Abstract
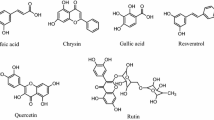
In continuation of our studies on the bioaccessibility of phenolic compounds from food grains as influenced by domestic processing, we examined the uptake of phenolics from native/sprouted finger millet (Eleucine coracana) and green gram (Vigna radiata) and native/heat-processed onion (Allium cepa) in human Caco-2 cells. Absorption of pure phenolic compounds, as well as the uptake of phenolic compounds from finger millet, green gram, and onion, was investigated in Caco-2 monolayer model. Transport of individual phenolic compounds from apical compartment to the basolateral compartment across Caco-2 monolayer was also investigated. Sprouting enhanced the uptake of syringic acid from both these grains. Open-pan boiling reduced the uptake of quercetin from the onion. Among pure phenolic compounds, syringic acid was maximally absorbed, while the flavonoid isovitexin was least absorbed. Apparent permeability coefficient P(app) of phenolic compounds from their standard solutions was 2.02 × 10−6 cm/s to 8.94 × 10−6 cm/s. Sprouting of grains enhanced the uptake of syringic acid by the Caco-2 cells. Open-pan boiling drastically reduced the uptake of quercetin from the onion. The permeability of phenolic acids across Caco-2 monolayer was higher than those of flavonoids.






Similar content being viewed by others
References
Adam A, Crespy V, Levrat-Verny MA, Leenhardt F, Leuillet M, Demigné C and Rémésy C 2002 The bioavailability of ferulic acid is governed primarily by the food matrix rather than its metabolism in intestine and liver in rats. J. Nutr. 132 1962–1968
Borrás-Linares I, Herranz-López M, Barrajón-Catalán E, Arráez-Román D, Gonzálezlvarez I, Bermejo M and Segura-Carretero A 2015 Permeability study of polyphenols derived from a phenolic-enriched Hibiscus sabdariffa extract by UHPLC-ESI-UHR-Qq-TOF-MS. Int. J. Mol. Sci. 16 18396–18411
Calabriso N, Scoditti E, Massaro M, Pellegrino M, Storelli C, Ingrosso I, Giovinazzo G and Carluccio MA 2016 Multiple anti-inflammatory and anti-atherosclerotic properties of red wine polyphenolic extracts: differential role of hydroxycinnamic acids, flavonols and stilbenes on endothelial inflammatory gene expression. Eur. J. Nutr. 55 477–489
Cao D, Li H, Yi J, Zhang J, Che H, Cao J and Jiang W 2011 Antioxidant properties of the mung bean flavonoids on alleviating heat stress. PLoS ONE 6 e21071
Caridi D, Trenerry VC, Rochfort S, Duong S, Laugher D and Jones R 2007 Profiling and quantifying quercetin glucosides in onion (Allium cepa L.) varieties using capillary zone electrophoresis and high performance liquid chromatography. Food Chem. 105 691–699
Chitindingu K, Benhura MAN and Muchuweti M 2015 In vitro bioaccessibility assessment of phenolic compounds from selected cereal grains: A prediction tool of nutritional efficiency. LWT Food Sci. Tech. 63 575–581
Devi PB, Vijayabharathi R, Sathyabama S, Malleshi NG and Priyadarisini VB 2014 Health benefits of finger millet (Eleusine coracana L.) polyphenols and dietary fiber: a review. J. Food Sci. Technol. 51 1021–1040
Galdón BR, Rodríguez EMR and Romero CD 2008 Flavonoids in onion cultivars (Allium cepa L.). J. Food Sci. 73 C599–C605
Graf BA, Milbury PE and Blumberg JB 2005 Flavonols, flavones, flavanones, and human health: epidemiological evidence. J. Med. Food 8 281–290
Guardia T, Rotelli AE, Juarez AO and Pelzer LE 2001 Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Farmaco (Società Chimica Italiana) 56 683–687
Henagan TM, Cefalu WT, Ribnicky DM, Noland RC, Dunville K, Campbell WW and Morrison CD 2014 In vivo effects of dietary quercetin and quercetin-rich red onion extract on skeletal muscle mitochondria, metabolism, and insulin sensitivity. Genes Nutr. 10 1–12
Hithamani G and Srinivasan K 2014a Effect of domestic processing on the polyphenol content and bioaccessibility in finger millet (Eleusine coracana) and pearl millet (Pennisetum glaucum). Food Chem. 164 55–62
Hithamani G and Srinivasan K 2014b Bioaccessibility of polyphenols from wheat (Triticum aestivum), sorghum (Sorghum bicolor), green gram (Vigna radiata), and chickpea (Cicer arietinum) as influenced by domestic food processing. J. Agric. Food Chem. 62 11170–11179
Hithamani G and Srinivasan K 2016a Bioaccessibility of polyphenols from onion (Allium cepa) as influenced by domestic heat processing and acidulants. Indian J. Nutr. Dietet. 53 391–404
Hithamani G and Srinivasan K 2016b Bioaccessibility of polyphenols from selected cereal grains and legumes as influenced by food acidulants. J. Sci. Food Agric. doi:10.1002/jsfa.7776
Hubatsch I, Ragnarsson EGE and Artursson P 2007 Determination of drug permeability and prediction of drug absorption in Caco-2 monolayers. Nat. Protoc. 2 2111–2119
Jakubowicz-Gil J, Rzeski W, Zdzisinska B, Dobrowolski P and Gawron A 2008 Cell death and neuronal arborization upon quercetin treatment in rat neurons. Acta Neurobiol. Exp. 68 139–146
Jiamboonsri P, Pithayanukul P, Bavovada R, Leanpolchareanchai J, Yin T, Gao S and Hu M 2017 Factors influencing oral bioavailability of Thai mango seed kernel extract and its key phenolic principles. Molecules 20 21254–21273
Kaulmann A, André CM, Schneider Y, Hoffmann L and Bohn T 2016 Carotenoid and polyphenol bioaccessibility and cellular uptake from plum and cabbage varieties. Food Chem. 197 325–332
Kaur M, Sandhu KS, Ahlawat RP and Sharma S 2015 In vitro starch digestibility, pasting and textural properties of mung bean: effect of different processing methods. J. Food Sci. Technol. 52 1642–1648
Konoshi Y and Shimizu M 2003 Transepithelial transport of ferulic acid by monocarboxylic acid transporter in Caco-2 cell monolayers. Biosci. Biotechnol. Biochem. 67 856–862
Lafay S and Gil-Izquierdo A 2008 Bioavailability of phenolic acids. Phytochem. Rev. 7 301–311
Lee HJ, Cha KH, Kim CY, Nho CW and Pan CH 2014 Bioavailability of hydroxycinnamic acids from Crepidiastrum denticulatum using simulated digestion and Caco-2 intestinal cells. J. Agric. Food Chem. 62 5290–5295
Lowry OH, Rosebrough NJ, Farr AL and Randall RJ 1951 Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193 265–275
Manach C, Scalbert A, Morand C, Rémésy C and Jiménez L 2004 Polyphenols- food sources and bioavailability. Am. J. Clin. Nutr. 79 727–747
Meinl W, Ebert B, Glatt H and Lampen A 2008 Sulfotransferase forms expressed in human intestinal Caco-2 and TC7 cells at varying stages of differentiation and role in benzo[a]pyrene metabolism. Drug Metab. Dispos. 36 276–283
Peng X, Zheng Z, Cheng KW, Shan F, Ren GX, Chen F and Wang M 2008 Inhibitory effect of mung bean extract and its constituents vitexin and isovitexin on the formation of advanced glycation end products. Food Chem. 106 475–481
Price KR, Bacon JR and Rhodes MJC 1997 Effect of storage and domestic Processing on the content and composition of flavonol glucosides in onion (Allium cepa). J. Agric. Food Chem. 45 938–942
Pu F, Mishima K, Irie K, Motohashi K, Tanaka Y, Orito K and Fujiwara M 2007 Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. J. Pharm. Sci. 104 329–334
Rastogi H and Jana S 2016 Evaluation of physicochemical properties and intestinal permeability of six dietary polyphenols in human intestinal colon adenocarcinoma Caco-2 cells. Eur. J. Drug Metab. Pharmacokinet. 41 33-43
Reddy RRL and Srinivasan K 2011 Dietary fenugreek and onion attenuate cholesterol gallstone formation in lithogenic diet–fed mice. Int. J. Exp. Pathol. 92 308–319
Seithel A, Karlsson J, Hilgendorf C, Björquist A and Ungell AL 2006 Variability in mRNA expression of ABC- and SLC-transporters in human intestinal cells: Comparison between human segments and Caco-2 cells. Eur. J. Pharm. Sci. 28 291–299
Shobana S, Harsha MR, Platel K, Srinivasan K and Malleshi NG 2010 Amelioration of hyperglycaemia and its associated complications by finger millet (Eleusine coracana L.) seed coat matter in streptozotocin-induced diabetic rats. Br. J. Nutr. 104 1787–1795
Sun D, Lennernäs H, Welage LS, Barnett JL, Landowski CP, Foster D, Fleisher D, Lee KD and Amidon GL 2002 Comparison of human duodenum and Caco-2 gene expression profiles for 12,000 gene sequences tags and correlation with permeability of 26 drugs. Pharmaceut. Res. 19 1400–1416
Tenore GC, Campiglia P, Giannetti D and Novellino E 2015 Simulated gastrointestinal digestion, intestinal permeation and plasma protein interaction of white, green, and black tea polyphenols. Food Chem. 169 320–326
Willenberg I, Michael M, Wonik J, Bartel LC, Empl MT and Schebb NH 2015 Investigation of the absorption of resveratrol oligomers in the Caco-2 cellular model of intestinal absorption. Food Chem. 167 245–250
Xiang D, Wang C, Wang W, Shi C, Xiong W, Wang M and Fang J 2017 Gastrointestinal stability of dihydromyricetin, myricetin, and myricitrin: an in vitro investigation. Int. J. Food Sci. Nutr. 68 704–711
Zhang P 2015 Polyphenols in health and disease. Cell Biochem. Biophys. 73 649–664
Zhang Y, Tie X, Bao B, Wu X and Zhang Y 2007 Metabolism of flavone C-glucosides and p-coumaric acid from antioxidant of bamboo leaves (AOB) in rats. Br. J. Nutr. 97 484–494
Acknowledgements
GH is grateful to the Indian Council of Medical Research, New Delhi, for the award of Senior Research Fellowship. This research received no specific grant from any funding agency in public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: María Eliano Lanio
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hithamani, G., Kizhakayil, D. & Srinivasan, K. Uptake of phenolic compounds from plant foods in human intestinal Caco-2 cells. J Biosci 42, 603–611 (2017). https://doi.org/10.1007/s12038-017-9705-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12038-017-9705-6