Abstract
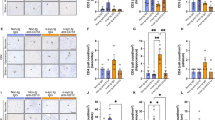
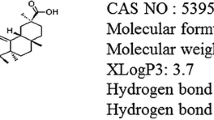
The increased α-synuclein (α-syn)-dependent activation of CD4 T cells leads to the progressive loss of dopaminergic (DA) neurons in the substantia nigra (SN) in Parkinson’s disease (PD). Astragaloside IV (AS-IV) protects DA neurons against neuroinflammation. The effects of AS-IV on CD4 T-cell-mediated immune responses in PD remain unknown. Rotenone (ROT) injected unilaterally into the substantia nigra pars compacta (SNc) of rats induced PD. AS-IV (20 mg/kg) was intraperitoneally injected once a day for 14 days. The limb hanging test and rotarod test were performed to evaluate the alteration of behavior at 4 and 6 weeks. Total gastrointestinal transit tests were performed at 4 weeks. Western blotting was used to detect the expression of proinflammatory cytokine proteins. Immunofluorescence staining was conducted to test the expression and localization of major histocompatibility complex class II (MHCII), cleaved caspase-1 and α-syn in astrocytes. Flow cytometry analysis, immunohistochemistry and immunofluorescence staining were used to measure the expression of CD4 T-cell subsets in the SN. The application of AS-IV protected against the loss of DA neurons and behavioral deficits in ROT-induced PD rat models. AS-IV administration inhibited the aggregation of α-syn in DA neurons and the expression of proinflammatory cytokines such as TNF-α, IL-18, IL-6 and IL-1β. AS-IV decreased the activation of CD4 T cells and three CD4 T-cell subsets: Tfh, Treg and Th1. AS-IV interrupted the ROT-induced interaction between astrocytes and CD4 T cells and the colocalization of MHCII and α-syn in astrocytes. AS-IV inhibited the expression of α-syn in astrocytes and the colocalization of α-syn and cleaved caspase-1 in astrocytes. AS-IV prevents the loss of DA neurons in PD by inhibiting the activation of α-syn-specific CD4 T cells, which is regulated by MHCII-mediated antigen presentation in astrocytes.








Similar content being viewed by others
Data Availability
The data used during the current study are available from the corresponding author on reasonable request.
References
Marogianni C, Sokratous M, Dardiotis E, Hadjigeorgiou GM, Bogdanos D, Xiromerisiou G (2020) Neurodegeneration and inflammation-an interesting interplay in Parkinson’s disease. Int J Mol Sci 21:8421
Xu Y, Li Y, Wang C, Han T, Liu H, Sun L, Hong J, Hashimoto M et al (2023) The reciprocal interactions between microglia and T cells in Parkinson’s disease: a double-edged sword. J Neuroinflammation 20:33
Benner EJ, Banerjee R, Reynolds AD, Sherman S, Pisarev VM, Tsiperson V, Nemachek C, Ciborowski P et al (2008) Nitrated alpha-synuclein immunity accelerates degeneration of nigral dopaminergic neurons. PLoS ONE 3:e1376
Costa IM, Lima FOV, Fernandes LCB, Norrara B, Neta FI, Alves RD, Cavalcanti J, Lucena EES et al (2019) Astragaloside IV supplementation promotes A neuroprotective effect in experimental models of neurological disorders: a systematic review. Curr Neuropharmacol 17:648–665
Zhong Y, Liu W, Xiong Y, Li Y, Wan Q, Zhou W, Zhao H, Xiao Q et al (2022) Astragaloside IV alleviates ulcerative colitis by regulating the balance of Th17/Treg cells. Phytomedicine 104:154287
Yang L, Xing F, Han X, Li Q, Wu H, Shi H, Wang Z, Huang F et al (2019) Astragaloside IV regulates differentiation and induces apoptosis of activated CD4(+) T cells in the pathogenesis of experimental autoimmune encephalomyelitis. Toxicol Appl Pharmacol 362:105–115
Zhang A, Zheng Y, Que Z, Zhang L, Lin S, Le V, Liu J, Tian J (2014) Astragaloside IV inhibits progression of lung cancer by mediating immune function of Tregs and CTLs by interfering with IDO. J Cancer Res Clin Oncol 140:1883–1890
Wang T, Shi C, Luo H, Zheng H, Fan L, Tang M, Su Y, Yang J et al (2022) Neuroinflammation in Parkinson’s disease: triggers, mechanisms, and immunotherapies. Neuroscientist 28:364–381
Choi I, Heaton GR, Lee Y-K, Yue Z (2022) Regulation of α-synuclein homeostasis and inflammasome activation by microglial autophagy. Sci Adv 8:eabn1298
Sofroniew MV (2020) Astrocyte reactivity: subtypes, states, and functions in CNS innate immunity. Trends Immunol 41:758–770
Rostami J, Fotaki G, Sirois J, Mzezewa R, Bergstrom J, Essand M, Healy L, Erlandsson A (2020) Astrocytes have the capacity to act as antigen-presenting cells in the Parkinson’s disease brain. J Neuroinflammation 17:119
Williams GP, Schonhoff AM, Jurkuvenaite A, Gallups NJ, Standaert DG, Harms AS (2021) CD4 T cells mediate brain inflammation and neurodegeneration in a mouse model of Parkinson’s disease. Brain 144:2047–2059
LindestamArlehamn CS, Dhanwani R, Pham J, Kuan R, Frazier A, Rezende Dutra J, Phillips E, Mallal S et al (2020) α-Synuclein-specific T cell reactivity is associated with preclinical and early Parkinson’s disease. Nat Commun 11:1875
Contaldi E, Magistrelli L, Comi C (2022) T lymphocytes in Parkinson’s disease. J Parkinsons Dis 12:S65–S74
Garretti F, Agalliu D, LindestamArlehamn CS, Sette A, Sulzer D (2019) Autoimmunity in Parkinson’s disease: the role of alpha-Synuclein-Specific T cells. Front Immunol 10:303
Yang L, Han X, Yuan J, Xing F, Hu Z, Huang F, Wu H, Shi H et al (2020) Early astragaloside IV administration attenuates experimental autoimmune encephalomyelitis in mice by suppressing the maturation and function of dendritic cells. Life Sci 249:117448
Wan C-P, Gao L-X, Hou L-F, Yang X-Q, He P-L, Yang Y-F, Tang W, Yue J-M et al (2013) Astragaloside II triggers T cell activation through regulation of CD45 protein tyrosine phosphatase activity. Acta Pharmacol Sin 34:522–530
Ma L, Yang C, Zhang X, Li Y, Wang S, Zheng L, Huang K (1864) C-terminal truncation exacerbates the aggregation and cytotoxicity of α-Synuclein: A vicious cycle in Parkinson’s disease. Biochim Biophys Acta Mol Basis Dis 2018:3714–3725
Wu S, Wen F, Zhong X, Du W, Chen M, Wang J (2023) Astragaloside IV ameliorate acute alcohol-induced liver injury in mice via modulating gut microbiota and regulating NLRP3/caspase-1 signaling pathway. Ann Med 55:2216942
Xiong N, Huang J, Zhang Z, Zhang Z, Xiong J, Liu X, Jia M, Wang F et al (2009) Stereotaxical infusion of rotenone: a reliable rodent model for Parkinson’s disease. PLoS ONE 4:e7878
Möller K, Stahl T, Boltze J, Wagner D-C (2012) Isolation of inflammatory cells from rat brain tissue after stroke. Exp Transl Stroke Med 4:20
Ahn H, Kim J, Jeung E-B, Lee G-S (2014) Dimethyl sulfoxide inhibits NLRP3 inflammasome activation. Immunobiology 219:315–322
Galvao J, Davis B, Tilley M, Normando E, Duchen MR, Cordeiro MF (2014) Unexpected low-dose toxicity of the universal solvent DMSO. FASEB J 28:1317–1330
Kunkl M, Amormino C, Tedeschi V, Fiorillo MT, Tuosto L (2022) Astrocytes and inflammatory T helper cells: a dangerous liaison in multiple sclerosis. Front Immunol 13:824411
Schetters STT, Gomez-Nicola D, Garcia-Vallejo JJ, Van Kooyk Y (2017) Neuroinflammation: microglia and T cells get ready to tango. Front Immunol 8:1905
Zhu J, Paul WE (2008) CD4 T cells: fates, functions, and faults. Blood 112:1557–1569
Zhang X, Ge R, Chen H, Ahiafor M, Liu B, Chen J, Fan X (2021) Follicular helper CD4+ T cells, follicular regulatory CD4+ T cells, and inducible costimulator and their roles in multiple sclerosis and experimental autoimmune encephalomyelitis. Mediators Inflamm 2021:2058964
Voet S, Srinivasan S, Lamkanfi M, van Loo G (2019) Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol Med 11:e10248
Mata-Martínez E, Díaz-Muñoz M, Vázquez-Cuevas FG (2022) Glial cells and brain diseases: inflammasomes as relevant pathological entities. Front Cell Neurosci 16:929529
Martynova E, Rizvanov A, Urbanowicz RA, Khaiboullina S (2022) Inflammasome contribution to the activation of Th1, Th2, and Th17 immune responses. Front Microbiol 13:851835
Sugama S, Wirz SA, Barr AM, Conti B, Bartfai T, Shibasaki T (2004) Interleukin-18 null mice show diminished microglial activation and reduced dopaminergic neuron loss following acute 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine treatment. Neuroscience 128:451–458
Wang W, Nguyen LTT, Burlak C, Chegini F, Guo F, Chataway T, Ju S, Fisher OS et al (2016) Caspase-1 causes truncation and aggregation of the Parkinson’s disease-associated protein α-synuclein. Proc Natl Acad Sci U S A 113:9587–9592
Terada M, Suzuki G, Nonaka T, Kametani F, Tamaoka A, Hasegawa M (2018) The effect of truncation on prion-like properties of α-synuclein. J Biol Chem 293:13910–13920
Xiao L, Dai Z, Tang W, Liu C, Tang B (2021) Astragaloside IV alleviates cerebral ischemia-reperfusion injury through NLRP3 inflammasome-mediated pyroptosis inhibition via activating Nrf2. Oxid Med Cell Longev 2021:9925561
Zhu X, Chen Y, Du Y, Wan Q, Xu Y, Wu J (2018) Astragaloside IV attenuates penicillin-induced epilepsy via inhibiting activation of the MAPK signaling pathway. Mol Med Rep 17:643–647
Qu YZ, Li M, Zhao YL, Zhao ZW, Wei XY, Liu JP, Gao L, Gao GD (2009) Astragaloside IV attenuates cerebral ischemia-reperfusion-induced increase in permeability of the blood-brain barrier in rats. Eur J Pharmacol 606:137–141
Yang Y, Hong M, Lian W-W, Chen Z (2022) Review of the pharmacological effects of astragaloside IV and its autophagic mechanism in association with inflammation. World J Clin Cases 10:10004–10016
Yu S, Peng W, Qiu F, Zhang G (2022) Research progress of astragaloside IV in the treatment of atopic diseases. Biomed Pharmacother 156:113989
Acknowledgements
The authors are grateful to Dr. Wang hongcai for technical support. We thank Medical Research Center of Binzhou Medical University Hospital for providing the high-precision experimental instruments.
Funding
This work was supported by grants from the National Natural Science Foundation of China (NSFC) (81601108), Natural Science Foundation of Shandong Province of China (ZR2016HQ14 and ZR2021MH135).
Author information
Authors and Affiliations
Contributions
Mengdi Wang and Hongcai Wang conceived and designed research. Mengdi Wang, Fengjiao Sun, Xiaofeng Han, Nan Wang, Yalan Liu, Jinfeng Cai, Shanshan Tong and Rui Wang performed the experiments. Mengdi Wang, Yalan Liu, Jinfeng Cai and Shanshan Tong analyzed and interpreted the data. Mengdi Wang and Hongcai Wang wrote the manuscript, which was read, edited, and approved by all the authors. Mengdi Wang, Fengjiao Sun, Nan Wang and Hongcai Wang contributed reagents, materials, and analysis tools. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All animal procedures were approved by the guidelines of the ethical standards for laboratory animals of Binzhou Medical University Hospital.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Sun, F., Han, X. et al. Astragaloside IV Inhibits Rotenone-Induced α-syn Presentation and the CD4 T-Cell Immune Response. Mol Neurobiol 61, 252–265 (2024). https://doi.org/10.1007/s12035-023-03566-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03566-7