Abstract
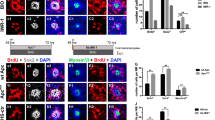
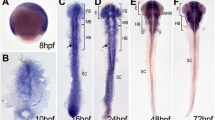
The regeneration of hair cells in zebrafish is a complex process involving the precise regulation of multiple signaling pathways, but this complicated regulatory network is not fully understood. Current research has primarily focused on finding molecules and pathways that can regulate hair cell regeneration and restore hair cell functions. Here, we show the role of N-Myc downstream regulated gene 2 (ndrg2) in zebrafish hair cell regeneration. We first found that ndrg2 was dynamically expressed in neuromasts of the developing zebrafish, and this expression was increased after neomycin-induced hair cell damage. Then, ndrg2 loss-of-function larvae showed reduced numbers of regenerated hair cells but increased numbers of supporting cells after neomycin exposure. By in situ hybridization, we further observed that ndrg2 loss of function resulted in the activation of Notch signaling and downregulation of atoh1a during hair cell regeneration in vivo. Additionally, blocking Notch signaling rescued the number of regenerated hair cells in ndrg2-deficient larvae. Together, this study provides evidence for the role of ndrg2 in regulating hair cell regeneration in zebrafish neuromasts.






Similar content being viewed by others
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Draf C, Wyrick T, Chavez E, Pak K, Kurabi A, Leichtle A, Dazert S, Ryan AF (2021) A screen of autophagy compounds implicates the proteasome in mammalian aminoglycoside-induced hair cell damage. Front Cell Dev Biol 9:762751
Han E, Lee DH, Park S, Rah YC, Park HC, Choi JW, Choi J (2022) Noise-induced hearing loss in zebrafish model: characterization of tonotopy and sex-based differences. Hear Res 418:108485
Lin FR, Yaffe K, Xia J, Xue QL, Harris TB, Purchase-Helzner E, Satterfield S, Ayonayon HN, Ferrucci L, Simonsick EM, Health ABCSG (2013) Hearing loss and cognitive decline in older adults. JAMA Intern Med 173:293–299
AitRaise I, Amalou G, Bousfiha A, Charoute H, Rouba H, Abdelghaffar H, Bonnet C, Petit C, Barakat A (2022) Genetic heterogeneity in GJB2, COL4A3, ATP6V1B1 and EDNRB variants detected among hearing impaired families in Morocco. Mol Biol Rep
Chen Y, Zhang S, Chai R, Li H (2019) Hair cell regeneration. Adv Exp Med Biol 1130:1–16
Adler HJ, Raphael Y (1996) New hair cells arise from supporting cell conversion in the acoustically damaged chick inner ear. Neurosci Lett 205:17–20
Balak KJ, Corwin JT, Jones JE (1990) Regenerated hair cells can originate from supporting cell progeny: evidence from phototoxicity and laser ablation experiments in the lateral line system. J Neurosci 10:2502–2512
Hardy K, Amariutei AE, De Faveri F, Hendry A, Marcotti W, Ceriani F (2021) Functional development and regeneration of hair cells in the zebrafish lateral line. J Physiol 599:3913–3936
Cruz IA, Kappedal R, Mackenzie SM, Hailey DW, Hoffman TL, Schilling TF, Raible DW (2015) Robust regeneration of adult zebrafish lateral line hair cells reflects continued precursor pool maintenance. Dev Biol 402:229–238
Kniss JS, Jiang L, Piotrowski T (2016) Insights into sensory hair cell regeneration from the zebrafish lateral line. Curr Opin Genet Dev 40:32–40
Millimaki BB, Sweet EM, Riley BB (2010) Sox2 is required for maintenance and regeneration, but not initial development, of hair cells in the zebrafish inner ear. Dev Biol 338:262–269
Romero-Carvajal A, Navajas Acedo J, Jiang L, Kozlovskaja-Gumbriene A, Alexander R, Li H, Piotrowski T (2015) Regeneration of sensory hair cells requires localized interactions between the Notch and Wnt pathways. Dev Cell 34:267–282
Baek S, Tran NTT, Diaz DC, Tsai YY, Acedo JN, Lush ME, Piotrowski T (2022) Single-cell transcriptome analysis reveals three sequential phases of gene expression during zebrafish sensory hair cell regeneration. Dev Cell 57(799–819):e796
Jiang L, Romero-Carvajal A, Haug JS, Seidel CW, Piotrowski T (2014) Gene-expression analysis of hair cell regeneration in the zebrafish lateral line. Proc Natl Acad Sci U S A 111:E1383-1392
Steiner AB, Kim T, Cabot V, Hudspeth AJ (2014) Dynamic gene expression by putative hair-cell progenitors during regeneration in the zebrafish lateral line. Proc Natl Acad Sci U S A 111:E1393-1401
Tang D, He Y, Li W, Li H (2019) Wnt/beta-catenin interacts with the FGF pathway to promote proliferation and regenerative cell proliferation in the zebrafish lateral line neuromast. Exp Mol Med 51:1–16
Lopez-Schier H, Hudspeth AJ (2006) A two-step mechanism underlies the planar polarization of regenerating sensory hair cells. Proc Natl Acad Sci U S A 103:18615–18620
Zhang Q, Li S, Wong HC, He XJ, Beirl A, Petralia RS, Wang YX, Kindt KS (2018) Synaptically silent sensory hair cells in zebrafish are recruited after damage. Nat Commun 9:1388
Liao H, Wu Z, Huang X, Qiu Z, Wu H (2015) NMyc downstream-regulated gene 2 suppresses proliferation and induces oncosis of OSRC2 human renal cancer cells. Mol Med Rep 11:1240–1245
Liu L, Shi M, Wang L, Hou S, Wu Z, Zhao G, Deng Y (2012) Ndrg2 expression in neurogenic germinal zones of embryonic and postnatal mouse brain. J Mol Histol 43:27–35
Zhu H, Zhao J, Zhou W, Li H, Zhou R, Zhang L, Zhao H, Cao J, Zhu X, Hu H, Ma G, He L, Yao Z, Yao L, Guo X (2012) Ndrg2 regulates vertebral specification in differentiating somites. Dev Biol 369:308–318
Yang J, Li Y, Wu L, Zhang Z, Han T, Guo H, Jiang N, Tao K, Ti Z, Liu X, Yao L, Dou K (2010) NDRG2 in rat liver regeneration: role in proliferation and apoptosis. Wound Repair Regen 18:524–531
Choi SC, Kim KD, Kim JT, Kim JW, Yoon DY, Choe YK, Chang YS, Paik SG, Lim JS (2003) Expression and regulation of NDRG2 (N-myc downstream regulated gene 2) during the differentiation of dendritic cells. FEBS Lett 553:413–418
Foletta VC, Prior MJ, Stupka N, Carey K, Segal DH, Jones S, Swinton C, Martin S, Cameron-Smith D, Walder KR (2009) NDRG2, a novel regulator of myoblast proliferation, is regulated by anabolic and catabolic factors. J Physiol 587:1619–1634
Qian F, Wei G, Gao Y, Wang X, Gong J, Guo C, Wang X, Zhang X, Zhao J, Wang C, Xu M, Hu Y, Yin G, Kang J, Chai R, Xie G, Liu D (2022) Single-cell RNA-sequencing of zebrafish hair cells reveals novel genes potentially involved in hearing loss. Cell Mol Life Sci 79:385
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59:307–321
Meyers JR, MacDonald RB, Duggan A, Lenzi D, Standaert DG, Corwin JT, Corey DP (2003) Lighting up the senses: FM1-43 loading of sensory cells through nonselective ion channels. J Neurosci 23:4054–4065
Grotek B, Wehner D, Weidinger G (2013) Notch signaling coordinates cellular proliferation with differentiation during zebrafish fin regeneration. Development 140:1412–1423
Ma EY, Rubel EW, Raible DW (2008) Notch signaling regulates the extent of hair cell regeneration in the zebrafish lateral line. J Neurosci 28:2261–2273
Thomas ED, Raible DW (2019) Distinct progenitor populations mediate regeneration in the zebrafish lateral line. Elife 8
Sarrazin AF, Villablanca EJ, Nunez VA, Sandoval PC, Ghysen A, Allende ML (2006) Proneural gene requirement for hair cell differentiation in the zebrafish lateral line. Dev Biol 295:534–545
Diensthuber M, Stover T (2018) Strategies for a regenerative therapy of hearing loss. HNO 66:39–46
Chen Y, Gu Y, Li Y, Li GL, Chai R, Li W, Li H (2021) Generation of mature and functional hair cells by co-expression of Gfi1, Pou4f3, and Atoh1 in the postnatal mouse cochlea. Cell Rep 35:109016
Jia Y, Zhao Y, Kusakizako T, Wang Y, Pan C, Zhang Y, Nureki O, Hattori M, Yan Z (2020) TMC1 and TMC2 proteins are pore-forming subunits of mechanosensitive ion channels. Neuron 105(310–321):e313
Shu Y, Li W, Huang M, Quan YZ, Scheffer D, Tian C, Tao Y, Liu X, Hochedlinger K, Indzhykulian AA, Wang Z, Li H, Chen ZY (2019) Renewed proliferation in adult mouse cochlea and regeneration of hair cells. Nat Commun 10:5530
Tao L, Yu HV, Llamas J, Trecek T, Wang X, Stojanova Z, Groves AK, Segil N (2021) Enhancer decommissioning imposes an epigenetic barrier to sensory hair cell regeneration. Dev Cell 56(2471–2485):e2475
Ai R, Sun Y, Guo Z, Wei W, Zhou L, Liu F, Hendricks DT, Xu Y, Zhao X (2016) NDRG1 overexpression promotes the progression of esophageal squamous cell carcinoma through modulating Wnt signaling pathway. Cancer Biol Ther 17:943–954
Kim KR, Kim KA, Park JS, Jang JY, Choi Y, Lee HH, Lee DC, Park KC, et al (2020) Structural and biophysical analyses of human N-Myc downstream-regulated gene 3 (NDRG3) protein. Biomolecules 10
Nakahata S, Ichikawa T, Maneesaay P, Saito Y, Nagai K, Tamura T, Manachai N, Yamakawa N, Hamasaki M, Kitabayashi I, Arai Y, Kanai Y, Taki T, Abe T, Kiyonari H, Shimoda K, Ohshima K, Horii A, Shima H, Taniwaki M, Yamaguchi R, Morishita K (2014) Loss of NDRG2 expression activates PI3K-AKT signalling via PTEN phosphorylation in ATLL and other cancers. Nat Commun 5:3393
Shen L, Qu X, Ma Y, Zheng J, Chu D, Liu B, Li X, Wang M, Xu C, Liu N, Yao L, Zhang J (2014) Tumor suppressor NDRG2 tips the balance of oncogenic TGF-beta via EMT inhibition in colorectal cancer. Oncogenesis 3:e86
Zhao C, Ren Y, Zhang Y (2022) NDRG4 alleviates myocardial infarction-induced apoptosis through the JAK2/STAT3 pathway. Comput Math Methods Med 2022:4869470
Berger P, Sirkowski EE, Scherer SS, Suter U (2004) Expression analysis of the N-Myc downstream-regulated gene 1 indicates that myelinating Schwann cells are the primary disease target in hereditary motor and sensory neuropathy-Lom. Neurobiol Dis 17:290–299
King RH, Chandler D, Lopaticki S, Huang D, Blake J, Muddle JR, Kilpatrick T, Nourallah M, Miyata T, Okuda T, Carter KW, Hunter M, Angelicheva D, Morahan G, Kalaydjieva L (2011) Ndrg1 in development and maintenance of the myelin sheath. Neurobiol Dis 42:368–380
Okuda T, Higashi Y, Kokame K, Tanaka C, Kondoh H, Miyata T (2004) Ndrg1-deficient mice exhibit a progressive demyelinating disorder of peripheral nerves. Mol Cell Biol 24:3949–3956
Wibowo I, Pinto-Teixeira F, Satou C, Higashijima S, Lopez-Schier H (2011) Compartmentalized Notch signaling sustains epithelial mirror symmetry. Development 138:1143–1152
Driver EC, Sillers L, Coate TM, Rose MF, Kelley MW (2013) The Atoh1-lineage gives rise to hair cells and supporting cells within the mammalian cochlea. Dev Biol 376:86–98
Zak M, Klis SF, Grolman W (2015) The Wnt and Notch signalling pathways in the developing cochlea: formation of hair cells and induction of regenerative potential. Int J Dev Neurosci 47:247–258
Takeichi T, Takarada-Iemata M, Hashida K, Sudo H, Okuda T, Kokame K, Hatano T, Takanashi M, Funabe S, Hattori N, Kitamura O, Kitao Y, Hori O (2011) The effect of Ndrg2 expression on astroglial activation. Neurochem Int 59:21–27
M, W (2000) The zebrafish book. A guide for the laboratory use of zebrafish (Danio rerio). Univ. of Oregon Press, Eugene
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310
Xiao T, Roeser T, Staub W, Baier H (2005) A GFP-based genetic screen reveals mutations that disrupt the architecture of the zebrafish retinotectal projection. Development 132:2955–2967
Bernardos RL, Raymond PA (2006) GFAP transgenic zebrafish. Gene Expr Patterns 6:1007–1013
Sato T, Takahoko M, Okamoto H (2006) HuC:Kaede, a useful tool to label neural morphologies in networks in vivo. Genesis 44:136–142
Huo L, Jiao L, Chen L, Yu Z, Hutvagner G, Li J (2021) Single-cell multi-omics sequencing: application trends, COVID-19, data analysis issues and prospects. Brief Bioinform 22:bbab229
Shi J, Fok KL, Dai P, Qiao F, Zhang M, Liu H, Sang M, Ye M, Liu Y, Zhou Y, Wang C, Sun F, Xie G, Chen H (2021) Spatio-temporal landscape of mouse epididymal cells and specific mitochondria-rich segments defined by large-scale single-cell RNA-seq. Cell Discov 7:34
Yu G, Wang L, Han Y, He Q (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287
Wang X, Ling CC, Li L, Qin Y, Qi J, Liu X, You B, Shi Y, Zhang J, Jiang Q, Xu H, Sun C, You Y, Chai R, Liu D (2016) MicroRNA-10a/10b represses a novel target gene mib1 to regulate angiogenesis. Cardiovasc Res 110:140–150
Qi J, Dong Z, Shi Y, Wang X, Qin Y, Wang Y, Liu D (2016) NgAgo-based fabp11a gene knockdown causes eye developmental defects in zebrafish. Cell Res 26:1349–1352
He Y, Tang D, Cai C, Chai R, Li H (2016) LSD1 is required for hair cell regeneration in zebrafish. Mol Neurobiol 53:2421–2434
Gong J, Wang X, Zhu C, Dong X, Zhang Q, Wang X, Duan X, Qian F, Shi Y, Gao Y, Zhao Q, Chai R, Liu D (2017) Insm1a regulates motor neuron development in zebrafish. Front Mol Neurosci 10:274
Harris JA, Cheng AG, Cunningham LL, MacDonald G, Raible DW, Rubel EW (2003) Neomycin-induced hair cell death and rapid regeneration in the lateral line of zebrafish (Danio rerio). J Assoc Res Otolaryngol 4:219–234
Funding
This work was supported by grants from the National Natural Science Foundation of China (Nos. 82192860, 82192862, 81830029, 81970879), the Natural Science Foundation of China (2018YFA0801004), the Shanghai Science and Technology Committee (STCSM) Science and Technology Innovation Program (No. 20MC1920200), and the Research Projects of the Shanghai Municipal Health Committee (2020YJZX0110, 2022XD059).
Author information
Authors and Affiliations
Contributions
All authors made a significant contribution to the work reported, whether in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; agreed on the journal to which the article has been submitted; and agreed to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Ethics Approval
All animal procedures were carried out in accordance with guidelines established by the Institutional Animal Care and Use Committee of Fudan University.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Gu, X., Wang, C. et al. Loss of ndrg2 Function Is Involved in Notch Activation in Neuromast Hair Cell Regeneration in Zebrafish. Mol Neurobiol 60, 3100–3112 (2023). https://doi.org/10.1007/s12035-023-03262-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03262-6