Abstract
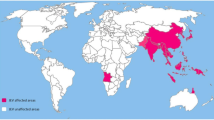
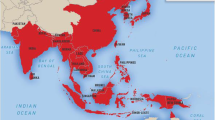
Japanese encephalitis (JE) remains the most important cause of acute viral encephalitis and continues to spread to hitherto unaffected regions like Indonesia, Pakistan and Australia. Approximately 60% of the world population inhabits JE endemic areas. Despite its restricted range mostly in the developing countries, a high annual incidence of 50,000 cases and about 10,000 deaths has been reported. Disease can be fatal in 25% cases. Magnitude of the problem is even more alarming since the survivors are left with serious long-term neuropsychiatric sequelae. Almost every two years, epidemics of JE occur in Indian subcontinent with a high mortality. JE virus infection results in different disease manifestations in host from mild subclinical febrile illness to clinical infections leading to encephalitis. No antiviral treatment is so far available for JE. The prevention of JE can be achieved by controlling the vector or by immunization regime. The vector control in the rural areas, which are the worst affected ones, is practically almost impossible. Three vaccines that have been implicated against JE include inactivated mouse brain derived, inactivated cell culture derived and cell culture derived live attenuated JE vaccine. But each has its own limitation. Currently, attempts to synthesize recombinant DNA vaccine are being made. New therapeutics are on the way of development like use of minocycline, short interfering RNA, arctigenin, rosmarinic acid, DNAzymes etc. However, the immune mechanisms that lead to JE are complex and need to be elucidated further for the development of therapeutics as well as safe and efficacious JE vaccines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CFR:
-
case fatality rate
- GM-CSF:
-
granulocyte-macrophage colony-stimulating factor
- JE:
-
Japanese encephalitis
- JEV:
-
JE virus
- MHC:
-
major histocompatibility complex
- NS:
-
non-structural
- PHK:
-
primary hamster kidney
References
Appaiahgari M B and Vrati S 2004 Immunogenicity and protective efficacy in mice of a formaldehyde-inactivated Indian strain of Japanese encephalitis virus grown in Vero cells; Vaccine 22 3669–3675
Appaiahgari M B and Vrati S 2007 DNAzyme-mediated inhibition of Japanese encephalitis virus replication in mouse brain; Mol. Ther. 15 1593–1599
Appaiahgari M B, Saini M, Rauthan M, Jyoti and Vrati S 2006 Immunization with recombinant adenovirus synthesizing the secretory form of Japanese encephalitis virus envelope protein protects adenovirus-exposed mice against lethal encephalitis; Microbes Infect. 8 92–104
Ashok M S and Rangarajan P N 1999 Immunization with plasmid DNA encoding the envelope glycoprotein of Japanese Encephalitis virus confers significant protection against intracerebral viral challenge without inducing detectable antiviral antibodies; Vaccine 18 68–75
Bharati K and Vrati S 2006 Japanese encephalitis: Development of new candidate vaccines; Expert Rev. Anti Infect. Ther. 4 313–324
Bharati K, Appaiahgari M B and Vrati S 2005 Effect of cytokine-encoding plasmid delivery on immune response to Japanese encephalitis virus DNA vaccine in mice; Microbiol. Immunol. 49 349–353
Burke D S and Morill J C 1987 Levels of interferon in the plasma and cerebrospinal fluid of patients with acute Japanese encephalitis; J. Infect. Dis. 155 797–799
Carey D E, Myers R M and Pavri K M 1968 Japanese encephalitis studies in Vellore, South India. II. Antibody response of patients; Indian J. Med. Res. 56 1319–1329
Chakravarty S K, Sarkar J K, Chakravarty M S, Mukherjee M K, Mukherjee K K, Das B C and Hati A K 1975 The first epidemic of Japanese encephalitis studied in India—virologicalstudies; Indian J. Med. Res. 63 77–82
Chang G J, Hunt A R and Davis B 2000 A single intramuscular injection of recombinant plasmid DNA induces protective immunity and prevents Japanese encephalitis in mice; J. Virol. 74 4244–4252
Chaturvedi U C, Mathur A, Chandra A, Das S K, Tandon H O and Singh U K 1980 Transplacental infection with Japanese encephalitis virus; J. Infect. Dis. 141 712–715
Chen C J, Raung S L, Kuo M D and Wang Y M 2002 Suppression of Japanese encephalitis virus infection by non-steroidal anti-inflammatory drugs; J. Gen. Virol. 83 1897–1905
Chen H W, Pan C H, Huan H W, Liau M Y, Chiang J R and Tao M H 2001 Suppression of immune response and protective immunity to a Japanese encephalitis virus DNA vaccine by coadministration of an IL-12-expressing plasmid; J. Immunol. 166 7419–7426
Das B P, Lal S and Saxena V K 2004 Outdoor resting preference of Culex tritaeniorhynchus, the vector of Japanese encephalitis in Warangal and Karim Nagar districts, Andhra Pradesh; J. Vector Borne Dis. 41 32–36
Gould E A, Zanotto P M and Holmes E C 1997 The genetic evolution of flaviviruses; in Factors in the emergence of arbovirus diseases (eds) J F Saluzzo and B Dodet (Paris: Elsevier) pp 51–63
Grossberg S E and Scherer W F 1966 The effect of host age, viral dose and route of inoculation on inapparent infection in mice with Japanese encephalitis virus; Proc. Soc. Exp. Biol. Med. 123 118–124
Guerin B and Pozzi N 2005. Viruses in boar semen: detection and clinical as well as epidemiological consequences regarding disease transmission by artificial insemination; Theriogenology 63 556–572
Guirakhoo F, Zhang Z X, Chambers T J, Delagrave S, Arroyo J, Barrett A D, Monath T P 1999 Immunogenicity, genetic stability, and protective efficacy of a recombinant, chimeric yellow fever-Japanese encephalitis virus (ChimeriVax-JE) as a live, attenuated vaccine candidate against Japanese encephalitis; Virology 257 363–372
Gupta A K, Lad V J and Koshy A A 2003 Protection of mice against experimental Japanese encephalitis virus infections by neutralizing anti-glycoprotein E monoclonal antibodies; Acta. Virol. 47 141–145
Halstead S B and Jacobson J 2003 Japanese encephalitis; Adv. Virus Res. 61 103–138
Hanna J N, Ritchie S A, Phillips D A, Lee J M, Hills S L, van den Hurk A F, Pyke A T, Johansen C A and Mackenzie J S 1999 Japanese encephalitis in north Queensland, Australia, 1998; Med. J. Aust. 170 533–536
Hanna J N, Ritchie S A, Phillips D A, Shield J, Bailey M C, Mackenzie J S, Poidinger M, McCall B J and Mills P J 1996 An outbreak of Japanese encephalitis in the Torres Strait, Australia, 1995; Med. J. Aust. 165 256–260
Harinasuta C, Nimmanitya S and Titsyakorn U 1985 The effect of interferon-alpha A on two cases of Japanese encephalitis in Thailand; Southeast Asian J. Trop. Med. Public Health 16 332–336
Harinasuta C, Wasi C and Vithanomsat S 1984 The effect of interferon on Japanese encephalitis virus in vitro; Southeast Asian J. Trop. Med. Public Health 15 564–568
Hennessy S, Liu Z, Tsai T F, Strom B L, Wan C M, Liu H L, Wu T X, Yu H J, Liu Q M, Karabatsos N, Bilker W B and Halstead S B 1996 Effectiveness of live-attenuated Japanese encephalitis vaccine (SA14-14-2): a case-control study; Lancet 347 1583–1586
Hoke C H, Nisalak A, Sangawhipa N, Jatanasen S, Laorakapongse T, Innis B L, Kotchasenee S, Gingrich J B, Latendresse J and Fukai K 1988 Protection against Japanese encephalitis by inactivated vaccines; N. Engl. J. Med. 319 608–614
Igarashi A, Tanaka M, Morita K, Takasu T, Ahmed A, Ahmed A, Akram D S and Waqar M A 1994 Detection of west Nile and Japanese encephalitis viral genome sequences in cerebrospinal fluid from acute encephalitis cases in Karachi, Pakistan; Microbiol. Immunol. 38 827–830
Innis B L 1995 Japanese encephalitis; in Exotic viral infections (ed.) J S Potterfield (London: Chapman and Hall) pp 147–174
Kabilan L, Rajendran R, Arunachalam N, Ramesh S, Srinivasan S, Samuel P P and Dash A P 2004a Japanese encephalitis in India: an overview; Indian J. Pediatr. 71 609–615
Kanesa-Thasan N, Smucny J J, Hoke C H, Marks D H, Konishi E, Kurane I, Tang D B, Vaughn D W, Mason P W and Shope R E 2000 Safety and immunogenicity of NYVAC-JEV and ALVAC-JEV attenuated recombinant Japanese encephalitis virus—poxvirus vaccines in vaccinia-nonimmune and vaccinia-immune humans; Vaccine 19 483–491
Kar N J, Bora D, Sharma R C, Bhattacharjee J, Datta K K and Sharma R S 1992 Epidemiological profile of Japanese encephalitis in Gorakhpur district, Uttar Pradesh, 1982–1988; J. Commun. Dis. 24 145–149
Kaur R, Rauthan M and Vrati S 2004 Immunogenicity in mice of a cationic microparticle-adsorbed plasmid DNA encoding Japanese encephalitis virus envelope protein; Vaccine 22 2776–2782
Kaur R, Sachdeva G and Vrati S 2002 Plasmid DNA immunization against Japanese encephalitis virus: Immunogenecity of membrane-anchored and secretory envelope protein; J. Infect. Dis. 185 1–12
Kimura-Kiroda J and Yasui K 1988 Protection of mice against Japanese encephalitis virus by passive administration of monoclonal antibodies; J. Immunol. 141 3606–3610
King N J, Getts D R, Getts M T, Rana S, Shrestha B and Kesson A M 2007 Immunopathology of flavivirus infections; Immunol. Cell Biol. 85 33–42
Konishi E, Ajiro N, Nukuzuma C, Mason P W and Kurane I 2003 Comparison of protective efficacies of plasmid DNAs encoding Japanese encephalitis virus proteins that induce neutralizing antibody or cytotoxic T lymphocytes in mice; Vaccine 21 3675–3683
Konishi E, Pincus S, Paoletti E, Shope R E and Wason P W 1994 Avipox virus-vectored Japanese encephalitis virus vaccines: use as vaccine candidates in combination with purified subunit immunogens; Vaccine 12 633–638
Konishi E, Yamaoka M, Khin-Sane-Win, Kurane I and Mason P W 1998 Induction of protective immunity against Japanese encephalitis in mice by immunization with a plasmid encoding Japanese encephalitis virus premembrane and envelope genes; J. Virol. 72 4925–4930
Kumar P, Krishna V D, Sulochana P, Nirmala G, Haridattatreya M and Satchidanandam V. 2004b Cell-mediated immune res-ponses in healthy children with a history of subclinical infec-tion with Japanese encephalitis virus: analysis of CD4+ and CD8+ T cell target specificities by intracellular delivery of viral proteins using the human immunodeficiency virus Tat protein transduction domain; J. Gen. Virol. 85 471–482
Kumar P, Lee S K, Shankar P and Manjunath N 2006 A single siRNA suppresses fatal encephalitis induced by two different flaviviruses; PLoS Med. 3(4) e96 doi:10.1371/journal.pmed.0030096
Kumar P, Sulochana P, Nirmala G, Haridattatreya M and Satchidanandam V. 2004a. Conserved amino acids 193–324 of non-structural protein 3 are a dominant source of peptide determinants for CD4+ and CD8+ T cells in a healthy Japanese encephalitis virus-endemic cohort; J. Gen. Virol. 85 1131–1143
Kumar R, Mathur A, Kumar A, Sharma S, Chakraborty S and Chaturvedi U C 1990 Clinical features & prognostic indicators of Japanese encephalitis in children in Lucknow (India); Indian J. Med. Res. 91 321–327
Liao C L, Lin Y L, Wu B C, Tsao C H, Wang M C, Liu C I, Huang Y L, Chen J H, Wang J P and Chen L K 2001 Salicylates inhibit flavivirus replication independently of blocking nuclear factor kappa B activation; J. Virol. 75 7828–7839
Lin Y L, Chen L K, Liao C L, Yeh C T, Ma S H, Chen J L, Huang Y L, Chen S S and Chiang H Y 1998 DNA immunization with Japanese encephalitis virus non-structural protein NS-1 elicits protective immunity in mice; J. Virol. 72 191–200
Mathur A, Arora K L and Chaturvedi U C 1981 Congenital infection of mice with Japanese encephalitis virus; Infect. Immun. 34 26–29
Mathur A, Arora K L and Chaturvedi U C 1982 Transplacental Japanese encephalitis virus (JEV) infection in mice during consecutive pregnancies; J. Gen. Virol. 59 213–217
McCown J, Cochran M, Putnak R, Feighny R, Burrous J, Henchal E and Hoke C 1990 Protection of mice against lethal Japanese encephalitis with a recombinant baculovirus vaccine; Am. J. Trop. Med. Hyg. 42 491–499
Monath T P, Guirakhoo F, Nichols R, Yoksan S, Schrader R, Murphy C, Blum P, Woodward S, McCarthy K, Mathis D, Johnson C and Bedford P 2003 Chimeric live, attenuated vaccine against Japanese encephalitis (ChimeriVax-JE): phase 2 clinical trials for safety and immunogenicity, effect of vaccine dose and schedule, and memory response to challenge with inactivated Japanese encephalitis antigen; J. Infect. Dis. 188 1213–1230
Monath T P, Soike K, Levenbook I, Zhang Z X, Arroyo J, Delagrave S, Myers G, Barrett A D, Shope R E, Ratterree M, Chambers T J and Guirakhoo F 1999 Recombinant, chimaeric live, attenuated vaccine (ChimeriVax) incorporating the envelope genes of Japanese encephalitis (SA14-14-2) virus and the capsid and nonstructural genes of yellow fever (17D) virus is safe, immunogenic and protective in non-human primates; Vaccine 17 1869–1882
Namachivayam V and Umayal K 1982 Proceedings of National Conference on Japanese Encephalitis (New Delhi: Indian Council of Medical Research) pp 30–33
Ni H, Burns N J, Chang G J, Zhang M J, Wills M R, Trent D W, Sanders P G and Barrett A D 1994 Comparison of nucleotide and deduced amino acid sequence of the 5′ non-coding region and structural protein genes of the wild-type Japanese encephalitis virus strain SA14 and its attenuated vaccine derivatives; J. Gen. Virol. 75 1505–1510
Nukuzuma C, Ajiro N, Wheeler C J and Konishi E 2003 Enhancing effect of vaxfectin on the ability of a Japanese encephalitis DNA vaccine to induce neutralizing antibody in mice; Viral Immunol. 16 183–189
Okamoto S, Yoshii H, Ishikawa T, Akagi T, Akashi M, Takahashi M, Yamanishi K and Mori Y 2008 Single dose of inactivated Japanese encephalitis vaccine with poly(gamma-glutamic acid) nanoparticles provides effective protection from Japanese encephalitis virus; Vaccine 26 589–594
Pan C H, Chen H W, Huang H W and Tao M H 2001 Protective mechanisms induced by a Japanese encephalitis virus vaccine: requirement for antibody but not CD8+ cytotoxic T cell responses; J. Virol. 75 11457–11463
Parida M, Dash P K, Tripathi N K, Ambuj, Sannarangaiah S, Saxena P, Agarwal S, Sahni A K, Singh S P, Rathi A K, Bhargava R, Abhyankar A, Verma S K, Rao P V and Shekhar K 2006 Japanese Encephalitis Outbreak, India, 2005; Emerg. Infect. Dis. 12 1427–1430
Phukan A C, Borah P K and Mahanta J 2004 Japanese encephalitis in Assam, northeast India; Southeast Asian J. Trop. Med. Public Health. 35 618–622
Plesner A M 2003 Allergic reactions to Japanese encephalitis vaccine; Immunol. Allergy Clin. North Am. 23 665–697
Rao J S, Misra S P, Patanayak S K, Rao T V, Das Gupta R K and Thapar B R 2000 Japanese Encephalitis epidemic in Anantapur district, Andhra Pradesh (October–November, 1999); J. Commun. Dis. 32 306–312
Rathi A K, Kushwaha K P, Singh Y D, Singh J, Sirohi R, Singh R K and Singh U K 1993 JE virus encephalitis: 1988 epidemic at Gorakhpur; Indian Pediatr. 30 325–333
Rauthan M, Kaur R, Appaiahgari M B and Vrati S 2004 Oral immunization of mice with Japanese encephalitis virus envelope protein synthesized in Escherichia coli induces antiviral antibodies; Microbes Infect. 6 1305–1311
Reuben R and Gajanana A 1997 Japanese encephalitis in India; Indian J. Pediatr. 64 243–251
Rosen L 1986 The natural history of Japanese encephalitis virus; Annu. Rev. Microbiol. 40 395–414
Saini M and Vrati S 2003 High-level synthesis of Johnson grass mosaic virus coat protein in Escherichia coli and its autoassembly to form virus-like particles; Protein Exp. Purif. 28 86–92
Scherer W F, Kitaoka M, Okuno T and Ogata T 1959 Ecologic studies of Japanese encephalitis virus in Japan. VII. Human infection; Am. J. Trop. Med. Hyg. 8 707–715
Seif S A, Morita K and Igarashi A 1996 A 27 amino acid coding region of JE virus E protein expressed in E. coli as fusion protein with glutathione-S-transferase elicit neutralizing antibody in mice; Virus Res. 43 91–96
Seif S A, Morita K, Matsuo S, Hasebe F and Igarashi A 1995 Finer mapping of neutralizing epitope(s) on the C-terminal of Japanese encephalitis virus E-protein expressed in recombinant Escherichia coli system; Vaccine 13 1515–1521
Solomon T 1997 Viral encephalitis in Southeast Asia; Neurol. Infect. Epidemiol. 2 191–199
Solomon T, Dung M N, Kneen R, Gainsborough M, Vaughn D W and Khanh V T 2000 Japanese Encephalitis; J. Neurol. Neurosurg. Psychiatry 68 405–415
Solomon T, Dung N M, Wills B, Kneen R, Gainsborough M, Diet T V, Thuy T T, Loan H T, Khanh V C, Vaughn D W, White N J and Farrar J J 2003 Interferon alfa-2a in Japanese encephalitis: a randomized double-blind placebo-controlled trial; Lancet. 361 821–826
Solomon T, Kneen R, Dung N M, Khanh V C, Thuy T T, Ha D Q, Day N P, Nisalak A, Vaughn D W and White N J 1998 Poliomyelitis-like illness due to Japanese encephalitis virus; Lancet 351 1094–1097
Srivastava A K, Putnak J R, Lee S H, Hong S P, Moon S B, Barvir D A, Zhao B, Olson R A, Kim S O, Yoo W D, Towle A C, Vaughn D W, Innis B L and Eckels K H. 2001 A purified inactivated Japanese encephalitis virus vaccine made in Vero cells; Vaccine 19 4557–4565
Swarup V, Ghosh J, Ghosh S, Saxena A and Basu A 2007 Antiviral and anti-inflammatory effects of rosmarinic acid in an experimental murine model of Japanese encephalitis; Antimicrob. Agents Chemother. 51 3367–3370
Swarup V, Ghosh J, Mishra M K and Basu A 2008 Novel strategy for treatment of Japanese encephalitis using arctigenin, a plant lignan; J. Antimicrob. Chemother. 61 679–688
Takegami T, Simamura E, Hirai K and Koyama J 1998 Inhibitory effect of furanonaphthoquinone derivatives on the replication of Japanese encephalitis virus; Antiviral Res. 37 37–45
Tiroumourougane S V, Raghava P and Srinivasan S 2002 Japanese viral encephalitis; Postgrad. Med. J. 78 205–215
Tsai T F, Chang G J and Yu X Y 1999 Japanese encephalitis vaccine. In: Plotkins SA, Orenstein ok, Thailand, 13–15 October 1998; Vaccine 18 1–25
Vajpayee A, Dey P N, Chakraborty A K and Chakraborty M S 1992 Study of the outbreak of Japanese encephalitis in Lakhimpur district of Assam in 1989; J. Indian Med. Assoc. 90 114–115
Vaughn D W and Hoke Jr C H 1992 The epidemiology of Japanese encephalitis: Prospects for prevention; Epidemiol. Rev. 14 197–221
Victor T J, Malathi M, Ravi V, Palani G and Appavoo N C 2000 First outbreak of Japanese encephalitis in two villages of Dharmapuri district in Tamil Nadu; Indian J. Med. Res. 112 193–197
Vrati S 2000 Comparison of the genome sequences and the phylogenetic analyses of the GP78 and the Vellore P20778 isolates of Japanese encephalitis virus from India; J. Biosci. 25 257–262
Xin Y Y, Ming Z G, Peng G Y, Jian A and Min L H 1988 Safety of a live-attenuated Japanese encephalitis virus vaccine (SA14-14-2) for children; Am. J. Trop. Med. Hyg. 39 214–217
Xin Y Y, Ming Z G, Peng G Y, Jian A and Min L H 1988 Safety of a live-attenuated Japanese encephalitis virus vaccine (SA14-14-2) for children; Am. J. Trop. Med. Hyg. 39 214–217
Yang D K, Kweon C H, Kim B H, Lim S I, Kwon J H, Kim S H, Song J Y and Han H R 2005 Immunogenicity of baculovirus expressed recombinant proteins of Japanese encephalitis virus in mice; J. Vet. Sci. 6 125–133
Yang D K, Kweon C H, Kim B H, Lim S I, Kwon J H, Kim S H, Song J Y and Han H R 2005 Immunogenicity of baculovirus expressed recombinant proteins of Japanese encephalitis virus in mice; J. Vet. Sci. 6 125–133
Zhang M J, Wang M J, Jiang S Z and Ma W Y 1989 Passive protection of mice, goats, and monkeys against Japanese encephalitis with monoclonal antibodies; J. Med. Virol. 29 133–138
Zhang M J, Wang M J, Jiang S Z and Ma W Y 1989 Passive protection of mice, goats, and monkeys against Japanese encephalitis with monoclonal antibodies; J. Med. Virol. 29 133–138
Zimmerman M D, Scott R M, Vaughn D W, Rajbhandari S, Nisalak A and Shrestha M P 1997 Short report: an outbreak of Japanese encephalitis in Kathmandu, Nepal; Am. J. Trop. Med. Hyg. 57 283–284
Zimmerman M D, Scott R M, Vaughn D W, Rajbhandari S, Nisalak A and Shrestha M P 1997 Short report: an outbreak of Japanese encephalitis in Kathmandu, Nepal; Am. J. Trop. Med. Hyg. 57 283–284
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saxena, V., Dhole, T.N. Preventive strategies for frequent outbreaks of Japanese encephalitis in Northern India. J Biosci 33, 505–514 (2008). https://doi.org/10.1007/s12038-008-0069-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12038-008-0069-9