Abstract
Density functional theory (DFT) calculations have been performed to explore the potential energy surfaces of C–O bond activation in CO2 molecule by gas-phase Mo + cation and Mo atom, in order to better understanding the mechanism of second-row metal reacting with CO2. The minimum energy reaction path is found to involve the spin inversion in the different reaction steps. This potential energy curve-crossing dramatically affects reaction exothermic. The present results show that the reaction mechanism is insertion-elimination mechanism along the C–O bond activation branch. All the theoretical results not only support the existing conclusions inferred from early experiment, but also complement the pathway and mechanism for this reaction.

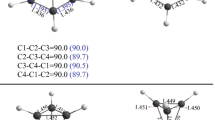
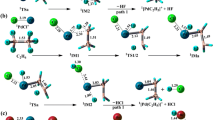
For the reaction between Mo atom and CO2, it is found that the reaction system would likely to change its spin multiplicity twice in going from the entrance channel to the exit channel. Specifically, it can be described as 5Mo+CO2→5IM1→5TS12→5IM2→5TS23→CP4→3IM3→CP6→5MoO+CO.
Similar content being viewed by others
References
Leitner W 2002 Acc. Chem. Res. 35 746
Rondinelli F, Russo N and Toscano M 2006 Theor. Chem. Acc. 115 434
Tommaso S D, Marino T, Rondinelli F, Russo N and Toscano M 2007 J. Chem. Theor. Comput. 3 811
Kafafi Z H, Hauge R H, Billups W E and Margrave J L 1983 J. Am. Chem. Soc. 105 3886
Andrews L and Tague T J 1998 J. Am. Chem. Soc. 120 13230
Solov’ev V N, Polikarpov E V, Nemukhin A V and Sergeev G B 1999 J. Phys. Chem. A. 103 6721
Zhou M F and Andrews L 1998 J. Am. Chem. Soc. 120 13230
Galan F, Fouassier M, Tranquille M, Mascetti J and Papai I 1997 J. Phys. Chem. A. 101 2626
Chertihin G V and Andrews L 1995 J. Am. Chem. Soc. 117 1595
Zhou M F and Andrews L 1999 J. Phys. Chem. A. 103 2066
Sievers M R and Armentrout P B 1999 Int. J. Mass. Spectrom. 185 117
Sievers M R and Armentrout P B 1998 Int. J. Mass. Spectrom. 179 115
Sievers M R and Armentrout P B 1999 Inorg. Chem. 38 397
Sievers M R and Armentrout P B 1995 J. Chem. Phys. 102 754
Sievers M R and Armentrout P B 1998 Int. J. Mass. Spectrom. 179 103
Kretzschmar I, Schroder D, Schwarz H and Armentrout P B 2006 Int. J. Mass. Spectrom. 249 263
Chen M, Wang X, Zhang L and Qin Q Z 2000 J. Phys. Chem. A. 104 7010
Souter P F and Andrews L 1997 Chem. Commun. 777
Wang X, Chen M, Zhang L and Qin Q Z 2000 J. Phys. Chem. A. 104 758
Souter P F and Andrews L 1997 J. Am. Chem. Soc. 119 7350
Zhou M F, Liang B and Andrews L 1999 J. Phys. Chem. A. 103 2013
Liang B and Andrews L 2002 J. Phys. Chem. A. 106 595
Liang B and Andrews L 2002 J. Phys. Chem. A. 106 4042
Andrews L, Zhou M F, Liang B, Li J and Bursten B E 2000 J. Am. Chem. Soc. 122 11440
Quere A M L, Xu C and Manceron L 1991 J. Phys. Chem. 95 3031
Papai I, Schubert G, Hannachi Y and Mascetti J 2002 J. Phys. Chem. A. 106 9551
Papai I, Mascetti J and Fournier R 1997 J. Phys. Chem. A. 101 4465
Papai I, Hannachi Y, Gwizdala S and Mascetti J 2002 J. Phys. Chem. A. 106 4181
Hannachi Y, Mascetti J, Stirling A and Papai I 2003 J. Phys. Chem. A. 107 6708
Dobrogorskaya Y, Mascetti J, Papai I and Hannachi Y 2005 J. Phys. Chem. A. 109 7932
Zhou M F, Tsumori N, Li Z, Fan K N, Andrews L and Xu Q 2002 J. Am. Chem. Soc. 124 12936
Jiang L and Xu Q 2005 J. Am. Chem. Soc. 127 42
Xu Q, Jiang L and Tsumori N 2005 Angew. Chem. Int. Ed. 44 4338
Jiang L and Xu Q 2005 J. Am. Chem. Soc. 127 8906
Campbell M L 1999 Phys. Chem. Chem. Phys. 1 3731
Campbell M L 2000 Chem. Phys. Lett. 330 547
Yrsson R and Mascetti J 2005 React. Kinet. Catal. Lett. 285 107
Jiang L, Zhang X B, Han S and Xu Q 2008 Inorg. Chem. 47 4826
Jiang L and Xu Q 2007 J. Phys. Chem. A. 111 3519
Sievers M R and Armentrout P B 1998 J. Phys. Chem. A. 102 10754
Fischer G, Goursot A, Coq Bernard, Delahay G and Pal S 2006 ChemPhysChem. 7 1795
Cho H G and Andrews L 2005 J. Am. Chem. Soc. 127 8226
Armentrout P B 2007 Organometallics. 26 5473
Armentrout P B 2007 Organometallics. 26 5486
Rutkowska-Zbik D, Tokarz-Sobieraj R and Witko M 2007 J. Chem. Theor. Comput. 3 914
Becke A D 1993 J. Chem. Phys. 98 5648
Lee C, Yang W and Parr R G 1988 Phys. Rev. B. 37 785
Holthausen M C and Koch W 1996 J. Am. Chem. Soc. 118 9932
Holthausen M C, Fiedler A, Schwarz H and Koch W 1996 J. Phys. Chem. 100 6236
Guo Z, Ke Z F, Phillips D L and Zhao C Y 2008 Organometallics. 27 181
Dolg M, Stoll H, Savin A and Preuss H 1989 Theor. Chim. Acta. 75 173
Fukui K 1981 Accounts. Chem. Res. 14 363
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R, Zakrzewski V G, Montgomery J A Jr, Stratmann R E, Burant J C, Dapprich S, Millam J M, Daniels A D, Kudin K N, Strain M C, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson G A, Ayala P Y, Cui Q, Morokuma K, Malick D K, Rabuck A D, Raghavachari K, Foresman J B, Cioslowski J, Ortiz J V, Baboul A G, Stefanov B B, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin R L, Fox D J, Keith T, Al-Laham M A, Peng C Y, Nanayakkara A, Gonzalez C, Challacombe M, Gill P M W, Johnson B, Chen W, Wong M W, Andres J L, Gonzalez C, Head-Gordon M, Replogle E S and Pople J A 2003 Gaussian 03, Revision B04, Pittsburgh PA: Gaussian Inc.
Broclawik E 1995 Int. J. Quantum. Chem. 56 779
Kretzschmar I, Fiedler A, Harvey J N, Schroder D and Schwarz H 1997 J. Phys. Chem. A. 101 6252
Yoshizawa K, Shiota Y and Yamabe T 1999 J. Chem. Phys. 111 538
Fiedler A, Schroder D, Shaik S and Schwarz H 1994 J. Am. Chem. Soc. 116 3563
Harvey J N, Poli R and Smith K M 2003 Coord. Chem. Rev. 238 347
Zhang G B, Li S H and Jiang Y S 2003 Organometallics. 22 3820
Schroder D, Shaik S and Schwarz H 2000 Acc. Chem. Res. 33 139
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
HAN, D., DAI, G., CHEN, H. et al. DFT study of the reactions of Mo and Mo + with CO2 in gas phase. J Chem Sci 123, 299–309 (2011). https://doi.org/10.1007/s12039-011-0072-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-011-0072-9