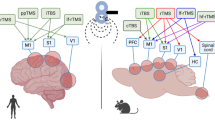
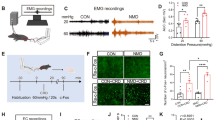
Abstract
Emerging evidence suggest that parvalbumin neurons in zona incerta (ZI) modulate pain and itch behavior in opposite manners. However, the role of ZI glutamatergic neurons, a unique incertal neuronal subpopulation residing in the caudal division, in pain and itch modulation remains unknown. In the present study, by combining chemogenetic manipulation, fiber photometry, and behavioral tests, we proved that incertal glutamatergic neurons served as an endogenous negative diencephalic modulator for both pain and itch processing. We demonstrated that ZI vesicular glutamate transporter 2 (VGluT2) neurons exhibited increased calcium signal upon hindpaw withdrawal in response to experimental mechanical and thermal stimuli. Behavioral tests further showed that pharmacogenetic activation of this specific type of neurons reduced nocifensive withdrawal responses in both naïve and inflammatory pain mice. Similar neural activity and modulatory role of ZI VGluT2 neurons were also observed upon histaminergic and non-histaminergic acute itch stimuli. Together, our study would expedite our understandings of brain mechanisms underlying somatosensory processing and modulation, and supply a novel therapeutic target for the management of chronic pain and itch disorders.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Wang X, Chou XL, Zhang LI, Tao HW (2020) Zona Incerta: an integrative node for global behavioral modulation. Trends Neurosci 43(2):82–87. https://doi.org/10.1016/j.tins.2019.11.007
Zhou M, Liu Z, Melin MD, Ng YH, Xu W, Südhof TC (2018) A central amygdala to zona incerta projection is required for acquisition and remote recall of conditioned fear memory. Nat Neurosci 21(11):1515–1519. https://doi.org/10.1038/s41593-018-0248-4
Li Z, Rizzi G, Tan KR (2021) Zona incerta subpopulations differentially encode and modulate anxiety. Sci Adv 7(37):eabf6709. https://doi.org/10.1126/sciadv.abf6709
Zhao ZD, Chen Z, Xiang X, Hu M, Xie H, Jia X, Cai F, Cui Y, Chen Z, Qian L, Liu J, Shang C, Yang Y, Ni X, Sun W, Hu J, Cao P, Li H, Shen WL (2019) Zona incerta GABAergic neurons integrate prey-related sensory signals and induce an appetitive drive to promote hunting. Nat Neurosci 22(6):921–932. https://doi.org/10.1038/s41593-019-0404-5
Wang X, Chou X, Peng B, Shen L, Huang JJ, Zhang LI, Tao HW (2019) A cross-modality enhancement of defensive flight via parvalbumin neurons in zona incerta. eLife 8. https://doi.org/10.7554/eLife.42728
Chou XL, Wang X, Zhang ZG, Shen L, Zingg B, Huang J, Zhong W, Mesik L, Zhang LI, Tao HW (2018) Inhibitory gain modulation of defense behaviors by zona incerta. Nat Commun 9(1):1151. https://doi.org/10.1038/s41467-018-03581-6
Liu K, Kim J, Kim DW, Zhang YS, Bao H, Denaxa M, Lim SA, Kim E, Liu C, Wickersham IR, Pachnis V, Hattar S, Song J, Brown SP, Blackshaw S (2017) Lhx6-positive GABA-releasing neurons of the zona incerta promote sleep. Nature 548(7669):582–587. https://doi.org/10.1038/nature23663
Wang H, Dong P, He C, Feng XY, Huang Y, Yang WW, Gao HJ, Shen XF, Lin S, Cao SX, Lian H, Chen J, Yan M, Li XM (2020) Incerta-thalamic Circuit Controls Nocifensive Behavior via Cannabinoid Type 1 receptors. Neuron 107(3):538–551e537. https://doi.org/10.1016/j.neuron.2020.04.027
Mitrofanis J (2005) Some certainty for the “zone of uncertainty”? Exploring the function of the zona incerta. Neuroscience 130(1):1–15. https://doi.org/10.1016/j.neuroscience.2004.08.017
Li J, Bai Y, Liang Y, Zhang Y, Zhao Q, Ge J, Li D, Zhu Y, Cai G, Tao H, Wu S, Huang J (2022) Parvalbumin neurons in Zona Incerta regulate itch in mice. Front Mol Neurosci 15:843754. https://doi.org/10.3389/fnmol.2022.843754
McCarson KE, Fehrenbacher JC (2021) Models of inflammation: Carrageenan- or complete Freund’s adjuvant (CFA)-Induced Edema and Hypersensitivity in the rat. Curr protocols 1(7):e202. https://doi.org/10.1002/cpz1.202
Muley MM, Krustev E, McDougall JJ (2016) Preclinical Assessment of Inflammatory Pain. CNS Neurosci Ther 22(2):88–101. https://doi.org/10.1111/cns.12486
Zhu X, He X, Liu Y, Wen P, Wang L, Zhang Z, Xu F (2018) A convenient semi-automatic method for analyzing brain sections: Registration, Segmentation and Cell counting. Chin J Magn Reson 35(2):133–140. https://doi.org/10.11938/cjmr20172608
Li LX, Li YL, Wu JT, Song JZ, Li XM (2022) Glutamatergic neurons in the Caudal Zona Incerta regulate Parkinsonian Motor symptoms in mice. Neurosci Bull 38(1):1–15. https://doi.org/10.1007/s12264-021-00775-9
Ma Q (2010) Labeled lines meet and talk: population coding of somatic sensations. J Clin Investig 120(11):3773–3778. https://doi.org/10.1172/jci43426
LaMotte RH, Dong X, Ringkamp M (2014) Sensory neurons and circuits mediating itch. Nat Rev Neurosci 15(1):19–31. https://doi.org/10.1038/nrn3641
Davidson S, Giesler GJ (2010) The multiple pathways for itch and their interactions with pain. Trends Neurosci 33(12):550–558. https://doi.org/10.1016/j.tins.2010.09.002
Samineni VK, Grajales-Reyes JG, Copits BA, O’Brien DE, Trigg SL, Gomez AM, Bruchas MR, Gereau RWt (2017) Divergent modulation of nociception by glutamatergic and GABAergic neuronal subpopulations in the Periaqueductal Gray. eNeuro 4(2). https://doi.org/10.1523/eneuro.0129-16.2017
Samineni VK, Grajales-Reyes JG, Sundaram SS, Yoo JJ, Gereau RWt (2019) Cell type-specific modulation of sensory and affective components of itch in the periaqueductal gray. Nat Commun 10(1):4356. https://doi.org/10.1038/s41467-019-12316-0
Liu Y, Latremoliere A, Li X, Zhang Z, Chen M, Wang X, Fang C, Zhu J, Alexandre C, Gao Z, Chen B, Ding X, Zhou JY, Zhang Y, Chen C, Wang KH, Woolf CJ, He Z (2018) Touch and tactile neuropathic pain sensitivity are set by corticospinal projections. Nature 561(7724):547–550. https://doi.org/10.1038/s41586-018-0515-2
Wu ZH, Shao HY, Fu YY, Wu XB, Cao DL, Yan SX, Sha WL, Gao YJ, Zhang ZJ (2021) Descending modulation of spinal itch transmission by primary somatosensory cortex. Neurosci Bull 37(9):1345–1350. https://doi.org/10.1007/s12264-021-00713-9
Nguyen E, Smith KM, Cramer N, Holland RA, Bleimeister IH, Flores-Felix K, Silberberg H, Keller A, Le Pichon CE, Ross SE (2022) Medullary kappa-opioid receptor neurons inhibit pain and itch through a descending circuit. Brain 145(7):2586–2601. https://doi.org/10.1093/brain/awac189
Li JN, Ren JH, He CB, Zhao WJ, Li H, Dong YL, Li YQ (2021) Projections from the lateral parabrachial nucleus to the lateral and ventral lateral periaqueductal gray subregions mediate the itching sensation. Pain 162(6):1848–1863. https://doi.org/10.1097/j.pain.0000000000002193
Liu PF, Wang Y, Xu L, Xiang AF, Liu MZ, Zhu YB, Jia X, Zhang R, Li JB, Zhang L, Mu D (2022) Modulation of itch and pain signals processing in ventrobasal thalamus by thalamic reticular nucleus. iScience 25(1):103625. https://doi.org/10.1016/j.isci.2021.103625
Yang Y, Jiang T, Jia X, Yuan J, Li X, Gong H (2022) Whole-brain connectome of GABAergic neurons in the mouse Zona Incerta. Neurosci Bull. https://doi.org/10.1007/s12264-022-00930-w
Casas-Torremocha D, Rubio-Teves M, Hoerder-Suabedissen A, Hayashi S, Prensa L, Molnár Z, Porrero C, Clasca F (2022) A combinatorial input landscape in the “higher-order relay” posterior thalamic nucleus. J neuroscience: official J Soc Neurosci 42(41):7757–7781. https://doi.org/10.1523/jneurosci.0698-22.2022
Beitz AJ (1989) Possible origin of glutamatergic projections to the midbrain periaqueductal gray and deep layer of the superior colliculus of the rat. Brain Res Bull 23(1–2):25–35. https://doi.org/10.1016/0361-9230(89)90159-7
Ziegler DR, Edwards MR, Ulrich-Lai YM, Herman JP, Cullinan WE (2012) Brainstem origins of glutamatergic innervation of the rat hypothalamic paraventricular nucleus. J Comp Neurol 520(11):2369–2394. https://doi.org/10.1002/cne.23043
Luan Y, Tang D, Wu H, Gu W, Wu Y, Cao JL, Xiao C, Zhou C (2020) Reversal of hyperactive subthalamic circuits differentially mitigates pain hypersensitivity phenotypes in parkinsonian mice. Proc Natl Acad Sci USA 117(18):10045–10054. https://doi.org/10.1073/pnas.1916263117
Velasco FC, Molina-Negro P, Bertrand C, Hardy J (1972) Further definition of the subthalamic target for arrest of tremor. J Neurosurg 36(2):184–191. https://doi.org/10.3171/jns.1972.36.2.0184
Shanker V (2019) Essential tremor: diagnosis and management. BMJ (Clinical research ed) 366:l4485. https://doi.org/10.1136/bmj.l4485
Mostofi A, Evans JM, Partington-Smith L, Yu K, Chen C, Silverdale MA (2019) Outcomes from deep brain stimulation targeting subthalamic nucleus and caudal zona incerta for Parkinson’s disease. NPJ Parkinson’s disease 5:17. https://doi.org/10.1038/s41531-019-0089-1
Ossowska K (2020) Zona incerta as a therapeutic target in Parkinson’s disease. J Neurol 267(3):591–606. https://doi.org/10.1007/s00415-019-09486-8
Plaha P, Ben-Shlomo Y, Patel NK, Gill SS (2006) Stimulation of the caudal zona incerta is superior to stimulation of the subthalamic nucleus in improving contralateral parkinsonism. Brain 129(Pt 7):1732–1747. https://doi.org/10.1093/brain/awl127
Plaha P, Khan S, Gill SS (2008) Bilateral stimulation of the caudal zona incerta nucleus for tremor control. J Neurol Neurosurg Psychiatry 79(5):504–513. https://doi.org/10.1136/jnnp.2006.112334
Lu CW, Harper DE, Askari A, Willsey MS, Vu PP, Schrepf AD, Harte SE, Patil PG (2021) Stimulation of zona incerta selectively modulates pain in humans. Sci Rep 11(1):8924. https://doi.org/10.1038/s41598-021-87873-w
de Groot A, van den Boom BJ, van Genderen RM, Coppens J, van Veldhuijzen J, Bos J, Hoedemaker H, Negrello M, Willuhn I, De Zeeuw CI, Hoogland TM (2020) NINscope, a versatile miniscope for multi-region circuit investigations. eLife 9. https://doi.org/10.7554/eLife.49987
Zhang C, Zhu H, Ni Z, Xin Q, Zhou T, Wu R, Gao G, Gao Z, Ma H, Li H, He M, Zhang J, Cheng H, Hu H (2021) Dynamics of a disinhibitory prefrontal microcircuit in controlling social competition. Neuron. https://doi.org/10.1016/j.neuron.2021.10.034
Acknowledgements
We thank the members of our lab for their support and help.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82271243, 82071235 and 82101318), Outstanding Youth Fund of Shaanxi Province (2021JC-32), Youth development programme funding (21QNPY065), support funding from the Fourth Military Medical University (2020AXJHHJ) and Sanming Project of Medicine in Shenzhen (SZSM201911011).
Author information
Authors and Affiliations
Contributions
JH and S-XW conceived and designed the experiments. J-QL, YB, S-HP performed experiments, analyzed data and wrote the manuscript. S-HP, Y-WZ, S-SG and Y-YZhu assisted J-QL with animal studies and histology. J-QL, J-YG and S-HP interpreted the data and statistical analyses. S-XW and JH conceived this study, analyzed the data and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the Institutional Animal Care and Use Committee of the Fourth Military Medical University and conformed to the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health.
Consent to Participate
Not applicable.
Consent for publication
Not applicable.
Competing Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Peng, S., Zhang, Y. et al. Glutamatergic Neurons in the Zona Incerta Modulate Pain and Itch Behaviors in Mice. Mol Neurobiol 60, 5866–5877 (2023). https://doi.org/10.1007/s12035-023-03431-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03431-7