnamm C, Bensoussan C, Reymond S and Cossy J 2010 Org. Lett. 12 1808; (p) Rueping M, Nachtsheim B J 2010 Beilstein J. Chem. 6 6
(a) Rowe B J and Spilling C D 2003 J. Org. Chem. 68 9502; (b) Yan B and Spilling C D 2004 J. Org. Chem. 69 2859; (c) He A, Yan B, Thanavaro A, Spilling C D and Rath N P 2004 J. Org. Chem. 69 8643; (d) Point V, Malla R K, Diomande S, Martin B P, Delorme V, Carriere F, Cannan S, Rath N P, Spilling C D and Cavalier J -F 2012 J. Med. Chem. 55 10204; (e) He A, Sutivisedsak N and Spilling C D 2009 Org. Lett. 11 3124; (d) Roy S and Spilling C D 2012 Org. Lett. 14 2230; (g) Sanders T C and Hammond G B 1993 J. Org. Chem. 58 5598
Yan B and Spilling C D 2008 J. Org. Chem. 73 5385
Malla R K, Ridenour J N and Spilling C D 2014 Beilstein J. Org. Chem. 10 1933
(a) Minami T, Okauchi T and Kouno R 2001 Synthesis 349; (b) Maffei M 2004 Curr. Org. Synth. 355; (b) Dembitsky V M, Quntar A A A, Haj-Yehia A and Srebnik M 2005 Mini-Rev. Org. Chem. 2 91
Janecki T, Kedzia J and Wasek T 2009 Synthesis 1227
Janecki T, Albrecht A, Koszuk J F, Modranka J and Słowak D 2010 Tetrahedron Lett. 51 2274
Selected recent articles: (a) Tarabay J, Al-Maksoud W, Jaber F, Pinel C, Prakash S and Djakovitch L 2010 Applied Catal., A: General 388 124; (b) Garzon C, Attolini M and Maffei M 2011 Synthesis 3109; (c) Garzon C, Attolini M and Maffei M 2013 Eur. J. Org. Chem. 3653; (d) Opekar S, Pohl R, Eigner V and Beier P 2013 J. Org. Chem. 78 4573; (e) Liu Z, MacRitchie N, Pyne S, Pyne N J and Bittman R 2013 Bioorg. Med. Chem. 21 2503; (f) Sobhani S and Honarmand M 2013 Synlett 24 236; (g) Hernandez-Guerra D, Rodriguez M S and Suarez E 2013 Org Lett. 15 250; (h) Cai Y, Ge H, Yu C, Sun W, Zhan J and Miao Z 2014 RSC Advances 4 21492; (i) Adler P, Fadel A and Rabasso N 2014 Tetrahedron 70 4437; (j) Hernandez-Guerra D, Rodriguez M S and Suarez E 2014 Eur. J. Org. Chem. 5033; (k) Wu Y, Liu L, Yan K, Xu P, Gao Y and Zhao Y 2014 J. Org. Chem. 79 8118
Representative publications: (a) Macarie L and Ilia G 2010 Progr. Polymer Sci. 35 1078; (b) Salzinger S and Rieger B 2012 Macromol. Rapid Commun. 33 1327; (c) Li J, Ni X, Ling J and Shen Z 2013 J. Polym. Sci., Polymer Chem. 51 2409; (d) Sannigrahi A, Takamuku S and Jannasch P 2013 Polymer Chem. 4 4207; (e) Salzinger S, Soller B S, Plikhta A, Seemann U B, Herdtweck E and Rieger B 2013 J. Am. Chem. Soc. 135 13030; (f) Soller B S, Zhang N and Rieger B 2014 Macromol. Chem. Phys. 215 1946; (g) Yang J, Liang Y, Salzinger S, Zhang N, Dong D and Rieger B J 2014 Polymer Sci., Part A: Polymer Chem. 52 2919
Baszczyňski O, Jansa P, Dračínský M, Kaiser M M Špaček P and Janeba Z 2012 RSC Advances 2 1282
Duan S –W, Liu Y –Y, Ding W, Li T –R, Shi D –Q, Chen J –R and Xiao W –J 2013 Synthesis 45 1647
(a) Sikorski J A and Gruys K J 1997 Acc. Chem. Res. 30 2; (b) Mader M M and Bartlett P A 1997 Chem. Rev. 97 1281; (c) Allenberger F and Klare I 1999 Antimicrob. Chemother. 43 211; (d) Kukhar V and Hudson H R 2000 In Aminophosphinic and Aminophosphonic Acids: Chemistry and Biological Activity (Wiley-Blackwell: Chichester); (e) Demmer C S, Krogsgaard-Larsen N and Bunch L 2011 Chem. Rev. 111 7981; (f) McGrath J W, Chin J P and Quinn J P 2013 Nature Rev. Microbiol. 11 413
(a) Boojamra C G, Cannizzaro C, Chen X, Cho A, Chong L S, Fardis M, Huang A X, Kim C U, Kirschberg T, Krawczyk S, Lee C P, Lin K-Y, Mackman R L, Markevitch D Y, Nelson P H, Oare D A, Prasad V K, Pyun H-J, Ray A S, Swaminathan S, Watkins W J, Zhang J R and Zhang L Anti-cancer phosphonate analogs US7452901 B2 2008; (b) Queffeléc C, Petit M, Janvier P, Knight D A and Bujoli B 2012 Chem. Rev. 112 3777; (c) Monge S, Canniccioni B, Graillot A and Robin J –J 2011 Biomacromolecules 12 1973 (d) David G, Negrell-Guirao C, Iftene F, Boutevin B and Chougrani K 2012 Polym. Chem. 3 265; (e) Jin S and Gonsalves K E 1998 Macromolecules 31 1010; (f) Ebdon J R 1997 Recent Adv. Flame Retard. Polym. Mater. 8 161
(a) Kotikalapudi R and Kumara Swamy K C 2012 Tetrahedron Lett. 53 3831; (b) Kotikalapudi R 2013 In Gold Gold Catalyzed Cyclizations of Alkynols/Propargylic Esters and Allenylphosphonates/Allenylphosphine Oxides in Cycloaddition/Cyclization Reactions (PhD Thesis University of Hyderabad: Hyderabad); (c) Uravakilli A, Kotikalapudi R and Kumara Swamy K C 2014 Synthesis 46 1197
For earlier work, see: (a) Kumaraswamy S, Selvi R S and Kumara Swamy K C 1997 Synthesis 207; (b) Muthiah C, Praveen Kumar K, Aruna Mani C and Kumara Swamy K C 2000 J. Org. Chem. 65 3733
(a) Pallikonda G, Chakravarty M 2013 Eur. J. Org. Chem. 944; (b) Pallikonda G, Chakravarty M and Sahoo M K 2014 Org. Biomol. Chem. 12 7140
(a) Bhuvan Kumar N N, Nagarjuna Reddy M and KumaraSwamy K C 2009 J. Org. Chem. 74 5395; (b) Phani Pavan M, Chakravarty M and Kumara Swamy K C 2009 Eur. J. Org. Chem. 5927; (c) Phani Pavan M and Kumara Swamy K C 2011 Synlett 1288; (d) Srinivas V, Sajna K V and Kumara Swamy K C 2011 Tetrahedron Lett. 52 5323; (e) Phani Pavan M, Nagarjuna Reddy M, Bhuvan Kumar N N and Kumara Swamy K C 2012 Org. Biomol. Chem. 10 8113; (f) Srinivas V, Sajna K V and Kumara Swamy K C 2011 Chem. Commun. 47 5629; (g) Sajna K V and Kumara Swamy K C 2012 J. Org. Chem. 77 5345
(a) Texier-Boullet F and Foucaud A 1982 Synthesis 165; (b) Cen W, Dai X and Shen Y 1993 J. Fluorine Chem. 65 49; (c) Shen Y and Qi M 1994 J. Chem. Soc., Perkin Trans. 1 1179
Perrin D D, Armarego W L F and Perrin D R 1986 In Purification of Laboratory Chemicals (Pergamon: Oxford UK)
(a) Sheldrick G M 1996 SADABS, Siemens Area Detector Absorption Correction (University of Göttingen: Germany); (b) Sheldrick G M 1997 SHELX-97: A program for crystal structure solution and refinement (University of Göttingen: Germany; (c) Sheldrick G M 1999 SHELXTL NT Crystal Structure Analysis Package, Version 5 Bruker AXS Analytical X-ray System: WI (USA)
Acknowledgements
We thank the Department of Science and Technology (DST, New Delhi) and the University Grants Commission (UGC, New Delhi) for financial support. MA and RK thank University Grants Commission (UGC, New Delhi) for fellowship. KCK thanks DST for the J. C. Bose fellowship (No. SR/S2/JCB-53/2010) and UGC for a one-time grant [No. F4-10/2010 (BSR)].
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
Crystallographic data for the structure 7a, 9a and 11b have been deposited with the Cambridge Crystallo- graphic Data Centre as supplementary publication num- ber CCDC 1028043-1028045. Copies of the data can be obtained free of charge, on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK [fax: +44(1223) 336033 or e-mail: deposit@ccdc.cam.ac.uk]. Copies of 1H/13C/31P NMR spectra are available at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
ANITHA, M., KOTIKALAPUDI, R. & SWAMY, K.C.K. FeCl3 catalysed regioselective allylation of phenolic substrates with (α-hydroxy)allylphosphonates. J Chem Sci 127, 1465–1475 (2015). https://doi.org/10.1007/s12039-015-0903-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-015-0903-1