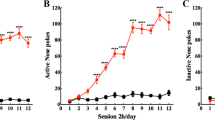
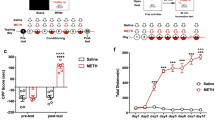
Abstract
Circular RNAs (circRNAs) are a novel type of non-coding RNAs. Despite the fact that the functional mechanisms of most circRNAs remain unknown, emerging evidence indicates that circRNAs could sponge microRNAs (miRNAs), bind to RNA binding proteins (RBP), and even be translated into protein. Recent research has demonstrated the crucial roles played by circRNAs in neuropsychiatric disorders. The medial prefrontal cortex (mPFC) is a crucial component of drug reward circuitry and exerts top-down control over cognitive functions. However, there is currently limited knowledge about the correlation between circRNAs and morphine-associated contextual memory in the mPFC. Here, we performed morphine-induced conditioned place preference (CPP) in mice and extracted mPFC tissue for RNA-sequencing. Our study represented the first attempt to identify differentially expressed circRNAs (DEcircRNAs) and mRNAs (DEmRNAs) in the mPFC after morphine-induced CPP. We identified 47 significantly up-regulated DEcircRNAs and 429 significantly up-regulated DEmRNAs, along with 74 significantly down-regulated DEcircRNAs and 391 significantly down-regulated DEmRNAs. Functional analysis revealed that both DEcircRNAs and DEmRNAs were closely associated with neuroplasticity. To further validate the DEcircRNAs, we conducted qRT-PCR, Sanger sequencing, and RNase R digestion assays. Additionally, using an integrated bioinformatics approach, we constructed ceRNA networks and identified critical circRNA/miRNA/mRNA axes that contributed to the development of morphine-associated contextual memory. In summary, our study provided novel insights into the role of circRNAs in drug-related memory, specifically from the perspective of ceRNAs.





Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Rybak-Wolf A, Stottmeister C, Glažar P, Jens M, Pino N, Giusti S, Hanan M, Behm M et al (2015) Circular RNAs in the mammalian brain are highly abundant, conserved, and dynamically expressed. Mol Cell 58(5):870–885. https://doi.org/10.1016/j.molcel.2015.03.027
Kyzar EJ, Bohnsack JP, Pandey SC (2022) Current and future perspectives of noncoding RNAs in brain function and neuropsychiatric disease. Biol Psychiatry 91(2):183–193. https://doi.org/10.1016/j.biopsych.2021.08.013
Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J (2019) The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet 20(11):675–691. https://doi.org/10.1038/s41576-019-0158-7
Bu Q, Long H, Shao X, Gu H, Kong J, Luo L, Liu B, Guo W et al (2019) Cocaine induces differential circular RNA expression in striatum. Transl Psychiatry 9(1):199. https://doi.org/10.1038/s41398-019-0527-1
Floris G, Gillespie A, Zanda MT, Dabrowski KR, Sillivan SE (2022) Heroin regulates orbitofrontal circular RNAs. Int J Mol Sci 23(3). https://doi.org/10.3390/ijms23031453
Mahmoudi E, Fitzsimmons C, Geaghan MP, Shannon Weickert C, Atkins JR, Wang X, Cairns MJ (2019) Circular RNA biogenesis is decreased in postmortem cortical gray matter in schizophrenia and may alter the bioavailability of associated miRNA. Neuropsychopharmacology 44(6):1043–1054. https://doi.org/10.1038/s41386-019-0348-1
Yu H, Xie B, Zhang J, Luo Y, Galaj E, Zhang X, Shen Q, Liu Y et al (2021) The role of circTmeff-1 in incubation of context-induced morphine craving. Pharmacol Res 170:105722. https://doi.org/10.1016/j.phrs.2021.105722
Shen Q, Xie B, Galaj E, Yu H, Li X, Lu Y, Zhang M, Wen D et al (2022) CircTmeff-1 in the nucleus accumbens regulates the reconsolidation of cocaine-associated memory. Brain Res Bull 185:64–73. https://doi.org/10.1016/j.brainresbull.2022.04.010
Chafee MV, Heilbronner SR (2022) Prefrontal cortex. Curr Biol 32(8):R346–R351. https://doi.org/10.1016/j.cub.2022.02.071
Volkow ND, Michaelides M, Baler R (2019) The neuroscience of drug reward and addiction. Physiol Rev 99(4):2115–2140. https://doi.org/10.1152/physrev.00014.2018
Yang X, Wen Y, Zhang Y, Gao F, Yang J, Yang Z, Yan C (2020) Dynamic changes of cytoskeleton-related proteins within reward-related brain regions in morphine-associated memory. Front Neurosci 14:626348. https://doi.org/10.3389/fnins.2020.626348
Franklin K, Paxinos G, Keith B (2008) The mouse brain in stereotaxic coordinates. Rat Brain in Stereotaxic Coordinates 3(2):6
Pertea M, Kim D, Pertea GM, Leek JT, Salzberg SL (2016) Transcript-level expression analysis of RNA-seq experiments with HISAT. StringTie and Ballgown Nat Protoc 11(9):1650–1667. https://doi.org/10.1038/nprot.2016.095
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11(10):R106. https://doi.org/10.1186/gb-2010-11-10-r106
Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL (2013) TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol 14(4):R36. https://doi.org/10.1186/gb-2013-14-4-r36
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S et al (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36(Database issue):D480–D484
Liberzon A, Birger C, Thorvaldsdóttir H, Ghandi M, Mesirov JP, Tamayo P (2015) The molecular signatures database (MSigDB) hallmark gene set collection. Cell Syst 1(6):417–425
Zhong S, Feng J (2022) CircPrimer 2.0: a software for annotating circRNAs and predicting translation potential of circRNAs. BMC Bioinformatics 23(1):215. https://doi.org/10.1186/s12859-022-04705-y
Enright AJ, John B, Gaul U, Tuschl T, Sander C, Marks DS (2003) MicroRNA targets in Drosophila. Genome Biol 5(1):R1
Krüger J, Rehmsmeier M (2006) RNAhybrid: microRNA target prediction easy, fast and flexible. Nucleic Acids Res 34(Web Server issue):W451–W454
Agarwal V, Bell GW, Nam J-W, Bartel DP (2015) Predicting effective microRNA target sites in mammalian mRNAs. Elife 4. https://doi.org/10.7554/eLife.05005
Sticht C, De La Torre C, Parveen A, Gretz N (2018) miRWalk: an online resource for prediction of microRNA binding sites. PLoS ONE 13(10):e0206239. https://doi.org/10.1371/journal.pone.0206239
Gruber AR, Lorenz R, Bernhart SH, Neuböck R, Hofacker IL (2008) The Vienna RNA websuite. Nucleic Acids Res 36(Web Server issue):W70–W74. https://doi.org/10.1093/nar/gkn188
Doyle TM, Hutchinson MR, Braden K, Janes K, Staikopoulos V, Chen Z, Neumann WL, Spiegel S et al (2020) Sphingosine-1-phosphate receptor subtype 1 activation in the central nervous system contributes to morphine withdrawal in rodents. J Neuroinflammation 17(1):314. https://doi.org/10.1186/s12974-020-01975-2
Wang Y-J, Yu C, Wu W-W, Ju Y-Y, Liu Y, Xu C, Long J-D, Zan G-Y et al (2021) Alteration of twinfilin1 expression underlies opioid withdrawal-induced remodeling of actin cytoskeleton at synapses and formation of aversive memory. Mol Psychiatry 26(11):6218–6236. https://doi.org/10.1038/s41380-021-01111-3
Ferrer-Alcón M, García-Fuster MJ, La Harpe R, García-Sevilla JA (2004) Long-term regulation of signalling components of adenylyl cyclase and mitogen-activated protein kinase in the pre-frontal cortex of human opiate addicts. J Neurochem 90(1):220–230
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102(43):15545–15550
Kauer JA, Malenka RC (2007) Synaptic plasticity and addiction. Nat Rev Neurosci 8(11):844–858
You X, Vlatkovic I, Babic A, Will T, Epstein I, Tushev G, Akbalik G, Wang M et al (2015) Neural circular RNAs are derived from synaptic genes and regulated by development and plasticity. Nat Neurosci 18(4):603–610. https://doi.org/10.1038/nn.3975
Liu Z, Ran Y, Tao C, Li S, Chen J, Yang E (2019) Detection of circular RNA expression and related quantitative trait loci in the human dorsolateral prefrontal cortex. Genome Biol 20(1):99. https://doi.org/10.1186/s13059-019-1701-8
Fernández-Chacón R, Königstorfer A, Gerber SH, García J, Matos MF, Stevens CF, Brose N, Rizo J et al (2001) Synaptotagmin I functions as a calcium regulator of release probability. Nature 410(6824):41–49
Barbier E, Barchiesi R, Domi A, Chanthongdee K, Domi E, Augier G, Augier E, Xu L et al (2021) Downregulation of synaptotagmin 1 in the prelimbic cortex drives alcohol-associated behaviors in rats. Biol Psychiatry 89(4):398–406. https://doi.org/10.1016/j.biopsych.2020.08.027
Viola TW, Schuch JB, Rovaris DL, Genovese R, Tondo L, Sanvicente-Vieira B, Zaparte A, Cupertino RB et al (2019) Association between cognitive performance and SYT1-rs2251214 among women with cocaine use disorder. J Neural Transm (Vienna) 126(12):1707–1711. https://doi.org/10.1007/s00702-019-02086-w
Ba W, Selten MM, van der Raadt J, van Veen H, Li LL, Benevento M, Oudakker AR, Lasabuda RSE et al (2016) ARHGAP12 functions as a developmental brake on excitatory synapse function. Cell Rep 14(6):1355–1368. https://doi.org/10.1016/j.celrep.2016.01.037
Williams JT, Christie MJ, Manzoni O (2001) Cellular and synaptic adaptations mediating opioid dependence. Physiol Rev 81(1):299–343
Hearing MC, Jedynak J, Ebner SR, Ingebretson A, Asp AJ, Fischer RA, Schmidt C, Larson EB et al (2016) Reversal of morphine-induced cell-type-specific synaptic plasticity in the nucleus accumbens shell blocks reinstatement. Proc Natl Acad Sci U S A 113(3):757–762. https://doi.org/10.1073/pnas.1519248113
Agrawal A, Lynskey MT (2009) Candidate genes for cannabis use disorders: findings, challenges and directions. Addiction 104(4):518–532. https://doi.org/10.1111/j.1360-0443.2009.02504.x
Fu R, Tang Y, Li W, Ren Z, Li D, Zheng J, Zuo W, Chen X et al (2021) Endocannabinoid signaling in the lateral habenula regulates pain and alcohol consumption. Transl Psychiatry 11(1):220. https://doi.org/10.1038/s41398-021-01337-3
Han W, Li J, Pelkey KA, Pandey S, Chen X, Wang Y-X, Wu K, Ge L et al (2019) Shisa7 is a GABA receptor auxiliary subunit controlling benzodiazepine actions. Science 366(6462):246–250. https://doi.org/10.1126/science.aax5719
Schmitz LJM, Klaassen RV, Ruiperez-Alonso M, Zamri AE, Stroeder J, Rao-Ruiz P, Lodder JC, van der Loo RJ et al (2017) The AMPA receptor-associated protein Shisa7 regulates hippocampal synaptic function and contextual memory. Elife 6. https://doi.org/10.7554/eLife.24192
Sabaie H, Talebi M, Gharesouarn J, Asadi MR, Jalaiei A, Arsang-Jang S, Hussen BM, Taheri M et al (2022) Identification and analysis of BCAS4/hsa-miR-185-5p/SHISA7 competing endogenous RNA axis in late-onset Alzheimer’s disease using bioinformatic and experimental approaches. Front Aging Neurosci 14:812169. https://doi.org/10.3389/fnagi.2022.812169
Sabaie H, Gharesouran J, Asadi MR, Farhang S, Ahangar NK, Brand S, Arsang-Jang S, Dastar S et al (2022) Downregulation of miR-185 is a common pathogenic event in 22q11.2 deletion syndrome-related and idiopathic schizophrenia. Metab Brain Dis 37(4):1175–1184. https://doi.org/10.1007/s11011-022-00918-5
Jahn R, Scheller RH (2006) SNAREs–engines for membrane fusion. Nat Rev Mol Cell Biol 7(9):631–643
Chen F, Chen H, Chen Y, Wei W, Sun Y, Zhang L, Cui L, Wang Y (2021) Dysfunction of the SNARE complex in neurological and psychiatric disorders. Pharmacol Res 165:105469. https://doi.org/10.1016/j.phrs.2021.105469
Ramos-Miguel A, Beasley CL, Dwork AJ, Mann JJ, Rosoklija G, Barr AM, Honer WG (2015) Increased SNARE protein-protein interactions in orbitofrontal and anterior cingulate cortices in schizophrenia. Biol Psychiatry 78(6):361–373. https://doi.org/10.1016/j.biopsych.2014.12.012
Das J (2020) SNARE complex-associated proteins and alcohol. Alcohol Clin Exp Res 44(1). https://doi.org/10.1111/acer.14238
Neasta J, Barak S, Hamida SB, Ron D (2014) mTOR complex 1: a key player in neuroadaptations induced by drugs of abuse. J Neurochem 130(2):172–184. https://doi.org/10.1111/jnc.12725
Ucha M, Roura-Martínez D, Ambrosio E, Higuera-Matas A (2020) The role of the mTOR pathway in models of drug-induced reward and the behavioural constituents of addiction. J Psychopharmacol 34(11):1176–1199. https://doi.org/10.1177/0269881120944159
Hansen TB, Jensen TI, Clausen BH, Bramsen JB, Finsen B, Damgaard CK, Kjems J (2013) Natural RNA circles function as efficient microRNA sponges. Nature 495(7441):384–388. https://doi.org/10.1038/nature11993
Beveridge NJ, Gardiner E, Carroll AP, Tooney PA, Cairns MJ (2010) Schizophrenia is associated with an increase in cortical microRNA biogenesis. Mol Psychiatry 15(12):1176–1189. https://doi.org/10.1038/mp.2009.84
Hollins SL, Zavitsanou K, Walker FR, Cairns MJ (2014) Alteration of imprinted Dlk1-Dio3 miRNA cluster expression in the entorhinal cortex induced by maternal immune activation and adolescent cannabinoid exposure. Transl Psychiatry 4(9):e452. https://doi.org/10.1038/tp.2014.99
Krol J, Loedige I, Filipowicz W (2010) The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet 11(9):597–610. https://doi.org/10.1038/nrg2843
Mahmoudi E, Cairns MJ (2017) MiR-137: an important player in neural development and neoplastic transformation. Mol Psychiatry 22(1):44–55. https://doi.org/10.1038/mp.2016.150
Vickstrom CR, Liu X, Liu S, Hu M-M, Mu L, Hu Y, Yu H, Love SL et al (2021) Role of endocannabinoid signaling in a septohabenular pathway in the regulation of anxiety- and depressive-like behavior. Mol Psychiatry 26(7):3178–3191. https://doi.org/10.1038/s41380-020-00905-1
Dever TE, Gutierrez E, Shin B-S (2014) The hypusine-containing translation factor eIF5A. Crit Rev Biochem Mol Biol 49(5):413–425. https://doi.org/10.3109/10409238.2014.939608
Schroeder S, Hofer SJ, Zimmermann A, Pechlaner R, Dammbrueck C, Pendl T, Marcello GM, Pogatschnigg V et al (2021) Dietary spermidine improves cognitive function. Cell Rep 35(2):108985. https://doi.org/10.1016/j.celrep.2021.108985
Huang Y, Higginson DS, Hester L, Park MH, Snyder SH (2007) Neuronal growth and survival mediated by eIF5A, a polyamine-modified translation initiation factor. Proc Natl Acad Sci USA 104(10):4194–4199
Kar RK, Hanner AS, Starost MF, Springer D, Mastracci TL, Mirmira RG, Park MH (2021) Neuron-specific ablation of eIF5A or deoxyhypusine synthase leads to impairments in growth, viability, neurodevelopment, and cognitive functions in mice. J Biol Chem 297(5):101333. https://doi.org/10.1016/j.jbc.2021.101333
Gunasekaran S, Jacob RS, Omkumar RV (2022) Differential expression of miR-148b, miR-129-2 and miR-296 in animal models of schizophrenia-Relevance to NMDA receptor hypofunction. Neuropharmacology 210:109024. https://doi.org/10.1016/j.neuropharm.2022.109024
Ru X, Qu J, Li Q, Zhou J, Huang S, Li W, Zuo S, Chen Y et al (2021) MiR-706 alleviates white matter injury via downregulating PKCα/MST1/NF-κB pathway after subarachnoid hemorrhage in mice. Exp Neurol 341:113688. https://doi.org/10.1016/j.expneurol.2021.113688
Sun Z, Yu J-T, Jiang T, Li M-M, Tan L, Zhang Q, Tan L (2013) Genome-wide microRNA profiling of rat hippocampus after status epilepticus induced by amygdala stimulation identifies modulators of neuronal apoptosis. PLoS ONE 8(10):e78375. https://doi.org/10.1371/journal.pone.0078375
Zou T, Zhang J, Liu Y, Zhang Y, Sugimoto K, Mei C (2020) Antidepressant-like effect of in mice exposed to a chronic mild stress involves the microRNA-298-5p-mediated Nox1. Front Mol Neurosci 13:131. https://doi.org/10.3389/fnmol.2020.00131
Vallelunga A, Iannitti T, Capece S, Somma G, Russillo MC, Foubert-Samier A, Laurens B, Sibon I et al (2021) Serum miR-96-5P and miR-339-5P are potential biomarkers for multiple system atrophy and Parkinson’s disease. Front Aging Neurosci 13:632891. https://doi.org/10.3389/fnagi.2021.632891
Dhiraj DK, Chrysanthou E, Mallucci GR, Bushell M (2013) miRNAs-19b, -29b-2* and -339-5p show an early and sustained up-regulation in ischemic models of stroke. PLoS ONE 8(12):e83717. https://doi.org/10.1371/journal.pone.0083717
Long JM, Ray B, Lahiri DK (2014) MicroRNA-339-5p down-regulates protein expression of β-site amyloid precursor protein-cleaving enzyme 1 (BACE1) in human primary brain cultures and is reduced in brain tissue specimens of Alzheimer disease subjects. J Biol Chem 289(8):5184–5198. https://doi.org/10.1074/jbc.M113.518241
Zou H-Y, Guo L, Zhang B, Chen S, Wu X-R, Liu X-D, Xu X-Y, Li B-Y et al (2022) Aberrant miR-339–5p/neuronatin signaling causes prodromal neuronal calcium dyshomeostasis in mutant presenilin mice. J Clin Invest 132(8). https://doi.org/10.1172/JCI149160
Zhang H, Deng J, Huang K, He Y, Cai Z, He Y (2022) circNup188/miR-760-3p/Map3k8 axis regulates inflammation in cerebral ischemia. Mol Cell Probes 64:101830. https://doi.org/10.1016/j.mcp.2022.101830
Chen D, Lan G, Li R, Mei Y, Shui X, Gu X, Wang L, Zhang T et al (2022) Melatonin ameliorates tau-related pathology via the miR-504-3p and CDK5 axis in Alzheimer’s disease. Transl Neurodegener 11(1):27. https://doi.org/10.1186/s40035-022-00302-4
Massaly N, Dahan L, Baudonnat M, Hovnanian C, Rekik K, Solinas M, David V, Pech S et al (2013) Involvement of protein degradation by the ubiquitin proteasome system in opiate addictive behaviors. Neuropsychopharmacology 38(4):596–604. https://doi.org/10.1038/npp.2012.217
Massaly N, Francès B, Moulédous L (2014) Roles of the ubiquitin proteasome system in the effects of drugs of abuse. Front Mol Neurosci 7:99. https://doi.org/10.3389/fnmol.2014.00099
Werner CT, Viswanathan R, Martin JA, Gobira PH, Mitra S, Thomas SA, Wang Z-J, Liu J-F et al (2018) E3 ubiquitin-protein ligase SMURF1 in the nucleus accumbens mediates cocaine seeking. Biol Psychiatry 84(12):881–892. https://doi.org/10.1016/j.biopsych.2018.07.013
Zhou Y, Ge Y, Liu Q, Li Y-X, Chao X, Guan J-J, Diwu Y-C, Zhang Q (2021) LncRNA BACE1-AS promotes autophagy-mediated neuronal damage through the miR-214-3p/ATG5 signalling axis in Alzheimer’s disease. Neuroscience 455:52–64. https://doi.org/10.1016/j.neuroscience.2020.10.028
Irie K, Tsujimura K, Nakashima H, Nakashima K (2016) MicroRNA-214 promotes dendritic development by targeting the schizophrenia-associated gene quaking (Qki). J Biol Chem 291(26):13891–13904. https://doi.org/10.1074/jbc.M115.705749
Zhang M, Zhou H, He R, Yang J, Zou Y, Deng Y, Xie H, Yan Z (2022) Up-regulating microRNA-214-3p relieves hypoxic-ischemic brain damage through inhibiting TXNIP expression. Mol Cell Biochem. https://doi.org/10.1007/s11010-022-04530-0
Chavoshi H, Boroujeni ME, Abdollahifar M-A, Amini A, Tehrani AM, Moghaddam MH, Norozian M, Farahani RM et al (2020) From dysregulated microRNAs to structural alterations in the striatal region of METH-injected rats. J Chem Neuroanat 109:101854. https://doi.org/10.1016/j.jchemneu.2020.101854
Funding
This research was supported by the National Natural Science Foundation of China (81971792, and 81901920) and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
CXY and YXZ designed the study. XXY performed the experiment and bioinformatics analysis. DYY, FFG, JSY and ZNC analysed the data. JLL, XYY and LJL checked the data. XXY wrote the manuscript. CXY and YXZ revised the manuscript. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Animal experiments were approved ethically by the Institutional Animal Care and Use Committee of Xi’an Jiaotong University (No. 2022–1174).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, X., Yu, D., Gao, F. et al. Integrative Analysis of Morphine-Induced Differential Circular RNAs and ceRNA Networks in the Medial Prefrontal Cortex. Mol Neurobiol (2023). https://doi.org/10.1007/s12035-023-03859-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-023-03859-x